1
EEG Monitoring in Neonatal Epilepsies
Electroencephalographers often approach neonatal studies with trepidation. Neonatal studies vary from traditional electroencephalograms (EEGs) in both technical and visual aspects. Half of the full electrode set is used, placed at double distance, and the recording lasts for 60 minutes in order to catch a full sleep-wake cycle. Extra electrodes are also essential for interpreting the recording, such as ocular leads, chin electromyogram (EMG), and cardiac and respiratory monitoring. When interpreting the neonatal EEG, the paper speed is slowed to 15 mm/sec in order to more easily recognize the slower delta frequencies, which dominate in neonatal records. The low-frequency filter is set to 0.5 Hz in order to clearly interpret slow eye movements (1). Sensitivity is often lowered below the standard 7 mv/sec, given that amplitudes are not as high and scalp impedance is lower. Although these differences exist, with experience and knowledge of these EEG differences, interpretation in this age group is readily accomplished.
Much of the trepidation associated with the interpretation of neonatal EEG stems from the fact that “normal” background is somewhat of a moving target. Findings that are acceptable at 30 weeks conceptional age (CA) are grossly abnormal at 36 weeks. Therefore, neonatal EEG is best interpreted by first noting the infant’s current CA and then recognizing the characteristics that should be present in the EEG background of a normal neonate. CA is calculated by adding the estimated gestational age at birth to the current chronologic age (in weeks). If not given the correct gestational age, an age range can be estimated based on recognized patterns.
NEONATAL EEG BACKGROUND
Neonatal EEG studies should be systematically evaluated, with interpretation phrased in terms of several key features:
![]() Continuity
Continuity
![]() Amplitude
Amplitude
![]() Symmetry
Symmetry
![]() Interhemispheric synchrony
Interhemispheric synchrony
![]() Normal named patterns
Normal named patterns
In extreme prematurity, normal electrographic findings are typically discontinuous, with bursts of continuous cerebral activity separated by intervals of relative quiescence and lower amplitude. This discontinuity improves with age, with the interburst interval becoming progressively shorter and higher in amplitude as the baby approaches full term. By 40 to 44 weeks CA, the EEG background becomes continuous in both wake and sleep (2).
Differentiation between wake and sleep states initially appears around 30 weeks CA. By definition, the infant is awake whenever his/her eyes are open and asleep when eyes are closed. Sleep is further subdivided into active sleep (AS, characterized by irregular respirations, occasional limb movements, and rapid horizontal eye movements) and quiet sleep (QS), characterized by deep, regular respirations and paucity of limb/trunk movement. Electrographically, wakefulness and AS in infants more than 30 weeks CA demonstrate fairly continuous cerebral activity, developing into a characteristic mixed frequency, moderate-amplitude activité moyenne pattern.
Because neonatal background abnormalities may become most apparent during deeper sleep stages, a complete assessment of the EEG background requires thorough evaluation of QS. To this end, continuous EEG (cEEG) provides a significant advantage over routine EEG in ensuring that a generous sample of QS is captured for review. As the invariant, nonreactive pattern of burst suppression seen in extremely preterm infants transitions into more defined wake-sleep stages around 30 weeks CA, the final remnants of EEG discontinuity linger in QS. As development proceeds, QS discontinuity gradually resolves, with gradual improvement in the duration and amplitude of the interburst activity. Between 30 and 32 weeks CA, QS activity consists of a tracé discontinue pattern in which periods of cerebral activity are separated by nearly isoelectric periods of quiescence with voltage less than 25 µV. With time, the voltage of the interburst intervals gradually increases such that by 35 to 36 weeks CA, QS typically transitions to a tracé alternant pattern, in which cerebral activity is consistently maintained above 25 µV but cycles between higher-amplitude bursts and more quiescent periods. The interburst amplitude continues to increase until no periods of relative quiescence are perceived, and a continuous slow-wave sleep pattern is fully established around 44 weeks CA (3,4).
Bursts of activity appearing in one hemisphere within 1.5 seconds of the other hemisphere are considered to be synchronous. Prior to 30 weeks CA, cerebral activity occurs nearly simultaneously in both the right and left hemispheres, a phenomenon described as a hypersynchrony (5). The reason for early interhemispheric hypersynchrony is unknown, though it has been postulated to be related to prominent thalamic drivers without significant cortical input. Following 30 weeks, occasional asynchronous bursts are seen, which progressively diminish until 100% synchrony is reestablished around 37 weeks CA.
BACKGROUND PATTERNS
A. Excessive sharps
B. Excessive discontinuity
C. Brief ictal/interictal
rhythmic/repetitive discharges (BIRDs)
D. Other patterns (depressed/undifferentiated, low voltage)
EEG background findings (Table 1.1) are also frequently employed to assess the functional integrity of the neonatal brain and to aid in the evaluation of neurologic prognosis. At the same time, however, many patterns are nonspecific and of uncertain clinical significance.
TABLE 1.1 EEG Background in Prematurity
| CONCEPTIONAL AGE (WEEKS) | MAXIMUM INTERBURST DURATION (SEC) | EEG BACKGROUND FEATURES |
| 24–25 | 60 | No sleep organization or reactivity |
| 27–30 | 35 | Discontinuous in both wake and sleep, some reactivity |
| 31–33 | 20 | Differentiation between active and quiet sleep patterns Wake and active sleep: mixed frequency continuous (activité moyenne) Quiet sleep: interburst intervals amplitude nearly isoelectric, <25 µV (trace discontinue pattern) |
| 34–36 | 10 | Wake and active sleep: mixed frequency continuous (activité moyenne) Quiet sleep: Interburst intervals increase in amplitude, eventually exceeding 25 µV (trace alternant pattern) |
| 37–40 | 6 | Wake and active sleep: mixed frequency continuous (activité moyenne) Quiet sleep: Interburst intervals continue to increase in amplitude, increasing continuity (trace alternant) transitioning to continuous slow-wave sleep pattern |
Source: Adapted from Refs. 5, 8, 9.
Excessive Sharps
Temporal sharp transients are normally seen during sleep in the term neonate, are often bilateral and asynchronous, and should be surface negative in polarity. If they occur in runs, are unilateral, or appear in wakefulness, they are more likely to be considered abnormal. Sharp waves occurring outside of the temporal or centrotemporal regions would also be considered abnormal. No official criteria exist in which temporal sharps are defined as excessive, and it has been proposed that greater than 13 over the course of a 60-minute recording in a term neonate would be considered excessive [criteria adapted from (6,7)].
Excessive Discontinuity
In the term neonate, periods of attenuation during QS should not exceed 2 to 4 seconds in duration. Interburst intervals longer than this are considered excessively discontinuous. This pattern can be associated with dysmaturity or incorrect gestational dating but can also be a nonspecific marker for neonatal encephalopathy.
Brief Ictal/Interictal Rhythmic/Repetitive Discharges
First described by Shewmon in 1990, this pattern is considered interictal but on the ictal spectrum. It usually occurs in the context of electrographic seizures and is characterized by a run of epileptiform discharges with evolution but lasting less than 10 seconds. Their clinical significance is not yet completely understood, but given their presence in neonates with seizures, they may be associated with neurologic morbidity.
Depressed/Undifferentiated or Low Voltage
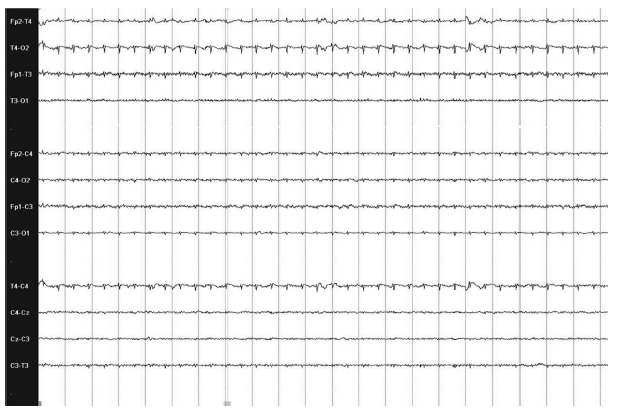
A depressed and undifferentiated pattern (Figure 1.1) is most commonly associated with severe underlying neurologic injury to the cortical generators of electrocerebral activity. Low voltage is considered to be background activity persistently less than 10 µV without normal background features. The recording will also show poor reactivity, no alteration in frequencies with external stimulation, and no sleep-wake cycling.
SEIZURE DETECTION
Seizure is the most common neurologic disorder in the neonatal period. There are numerous potential etiologies for neonatal seizures, and timing of presentation as well as electrographic findings can be of potential use in elucidating their etiology. Seizures can be transient due to an acute injury, markers of an underlying genetic or metabolic disorder, or signs of an underlying structural abnormality.
EEG evaluation and confirmation of seizure activity is particularly important in the neonatal population, given the high rate of subclinical or subtly clinical seizures and because newborns may often have unusual movements that can be mistaken for seizure activity. For instance, a systematic video review of 526 electrographic seizures in nine infants revealed that only 34% of seizures were associated with clinical manifestations, and only 27% of these clinical seizures (9% of overall seizures) were recognized by nursing staff. Of more concern, 73% of “seizures” documented by the neonatal intensive care unit (NICU) nursing staff were not epileptic seizures. Rather, the events marked by NICU nursing were not epileptic in nature. Instead, these movements commonly consisted of likely nonepileptic events such as jitteriness, mouthing, and fisting (10). Therefore neonatal seizure quantification solely by clinical observation is plagued by both high false-positive and high false-negative rates. To ensure an accurate assessment of seizure detection and treatment response, EEG monitoring is essential.
FIGURE 1.1 A 38-week-old baby boy born via emergency Caesarean-section (C-section) for polyhydramnios and nonreassuring fetal heart tracings with severe hypoxic-ischemic encephalopathy. Background shows low voltage (<10 µV) without reactivity.
Subclinical Seizures
EEG confirmation of seizure cessation following anticonvulsant treatment is also recommended. Neonates are particularly vulnerable to the phenomenon of electroclinical uncoupling, in which clinical evidence of seizure activity ceases, following the administration of seizure medications, while subclinical electrographic seizure activity continues unabated. Although subclinical seizures are known to occur in critically ill children and adults (11,12), features of chloride homeostasis unique to the immature brain contribute to a high likelihood of electroclinical uncoupling. The potassium-chloride cotransporter (KCC2), which is the predominant type of chloride channel in the adult brain, transport chloride ions outside of neurons and have a hyperpolarizing effect. In contrast, the predominant chloride channel in the immature brain is the sodium-potassium-chloride cotransporter (NKCC1), which transports chloride ions into neurons and has a depolarizing effect. Gamma aminobutyric acid (GABA), a neurotransmitter that activates chloride channels, can therefore have a paradoxically excitatory effect in developing neurons due to the predominance of NKCC1 channels (13). Because the transition from NKCC1 to KCC2 chloride channels occurs in a caudal-to-rostral progression, GABA initially becomes inhibitory in subcortical structures such as the brainstem and basal ganglia while remaining excitatory in the cortex. Commonly used medications such as phenobarbital, which exert their effects through GABA agonist activity, may therefore suppress brainstem motor output, while allowing electrographic seizure activity to continue in the cortex.
The high risk of subclinical seizures has been well documented in the NICU population (14–17). For instance, cEEG monitoring of neonates randomized to initial treatment with either phenobarbital or phenytoin demonstrated that while 24 of 50 infants responded completely to the first seizure medication administered, 15 of the remaining 26 neonates (58%) demonstrated electroclinical uncoupling, with suppression of clinical seizure activity during all or the majority of posttreatment electrographic seizures (18).
Neonatal Seizure Semiology
Seizure semiology in the newborn is variable but can be grouped into the following categories: clonic, tonic, and myoclonic (Table 1.2). These are focal, repetitive, and cannot be suppressed by the examiner. Due to incomplete myelination, infants cannot generate generalized tonic-clonic seizures, but they can have multifocal seizures that can appear generalized to the untrained or inexperienced examiner. Infants can also have generalized epileptic spasms that are hypothesized to be more subcortically driven.
Because infants often have repetitive movements which can be difficult to interpret, EEG is often relied upon to distinguish stereotyped or rhythmic movements as epileptic or nonepileptic. Oral automatisms, bicycling, roving eye movements, and other nonrhythmic but repetitive movements are often seen in critically ill infants. Without clear electrographic correlate, these had been previously termed clinical only seizures, but are now more commonly presumed to be nonepileptic in nature. These movements tend to occur more often in encephalopathic infants and are also associated with poor prognosis (19).
TABLE 1.2 Neonatal Seizure Types
| MOVEMENT TYPE | LOCALIZATION/CLINICAL | ELECTROGRAPHIC CORRELATE |
| Clonic | Focal rhythmic jerking of an extremity Nonsuppressible | Yes |
| Tonic | Focal sustained extension or flexion of an extremity Not able to overcome with external manipulation Sustained extension of the whole body | Yes Not usually |
| Myoclonic | Single jerk or multiple nonrhythmic jerks of an extremity | Usually |
| Spasms | Focal or generalized Flexor, extensor, or mixed flexor-extensor | Yes |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree