12 Endovascular Interventions for Skull Base Lesions The management of skull base lesions has undergone evolution in the last two decades thanks to advances in endovascular techniques to either help manage these lesions before or after open surgery, or as an alternative approach to an open procedure. • Interventional neuroradiology (INR) is a branch of interventional radiology that encompasses image-guided procedures involving the head, neck, and spine. The primary applications of endovascular techniques in the management of skull base lesions are embolization of lesions (e.g., skull base tumors and fistulas), balloon test occlusion, vessel sacrifice, and inferior petrosal sinus sampling. Embolization is a procedure to obliterate the vascular supply to a lesion. Since the first treatment of a posttraumatic carotid cavernous fistula in the 1930s, by placing a long muscle fragment marked with a clip in the internal carotid artery (Brooks’s1 technique), a number of studies on embolization have explored the value of presurgical tumor embolization, its cost-efficiency, and its ability to decrease surgical blood loss and to reduce operative and recovery time.2–9 A thorough understanding of the vascular anatomy is mandatory in all endovascular procedures. Awareness of normal anatomy of the blood vessels, as well as of the anatomic variants, the collateral pathways, and anastomoses between the extracranial and intracranial vessels, is important to ensure safe and successful treatment.10,11 Knowledge of the anastomoses, such as among the internal carotid artery (ICA), vertebral artery (VA), and external carotid artery (ECA) branches, as well as the arterial supply to the cranial nerves, is essential for neurointerventional procedures. In 1970, Charles Dotter’s group reported the first use of occlusive embolic particles to treat arterial bleeding.3 An autologous blood clot was delivered endovascularly via superselective catheter injection in order to control acute upper gastrointestinal bleeding. Since then, a plethora of occlusive, prothrombotic materials have been used in the endovascular treatment of arterial bleeding (including fascial strips and silk threads, which have now been replaced in clinical use by more modern products). • The new generation of embolic agents can be classified as permanent or temporary (enabling recanalization over time).12 Embolic agents can also be classified according to their physical properties: mechanical devices, particles, and liquid (Table 12.1). • Balloon: Inflatable balloons are used for the obliteration of large vessels, such as the carotid and vertebral arteries.13–16 • Coil: There are several types of coils (Table 12.1). The most commonly used coils, the detachable coils, enable accurate positioning before permanent deployment. Gelfoam is one of the most commonly used particles. • Gelfoam is used in either powder form (40 to 60 μm) or in solid form as strips. The strips are cut down to a size of a few millimeters and then are injected under pressure into the target vessel, often referred to as “torpedoes.” • The ability of Gelfoam to expand with hydration is one of its key advantages. • Used in most embolization after the use of Gelfoam, it offers temporary proximal arterial control, which enables the option of future access. Polyvinyl alcohol (PVA) comes in a variety of sizes (45–150 to 1,000–1,180 μm). • Cerebral angiography might not demonstrate all the small arterious micro-anastomoses, especially for vessels ranging from 50 to 80 μm in size.17 Knowledge of the local microanatomy is important in choosing the appropriate-sized PVA particles. With the use of Gelfoam or PVA, the vessel can recanalize over time (faster with the Gelfoam than with PVA). Thus, these materials are often used in the presurgical embolization of lesions, when definitive surgical treatment is planned within 24 to 72 hours.18 Cyanoacrylates (also called “glue”) and precipitated polymers are the most common liquid embolic agents used. Glue has classically been considered an effective and permanent agent for the occlusion of vascular lesions.19 INR Pitfalls The durability and permanence of glue has been called into question.20 Further complicating the use of glue in embolization procedures is the fact that glue is technically challenging to deliver into the cerebral circulation, and its use requires experience because polymerization of glue starts on contact with anions in the blood.21 Although this is somewhat altered by formulation with Ethiodol, it still poses a technical challenge. Rapid polymerization in a microcatheter can also cause glue to adhere to the microcatheter prior to its removal, with the risk of catheter breakage. Furthermore, a lesion treated with glue will often become less pliable and stiffer, making a subsequent surgical excision more difficult.22 • Precipitated polymers. Onyx (ev3, Irvine, CA), a precipitated polymer (also known as nonadhesive liquid embolic agents), is a newer embolic agent that has become an alternative to glue. • Neurologic • Nonneurologic32 The most common skull base–related embolized tumors are paragangliomas (glomus tumors), meningiomas, and angiofibromas.26,27,33 Less common are metastases, esthesioneurorablastomas, schwannomas, chordomas, and hemangiopericytomas.27 • Timing: Tumor embolization is usually performed 24 to 72 hours prior to surgery, although this a matter of debate. This timing is believed to enable maximal thrombosis of the vessels prior to recanalization.4–6,27 In meningioma, delaying surgery up to 7 to 9 days has been reported; it enables tumor softening and minimizes blood loss.34,35 This has not been routinely observed in all series, with the risk of confusing the grade and even overgrading due to findings of necrosis and enlarged nuclei, as found in embolized meningiomas.36 Although meningiomas are common, preoperative embolization is rarely done for convexity lesions whose blood supply can be easily taken at resection. Skull base tumors such as clival meningiomas have a deep blood supply (usually off the ICA) that is difficult to access; endovascular access to the feeding pedicle (usually the meningohypophyseal trunk) is often difficult, and embolic material can reflux into the ICA.37 • Olfactory groove meningiomas, which are commonly supplied by the anterior ethmoidal artery, cannot be embolized due to the risk of inadvertent introduction of embolic material into the ophthalmic artery via anastomotic connections.38 • Cavernous and parasellar meningiomas receive blood supply from the meningohypophyseal trunk and the inferolateral trunk; these vessels can be difficult to embolize and require surgeons with extensive experience.39,40 ICA-supplied skull base meningiomas have been embolized with a good outcome.41 • Some requirements for preoperative embolization have been elaborated42: • Regarding the outcome after embolization, complete tumor devascularization has been described as beneficial in many studies.37,43 • Regarding the choice of embolic agents, PVA is most commonly used. Glue is the best known agent, and it offers good result. It enables occlusion of feeders and intratumoral vessels, which prevent intratumoral bleeding.44 Onyx has been reported in few cases and no definitive conclusions may be drawn yet.45 Paragangliomas are highly vascular, and embolization is frequently performed prior to surgery.46–49 • In some cases, paragangliomas may secrete vasoactive substances. Release of vasoactive mediators can occur during angiography and lead to a hypertensive crisis. • Direct percutaneous puncture of the tumor can be performed when arterial feeders are too small to navigate from a peripheral access point (such as from femoral or radial access) or when the transarterial approach carries a high risk.26,50,51 • Facial nerve palsy has been reported after embolization of paragangliomas. Although some reports show less injury after embolization, this is not a consistent finding.52,53 • Decreased extent of the procedure and less blood loss have been observed after embolization.48,53,54 Blood loss dropped from 599 to 263 mL with embolization, and simple excision was achieved in 97% versus 82% with an estimated mean devascularization of 76%.54 Juvenile angiofibromas (JAFs) are rare lesions, accounting for 0.05% of all head and neck neoplasms.55 Although benign, JAFs can be locally invasive into the nasopharynx, and even intracranial extension is possible. They often arise in the sphenopalatine fossa.23 The precise origin can be difficult to determine, as they present commonly with considerable size. It is commonly agreed that the posterior choanal tissue is the origin of the JAF. Angiography is essential prior to surgery for two reasons: • To obtain detailed vessels architecture: • To provide treatment of embolization followed by complete surgical removal6: Vascular lesions of the skull base region encompass a variety of types, including CCF, dural arteriovenous fistula (dAVF), and AVM. Other vascular lesions, which are mostly low-flow lesions, include capillary, capillarovenous, venous, or lymphatic malformations.25–27,62,63 Carotid cavernous fistula can be classified using the Barrow classification64: • Type A: direct (high flow) between the carotid artery and the cavernous sinus (CS) (spontaneous intracavernous aneurysm rupture or traumatic) • Indirect (low flow) dural shunt (spontaneous) Urgent treatment is usually required in direct CCFs, as they are high flow and often present with vision loss, high intraocular pressure, and even venous re-flux into the cortical veins.65 • Endovascular repair has become the preferred strategy for CCF treatment; specifically, the transarterial approach is usually employed for direct CCFs and transarterial/transvenous approaches for indirect CCFs. • Detachable balloons, coils, PVA, liquid embolic agents, and stents have all been used in the treatment of CCFs, with the goal of closing the fistula while preserving flow within the ICA.66–71 • For indirect CCFs, the favored approach is transvenous embolization, starting from the femoral or jugular veins. Indirect CCFs have also been treated via direct canalization of the ophthalmic vein. The goal is to access the cavernous sinus and then pack the appropriate part with coil to shut down the shunting.72–76 CCFs supplied by the ICA branches are particularly thought to be more safely treated with transvenous embolization.77 • Transarterial embolization is possible for accessible feeders, especially those from the ECA. Once again, coils, PVA, or liquid embolic agents can also be used. • Up to a 98% rate of successful occlusion of direct CCF has been achieved in countries where the detachable balloon method is available. In institutions where series included balloon and other devices, the rate was between 88 and 94%.71,78 The largest series on indirect CCF reported a 90% success rate using the transvenous approach.78,79 As classified by Borden et al or Cognard et al (see Table 25.5, page 695), a dAVF is a shunt between extracranial (and meningeal) arteries and the dural venous sinus, dural veins, or cortical veins. Although some institutions consider open surgery as the best treatment, the advancements in endovascular treatment, with new devices, embolic agents, and catheters, have made it a valid and proven option for dAVF. The goal, which correlates with the surgical disconnection, is the obliteration of the fistulous connection. The vein is the known recruiting source, and obliteration has to include either both sides or at least the venous side. • Complete obliteration will result in cure.80–82 Incomplete embolization enables new arterial supply to form and necessitates a second endovascular attempt or a new approach, such as open surgery. If neurosurgical excision is advocated, the initial embolization provides partial devascularization and decreased bleeding, which can be significant and rapid in dAVF (including just incising the scalp).83 • Approaches to dAVF embolization can be divided into transarterial or transvenous, or both. Of note, when femoral transarterial or transvenous treatment is impossible or unsuccessful, options include direct access either by direct puncture (carotid), cutdown to expose vessels (superior ophthalmic vein to access cavernous sinus), or even through a bur hole position on the known cortical drainage vein.84–86 • Some factors have been shown to improve the cure rate: • Success rates depend on the type of embolic agent used—glue versus Onyx: Provocative testing is performed in an attempt to predict if a clinical deficit will appear following a vessel occlusion or sacrifice. Balloon test occlusion (BTO) of the ICA is a relatively common endovascular procedure to assess cerebrovascular reserve. • Balloon test occlusion incorporates multiple methods of clinical and radiographic evaluation. It requires: Temporary occlusion of a vessel is a well-accepted evaluation prior to surgery in which arterial sacrifice or prolonged temporary occlusion is considered.99 The following factors must be taken into consideration22: • Whenever possible, the vessel must be temporarily occluded at the level that would simulate the proposed sacrifice. • Neurologic testing should evaluate the possible consequences of the proposed vascular occlusion. There are several techniques for performing a BTO. One of the most common techniques is as follows: • A neurologic exam is completed prior to the test. Two or more days before the test a baseline single photon emission computed tomography (SPECT) study is performed to assess cerebral perfusion as a control. • Bilateral femoral arterial access is achieved (one side will be used during inflation for repeat angiography) with the patient awake. • The patient is heparinized to an activated clotting time (ACT) goal of above 300; at this level, clot formation decreases. • After inflation of the balloon and confirmation of proper occlusion with contrast stagnation, the angiogram is repeated in the contralateral ICA and dominant VA. The neurologic assessment should be reviewed with the patient prior to the procedure and the inflation, and the patient is examined continuously during occlusion. • Twenty-five minutes postocclusion, the radiotracer is administered intravenously and at the 30-minute mark the balloon is deflated and significant backflow is performed to minimize the risk of thrombus embolization; for extra safety an aspiration can be performed through the catheter. • The heparin is reversed, the sheaths are withdrawn, and hemostasis is achieved by manual compression. If at any moment, the patient’s clinical status changes, the procedure is aborted and this is considered a failed BTO. On the angiographic study, an important component is the timing of the venous delay: • The appearance of the first cortical vein on the injected side is the beginning of the venous phase. Images are reviewed, and one of the considerations for vessel sacrifice acceptance is when the delay between the appearance of the first cortical vein on the injected side and the contralateral and occluded hemisphere is no longer than 2 seconds.100,101 There are number of adjunctive tests that can be performed to improve either the clinical or imaging aspect: • Neurologic function. These tests either assess neurologic function or are used to increase the accuracy of the evaluation. • Cerebral blood flow
 Embolization of Skull Base Lesions
Embolization of Skull Base Lesions
General Principles
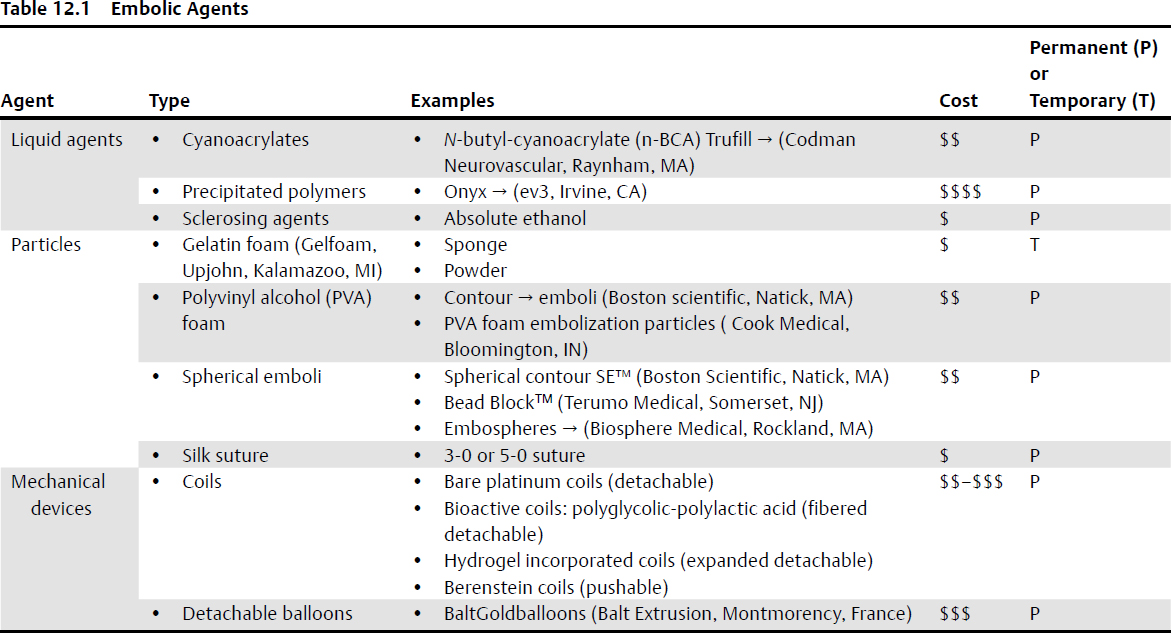
Embolic agents
Mechanical Devices
 Detachable balloons (Balt, Goldballoons, Balt Extrusion, Montmorency, France) are not currently Food and Drug Administration (FDA) approved, but are available in most countries, as of August 2014.
Detachable balloons (Balt, Goldballoons, Balt Extrusion, Montmorency, France) are not currently Food and Drug Administration (FDA) approved, but are available in most countries, as of August 2014.
 Care must be used when employing detachable balloons in the cerebral circulation, as they have the potential risk of spontaneously deflating over time or rupturing once deployed, and then embolizing into the distal arterial circulation.
Care must be used when employing detachable balloons in the cerebral circulation, as they have the potential risk of spontaneously deflating over time or rupturing once deployed, and then embolizing into the distal arterial circulation.
 Although widely available, coils are not usually the preferred tool in the embolization process. They enable a more proximal occlusion of the blood supply and are better for large vessel occlusion. But for tumors, coils block access for future embolization through the same vessel, which makes it not necessarily the best option.
Although widely available, coils are not usually the preferred tool in the embolization process. They enable a more proximal occlusion of the blood supply and are better for large vessel occlusion. But for tumors, coils block access for future embolization through the same vessel, which makes it not necessarily the best option.
Particles
Liquids
 Advantages of Onyx: It is nonadhesive, and although physically occupying spaces within the vasculature, it does not adhere to the vessel walls. This enables longer injections than with glue, and the ability to control the rate of embolization. Onyx can be deployed more slowly than glue, and often entails a more controlled delivery. For instance, injection of Onyx can be stopped, an angiogram injection performed, and then a decision regarding more embolization can be made. This can all be done with significantly less risk of embedding the catheter in a vessel than with the use of glue.21
Advantages of Onyx: It is nonadhesive, and although physically occupying spaces within the vasculature, it does not adhere to the vessel walls. This enables longer injections than with glue, and the ability to control the rate of embolization. Onyx can be deployed more slowly than glue, and often entails a more controlled delivery. For instance, injection of Onyx can be stopped, an angiogram injection performed, and then a decision regarding more embolization can be made. This can all be done with significantly less risk of embedding the catheter in a vessel than with the use of glue.21
 Disadvantages of Onyx: the need to flush the catheter in vivo with dimethyl sulfoxide (DMSO), which is potentially toxic to the endothelium, painful in awake patients, and requires a DMSO-compatible catheter. Onyx also produces a rather pungent smell of garlic on the patient’s breath.
Disadvantages of Onyx: the need to flush the catheter in vivo with dimethyl sulfoxide (DMSO), which is potentially toxic to the endothelium, painful in awake patients, and requires a DMSO-compatible catheter. Onyx also produces a rather pungent smell of garlic on the patient’s breath.
Indications for and Goals of Embolization
 Prior to surgery (head and neck surgeries)
Prior to surgery (head and neck surgeries)
 Adjuvant therapy (stabilize or reduce inoperable tumors, epistaxis)
Adjuvant therapy (stabilize or reduce inoperable tumors, epistaxis)
 Dural arteriovenous fistula
Dural arteriovenous fistula
 Relief of symptoms (pain) in palliative situation
Relief of symptoms (pain) in palliative situation
 Recurrent nonsurgical lesions
Recurrent nonsurgical lesions
 Devascularize lesion
Devascularize lesion
 Occlude surgically inaccessible blood supply
Occlude surgically inaccessible blood supply
 Sacrifice large vessels (endovascular approach enables prior testing before occlusion)
Sacrifice large vessels (endovascular approach enables prior testing before occlusion)
 Facilitate surgical access (e.g., allowing decrease blood loss at skin incision)
Facilitate surgical access (e.g., allowing decrease blood loss at skin incision)
Complications of Embolization
 Vessel injury with dissection or rupture, which may cause subarachnoid, subdural, or intracerebral hemorrhage
Vessel injury with dissection or rupture, which may cause subarachnoid, subdural, or intracerebral hemorrhage
 Frequency: The N-Butyl-Cyanoacrylate (n-BCA) Trial Investigators Study found an average vessel perforation rate of 1.9% in glue (flow-directed microcatheter) versus 5.8% in PVA.28
Frequency: The N-Butyl-Cyanoacrylate (n-BCA) Trial Investigators Study found an average vessel perforation rate of 1.9% in glue (flow-directed microcatheter) versus 5.8% in PVA.28
 Thromboembolic events
Thromboembolic events
 9% symptomatic.29
9% symptomatic.29
 Other studies found lower risk, which varies with the embolic agents and lesions treated; 1.9% thromboembolic events and 3.8 to 5.8% stroke.28
Other studies found lower risk, which varies with the embolic agents and lesions treated; 1.9% thromboembolic events and 3.8 to 5.8% stroke.28
 Trapped microcatheter
Trapped microcatheter
 Frequency: 7.4% for n-BCA versus 0% for PVA.28 As for Onyx, the percent varies but can be as low as 2.5% and as high as 8.5%.22,30
Frequency: 7.4% for n-BCA versus 0% for PVA.28 As for Onyx, the percent varies but can be as low as 2.5% and as high as 8.5%.22,30
 Cranial nerve injury
Cranial nerve injury
 Specific complications related to vascular lesion embolization:
Specific complications related to vascular lesion embolization:
 Rupture, hemorrhage (with or without intraparenchymal hematoma), venous drainage obstruction.
Rupture, hemorrhage (with or without intraparenchymal hematoma), venous drainage obstruction.
 In arteriovenous malformation (AVM) treatment, 14% treatment-related neurologic deficit rate, a 2% persistent disabling deficit rate, and 1% mortality.31
In arteriovenous malformation (AVM) treatment, 14% treatment-related neurologic deficit rate, a 2% persistent disabling deficit rate, and 1% mortality.31
 In carotid-cavernous fistula (CCF), worsening of symptoms is possible but usually transient (it occurs as the cavernous sinus thromboses).
In carotid-cavernous fistula (CCF), worsening of symptoms is possible but usually transient (it occurs as the cavernous sinus thromboses).
 Specific complications related to tumor embolization:
Specific complications related to tumor embolization:
 Most common: fever and localized pain25
Most common: fever and localized pain25
 Intratumoral hemorrhage
Intratumoral hemorrhage
 Swelling (generally responds to corticosteroids)
Swelling (generally responds to corticosteroids)
 Scalp infarction
Scalp infarction
 Allergic reactions
Allergic reactions
 Groin complications (infection, hematoma, pseudoaneurysm, arterial occlusion) in 0.2%, with a 0.5% risk of hematoma requiring surgery or transfusion
Groin complications (infection, hematoma, pseudoaneurysm, arterial occlusion) in 0.2%, with a 0.5% risk of hematoma requiring surgery or transfusion
 Renal failure 0.2%
Renal failure 0.2%
 Deep venous thrombosis (DVT) or pulmonary embolization when treating arteriovenous malformations from venous shunting18
Deep venous thrombosis (DVT) or pulmonary embolization when treating arteriovenous malformations from venous shunting18
Embolization of Tumors
Meningiomas
 A retrospective analysis of 470 treated meningiomas found an average of 27% embolized skull base location meningiomas, including olfactory groove, planum sphenoidale/tuberculum, sphenoid wing, and cerebellopontine angle.42
A retrospective analysis of 470 treated meningiomas found an average of 27% embolized skull base location meningiomas, including olfactory groove, planum sphenoidale/tuberculum, sphenoid wing, and cerebellopontine angle.42
 Tumors size above 3 cm in diameter and with 50% of feeders from accessible ECA branches.
Tumors size above 3 cm in diameter and with 50% of feeders from accessible ECA branches.
 Imaging-confirmed deep supply or hypervascularity of the lesion.
Imaging-confirmed deep supply or hypervascularity of the lesion.
 Eloquent locations.
Eloquent locations.
 Significant calcified meningiomas are not generally embolized.
Significant calcified meningiomas are not generally embolized.
Paragangliomas
Angiofibromas
 Assess the vascularization of JAFs for surgical planning, delineate normal and pathological anastomosis, and assess retinal supply.56
Assess the vascularization of JAFs for surgical planning, delineate normal and pathological anastomosis, and assess retinal supply.56
 Correlations have been made between the size of the JAF and its blood supply. In general, ECA branches and the target vessels for presurgical embolization are often branches of the internal maxillary artery (IMAX) such as the sphenopalatine artery, accessory meningeal artery, and superior pharyngeal artery.
Correlations have been made between the size of the JAF and its blood supply. In general, ECA branches and the target vessels for presurgical embolization are often branches of the internal maxillary artery (IMAX) such as the sphenopalatine artery, accessory meningeal artery, and superior pharyngeal artery.
 In the Andrews classification, stage III and IV JAFs have tumor invading the infratemporal fossa, orbit, and extending intracranially, and have supply from ICA branches, anterior and posterior ethmoidal arteries, the inferolateral trunk, meningohypophyseal trunk (MHT), and other vessels.57
In the Andrews classification, stage III and IV JAFs have tumor invading the infratemporal fossa, orbit, and extending intracranially, and have supply from ICA branches, anterior and posterior ethmoidal arteries, the inferolateral trunk, meningohypophyseal trunk (MHT), and other vessels.57
 Embolization is often unsafe.6,23,55,57,58
Embolization is often unsafe.6,23,55,57,58
 In some instances, direct percutaneous puncture can be an option, when the ICA is the main vascular supply.59,60
In some instances, direct percutaneous puncture can be an option, when the ICA is the main vascular supply.59,60
 Careful angiographic images should be obtained and thoroughly inspected prior to embolization to assess dangerous anastomosis; occasionally, the use of temporary balloon occlusion of the proximal blood supply from the ICA or VA may be an adjunctive safety measure.61
Careful angiographic images should be obtained and thoroughly inspected prior to embolization to assess dangerous anastomosis; occasionally, the use of temporary balloon occlusion of the proximal blood supply from the ICA or VA may be an adjunctive safety measure.61
 Intraoperative blood loss ranges from 915 to 3,000 mL versus 450 mL with embolization.56
Intraoperative blood loss ranges from 915 to 3,000 mL versus 450 mL with embolization.56
Embolization of Vascular Lesions
Carotid Cavernous Fistula
 Type B: shunt between ICA branches and the CS
Type B: shunt between ICA branches and the CS
 Type C: shunt between ECA branches and the CS
Type C: shunt between ECA branches and the CS
 Type D: shunt between both ECA and ICA branches and the CS
Type D: shunt between both ECA and ICA branches and the CS
Treatment
Dural Arteriovenous Fistula
Goal
 Transarterial: usually favored for small dAVFs or those with venous occlusion or stenosis, where the access is restricted. A common initial strategy is to shut down the small feeders, which increase the flow in the main arterial supply. This increases the chance for the embolic agent (glue or Onyx) to penetrate the venous side, the ultimate end point. This strategy can also be used as an initial step, before the transvenous approach.
Transarterial: usually favored for small dAVFs or those with venous occlusion or stenosis, where the access is restricted. A common initial strategy is to shut down the small feeders, which increase the flow in the main arterial supply. This increases the chance for the embolic agent (glue or Onyx) to penetrate the venous side, the ultimate end point. This strategy can also be used as an initial step, before the transvenous approach.
 Transvenous: usually selected for large, complex dAVFs with a nonfunctional and isolated dural sinus. Access with a microcatheter followed by occlusion of the disease segment with coil is commonly performed.
Transvenous: usually selected for large, complex dAVFs with a nonfunctional and isolated dural sinus. Access with a microcatheter followed by occlusion of the disease segment with coil is commonly performed.
 Treatment through meningeal arterial supply offers a higher success rate than using the superficial temporal or occipital arteries to penetrate via the transosseous branches of the skull, which are millimetric and tortuous.87
Treatment through meningeal arterial supply offers a higher success rate than using the superficial temporal or occipital arteries to penetrate via the transosseous branches of the skull, which are millimetric and tortuous.87
 High-flow dAVFs are better treated by using a balloon to occlude the flow, or by embolization using a coil.88
High-flow dAVFs are better treated by using a balloon to occlude the flow, or by embolization using a coil.88
 As described above, occlude the smaller supply to increase the flow in the main feeder.
As described above, occlude the smaller supply to increase the flow in the main feeder.
 Cyanoacrylate (glue). More studies are available considering this agent, as it has been available for a long time. On initial transarterial approach, cure was achieved in only 30% versus 81% for a transvenous approach. In one study, a combined approach offered a 54% cure rate. Delayed thrombosis of a residual shunt has been described, with a follow-up angiogram showing complete occlusion in 88% of patients with residual flow at the end of the initial procedure.89,90 In a study involving 170 patients, cure was achieved in 60% and 69% of patient without and with cortical venous drainage, respectively. At follow-up, these numbers increase to 85% and 89%, with overall clinical improvement in 93%. These numbers seem more accurate with larger groups being included compared with case series, with an initial cure rate of 63 to 100%.91–94 For those that do not occlude with surgery or embolization, Gamma Knife radiosurgery is an option.
Cyanoacrylate (glue). More studies are available considering this agent, as it has been available for a long time. On initial transarterial approach, cure was achieved in only 30% versus 81% for a transvenous approach. In one study, a combined approach offered a 54% cure rate. Delayed thrombosis of a residual shunt has been described, with a follow-up angiogram showing complete occlusion in 88% of patients with residual flow at the end of the initial procedure.89,90 In a study involving 170 patients, cure was achieved in 60% and 69% of patient without and with cortical venous drainage, respectively. At follow-up, these numbers increase to 85% and 89%, with overall clinical improvement in 93%. These numbers seem more accurate with larger groups being included compared with case series, with an initial cure rate of 63 to 100%.91–94 For those that do not occlude with surgery or embolization, Gamma Knife radiosurgery is an option.
 Onyx: Available studies showed an immediate cure rate ranging from 62 to 95%.30,95–98 At follow-up angiogram, the cure rate has even reached 100%.95,96
Onyx: Available studies showed an immediate cure rate ranging from 62 to 95%.30,95–98 At follow-up angiogram, the cure rate has even reached 100%.95,96
 Occlusion Test
Occlusion Test
Balloon Test Occlusion
 A detailed clinical exam before, during, and after the occlusion
A detailed clinical exam before, during, and after the occlusion
 Timed angiographic imaging
Timed angiographic imaging
 Cerebral blood flow imaging
Cerebral blood flow imaging
Technique
 Neuropsychological testing109–111
Neuropsychological testing109–111
 Electroencephalography (EEG)112–115
Electroencephalography (EEG)112–115
 Somatosensory evoked potentials (SSEP)116–119
Somatosensory evoked potentials (SSEP)116–119
 Transcranial Doppler (TCD)127–129
Transcranial Doppler (TCD)127–129
 Xenon imaging107
Xenon imaging107
 Xenon computed tomography (CT)99,122,130,131
Xenon computed tomography (CT)99,122,130,131
 Positron emission tomography (PET)115
Positron emission tomography (PET)115![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


 Hypotension challenge
Hypotension challenge Angiography
Angiography Stump pressure
Stump pressure CT perfusion (CTP)
CT perfusion (CTP)