21
CHAPTER
![]()
Functional Neuroimaging
Jorge Oldan, Terence Wong, and Jeffrey Petrella
Functional imaging, referring here to nuclear techniques such as PET and SPECT, as well as functional magnetic resonance imaging (fMRI), is primarily useful in the surgical treatment of epilepsy. PET and SPECT are primarily used to localize potential candidates for resection of an epileptogenic focus, whereas fMRI is principally used to map crucial areas and aid in surgical planning.
While 70% of epilepsy patients can be treated medically, others may require surgical resection of the epileptogenic focus. Surgery is successful (defined as resolution of seizures over a period of 2 years after resection) in 60% to 90% of cases of temporal lobe epilepsy (TLE) and 22% to 65% of cases of extratemporal epilepsy (1). Usually, MRI is the favored modality for detecting structural abnormalities such as hippocampal or mesial temporal sclerosis (where patients also have the best postsurgical prognosis) (1). However, if MRI is normal (as in 20% to 25% of cases (2)), or localizes the focus to a different location from EEG, nuclear medicine techniques, PET and SPECT, may be used to guide placement of, or in some cases, replace subdural electrodes (1–3) for further study. For defining an epileptogenic focus (EF), ictal SPECT is the most sensitive study, followed by interictal PET and interictal SPECT (1).
PET
Introduction and Technique
PET uses tracers labeled with positron emitters to visualize and quantify cerebral metabolism. The usual tracer is 2-deoxy-2-[18F] fluoroglucose, or FDG, a glucose analogue. Formerly used more extensively, it has been largely supplanted by high-quality MRI. In addition to localizing the EF and suggesting prognosis (particularly for suggesting seizure freedom with a focal abnormality) (4) it can detect subtle anomalies such as focal cortical dysplasia (2). As PET may often not match MRI and EEG, all positive regions should be considered for electrode placement (4).
Unfortunately, tracer availability is somewhat limited by the half-lives of the atoms they are attached to. While SPECT tracers can be obtained from a generator good for a week, PET tracers used in clinical epilepsy imaging must be made by a cyclotron. The most commonly used positron emitter, F-18, has a half-life of 108 minutes, and others have even shorter half-lives (20 minutes for C-11). While F-18-labeled tracers can be bought and transported, C-11 tracers must be manufactured by an on-site cyclotron (2).
PET has superior spatial resolution (about 4–10 mm) (1) than SPECT, but as brain glucose uptake is slower (over 30 to 40 minutes) (5) and since FDG-PET reflects metabolism during the uptake phase, ictal studies are difficult to obtain. PET scans are thus usually interictal (6). The patient is monitored with continuous electroencephalography (cEEG) during the scan (and/or the 45- to 60-minute uptake between injection and scan) to avoid accidentally performing an ictal PET study (1,4). Ideally, the patient should be seizure-free for 24 hours, although 2 to 12 hours is considered acceptable in some centers (1).
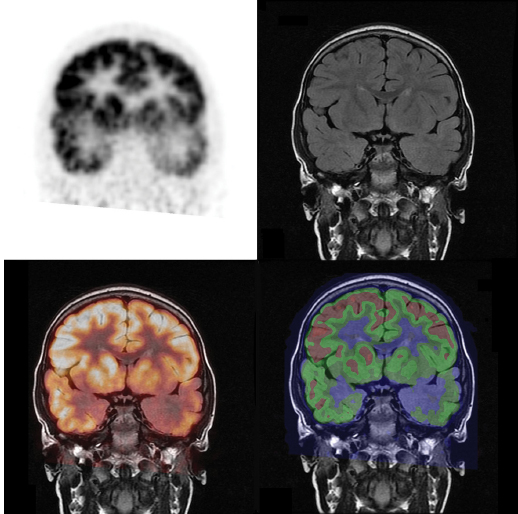
Interictal PET displays decreased activity in the affected area (Figure 21.1) reflecting hypometabolism, for unclear reasons (probably neuronal loss or functional disturbance) (1). Ictal PET scans that have been performed demonstrate significant hypermetabolism (3,5), but the mix of ictal, postictal, and interictal states usually due to proposed propagation is hard to interpret (4).
Whole-body effective dose for a PET of the brain is about 14.1 millisieverts (mSv) for a standard 20 microcurie (mCi) dose; this is about equal to a CT scan of the abdomen and pelvis (7).
Interpretation
The actual scan is usually evaluated visually. Quantitative analysis, using standardized uptake value (SUV), may be used as an adjunct (5). Usually, the most hypometabolic area is assumed to contain the epileptogenic zone (8), with relative temporal lobe hypometabolism of at least 15% to 40% shown to correlate with seizure-free outcomes (1). The onset zone is usually at the margin of the hypometabolic area (2,6).
FIGURE 21.1 Positive PET scan with hypometabolism in the left temporal lobe, corresponding to hypometabolic region. Upper left: PET image, upper right: MR image, lower left: fusion image using “hot iron” color map, lower left: fusion image using “rainbow” colormap. Note the additional decreased metabolism in the ipsilateral basal ganglia.
![]()
Findings may be related to semiology: a study of complex partial seizures found that hypometabolism was limited to the epileptogenic zone in focal limbic seizures, included the limbic cortex in widespread limbic seizures, included the extralimbic frontal lobe with posturing, and included cerebellum and parietal lobes with secondarily generalized seizures (3). Interictal hypometabolism in the putamen is associated with ictal dystonic posturing (4).
Temporal Lobe
The most common focal site of seizure onset is in the temporal lobe. It may be mesial, localized in the hippocampus and due to sclerosis, or lateral, usually neocortical in origin, due to a focal lesion or other cause. Mesial and lateral sites are difficult to distinguish on PET due to spatial resolution (4,5), volume averaging, and widespread temporal lobe hypometabolism in refractory mesial epilepsy (4).
Hypometabolism usually extends outside the lesion itself, although it is less severe extratemporally (4,5). Common sites are the ipsilateral temporal lobe, and less often the frontal (particularly insular and inferior frontal (4)), parietal, and occasionally contralateral temporal lobes (5), as well as, commonly, the dorsal thalamus (4). Mesial TLE specifically may involve the mesial and anterolateral temporal cortex, as well as the basal ganglia and thalamus (1,4), and more rarely the occipital lobe (4). Refractory patients have more widespread disease; conversely, nonrefractory TLE patients may have a normal PET (4,5). The inferolateral temporal lobe is a common site in MRI lesion negative disease (1).
Over time, the volume and severity of hypometabolism in mesial TLE increase (4). Executive function deficits and depression may be associated with orbitofrontal and prefrontal cortex abnormalities, with more frequent seizures causing worse impairment and larger abnormalities on PET; seizure control may decrease the size of these areas, and they may improve after surgery (3,8). Bilateral temporal lobe hypometabolism is more commonly associated with bilateral, diffuse, or extratemporal seizure onset and bilateral or diffuse MRI abnormalities (3,5). These patients have a lower probability of seizure remission after surgery (3,5) or medical treatment (3), and more cognitive deficits (3).
Children (but usually not adults) may have interictal hypermetabolism (4). Occasionally (1% to 2%) hypometabolism is seen contralateral to the intracranially recorded site (3,4), possibly due to subclinical epileptiform activity (5), prior surgery, and/or imaging artifacts (4). One way to distinguish focal hypermetabolism from contralateral hypometabolism may be quantification of temporal and occipital metabolism (4). While antiepileptic drugs will decrease metabolism, they will generally do so in a global fashion (4,5), so underlying asymmetries and bilateral symmetric abnormalities will still be visible (4).
Extratemporal Lobe
PET is less sensitive in the extratemporal region, particularly if MRI is normal (4). Extratemporal seizures tend to spread rapidly, so a rapid injection for ictal SPECT becomes more important (5), and detecting the precise focus is more difficult. However, PET can still be useful in guiding subdural electrode placement (5). The border with normal tissue is more gradual (4,5); if there is no lesion, the area may be quite small (4). Frontal lobe epilepsy frequently has a normal PET scan (3). If not, hypometabolism may be quite widespread, including ipsilateral temporal and parietal lobes and occasionally the thalami and basal ganglia (4,5). Parietal lobe foci appear to be more difficult to locate (3).
Aids to Interpretation
Potential aids include coregistration with MRI and voxel-based comparisons with normative data (2). These are particularly useful in extratemporal epilepsy (8).
Coregistering or fusing PET data with a structural MRI can help identify subtle findings such as cortical dysplasias, either by themselves or in association with tumors, and improve delineation of the epileptogenic zone for surgical planning (9). In general, the PET and MRI have to be registered by commercial software (most conventional PACS systems cannot do this well), which frequently allows for display of a fused image as well. A rainbow-color map protocol using a 15% difference corresponding to a color change and SUV calculation with differences of 10% considered significant has shown some effectiveness in the detection of focal cortical dysplasia (6). The resolution is superior to SPECT, but partial volume correction remains useful (8).
In statistical parametric mapping (SPM), each pixel is treated as a z-score using the mean and standard deviation for that patient’s overall study. Initial findings have shown successful lateralization and correlation with seizure duration, with the anterolateral temporal region being more important in predicting response to surgery (3). SPM may also be more useful in identifying bilateral abnormalities (3), particularly if asymmetric (4). Some authors suggest using a volume threshold to avoid focusing on very tiny, statistically insignificant foci (4).
Validity
Temporal Lobe
In the temporal lobe, PET sensitivity ranges from 80% to 90% (1,2,6), increasing to 90% with quantitative analysis (comparing to standard normal) in some trials (1,5). However, its sensitivity in MRI-normal cases may be as low as 44% (8). Concordance with the ultimate site of resection producing relief is high at 96% (10). Some preliminary work suggests flumazenil (FMZ) may be more effective at determining the area to be resected (5). As this is a C11-tracer, availability is limited to centers having a cyclotron.
Postsurgical outcomes are better with more severe abnormalities restricted to the temporal lobe (3,4) (especially mesial (3,5)), a single focus of hypometabolism (4) ipsilateral to the resection site (3,4,6,8), a greater degree of asymmetry (3,8), a greater portion of the hypometabolic area resected (2–4,6), white matter changes on MRI (3) and no or ipsilateral (versus contralateral) thalamic asymmetry (3). Conversely, extratemporal and bilateral temporal hypometabolism make postsurgical seizure control less likely (4). A lack of asymmetry may indicate extratemporal or bilateral foci (3). Essentially, the more likely it is that the temporal lobe is the problem, the more likely removing a portion of the temporal lobe is to help.
In some cases of refractory mesial TLE, use of intracranial electrodes may be avoided if there is unilaterally predominant hypometabolism when extracranial EEG electrodes show ictal onset from the ipsilateral temporal lobe, MRI is normal, nonspecific, or localized to the ipsilateral temporal lobe, and other data are not discordant (4). However, simply because the most severe abnormality on PET is in a temporal lobe does not indicate that all seizures arise from that location; thus, no other contradictory data should be present. If the EEG is nonlocalizing, intracranial electrodes may still be necessary (4).
Extratemporal
PET sensitivity for extratemporal epilepsy is lower than for temporal lobe epilepsy. It is somewhat higher at 70% to 90% than the sensitivity of 30% to 80% for MRI (6) (although concordance may be somewhat lower at 68% (10)). In the case of frontal lobe epilepsy, sensitivity is lower at 45% to 73%, and even lower at 36% for MRI lesion-negative patients; performance may be somewhat higher in children (5).
Patients with extratemporal cortical hypometabolism ipsilateral to the site of surgery generally had a lower frequency of successful surgery than those with temporal cortical hypometabolism (45%–60% vs. 75%) (3,5,8). PET is of more utility in guiding subdural electrode placement than in detecting the epileptic foci (EF), due to the relatively rapid spread (5). In addition, work using high-resolution registration has suggested that small mismatches between hypometabolic and epileptogenic areas are common in extratemporal epilepsy. PET may also find additional foci (4).
Cortical Dysplasias
PET has been shown to be particularly useful in cases of multiple or focal cortical dysplasias (FCD) that are often subtle on MRI. It is 75% to 100% sensitive (3,9) and 78% concordant with MRI (1) in localizing areas with FCD (usually appearing as a hypometabolic zone); more areas (and more extensive areas) may be visible with PET than with MRI (1,6). Sensitivity for cortical dysplasia in general may reach 70% to 80% with coregistration (6), and coregistration will allow for localization of an EF in 33% of patients with negative or discordant EEG and MRI (6).
Other Uses
PET is useful in neonates with intractable seizures of unknown etiology. A focal abnormality suggests a focal developmental abnormality, whereas diffuse abnormalities suggest a metabolic cause (11).
PET has been shown to detect abnormalities not seen on MRI in cryptogenic infantile spasms (West syndrome). Surgical removal of abnormal foci of cortical dysplasia can stop seizures and sometimes reverse developmental arrest (3,4,11), particularly if PET and EEG are concordant and/or PET anomalies normalize on medical therapy (4).
PET in Lennox-Gastaut syndrome may suggest EF (3). Cortical tubers are hypometabolic on FDG-PET (to about 30% to 50% vs. the other side) (1), and show increased uptake with 11C-AMT—in two-thirds of epileptogenic cases (5). In Sturge-Weber, PET shows hypometabolism ipsilateral to the nevus and demonstrates the extent of involvement in the cortex below, guiding resection and detecting contralateral hypometabolism when hemispherectomy is considered (3–5). Patients with cutaneous but no intracranial anomalies have a normal PET (4). Patients with GLUT1 transporter deficiency show generalized reduction in FDG activity, but relative preservation in the basal ganglia (4). PET is generally not used in primary generalized epilepsy to exclude surgical candidates, as such patients are usually diagnosed from history and EEG; if performed, interictal FDG studies are usually normal, as is sometimes the case with secondarily generalized epilepsy (4).
New Directions
Simultaneous acquisition in a PET-MRI system may improve coregistration, as well as helping to determine motion correction, or obtain multifunctional imaging such as combining PET ligand uptake with processes such as arterial spin labeling, MR spectroscopy, or blood oxygenation-level-dependent imaging (12).
While FDG is the most common tracer used, other tracers can be used to investigate other pathways, such as 11C-FMZ for GABA-A receptors or 11C-alpha-methyltryptophan (AMT), a serotonin precursor (1). Many of these are limited by use of 11C as a radionuclide, whose half-life of 20 minutes not only allows for a smaller radiation dose but also limits administration to institutions with a cyclotron.
Much like FDG, FMZ binding is significantly lower in the EF (1,16), the degree of which may correlate with seizure frequency (3). 11C-flumazenil may have a more restricted area of hypometabolism than FDG (1,9,11), and may be more sensitive for contralateral abnormalities (9) and mesial temporal sclerosis (11). Changes have been seen in the thalamus in temporal lobe epilepsy and contralateral cerebellum in frontal lobe epilepsy as well (3). Larger regions of FMZ uptake may correlate with seizure frequency (1), and periventricular uptake before surgery may correlate with a poor outcome (1). It may be more accurate in extratemporal, in particular, frontal, epilepsy as well (1,3).
11C-AMT has shown to be specific for seizure foci, particularly in tuberous sclerosis (8), as described previously, where epileptogenic tubers have a higher uptake (1). It may also be useful in identification of nonresected epileptogenic cortex (5) or developmental malformations (11).
Among the many other tracers in investigative use are 11C-carfentanil and 11C-methylnaltrindole for opioid receptors and 18-MPPF, a selective 5-HT1A agonist (3). Opioid-receptor tracer uptake may be more localized than FDG and may increase postictally. Synaptic opioid levels may correlate with seizure activity (3). 18-MPPF, a selective 5-HT1A agonist, may be more sensitive and accurate (3) in MRI lesion-negative temporal lobe cases.
SPECT
Introduction and Technique
SPECT evaluates regional cerebral perfusion (rCP), which is generally coupled to metabolism. The seizure zone is hyperperfused during epileptic activity because of local neuronal hyperactivity (8), whereas it is hypoperfused between seizures (6). SPECT, unlike PET, allows for ictal imaging (13). Spatial resolution is generally not as good as PET (about 8–10 mm) (6).
Of the three FDA-approved radiopharmaceuticals, one (IMP, or isopropyl-iodo-amphetamine) is primarily used in Japan. The other two, both using Tc-99m as the radionuclide, are HMPAO (hexamethylpropylamine oxime, Ceretec, GE Healthcare, Chalfont St Giles, UK) and ECD (ethyl cysteinate dimer; Neurolite, Bristol-Myers Squibb, North Billerica, MA). The Tc-99m radionuclide has a longer half-life (6 hours) and is obtainable from generators, allowing for easier availability relative to PET. In general, the compound is bound to a specific area of brain on its first pass through and remains there (being converted to a polar metabolite) for several hours. The patient can thus be injected during a seizure, and the SPECT scan be done hours (up to 2 (13) to 4(1)) later when the patient has recovered and is cooperative. One important difference between the two compounds is that HMPAO must be used within 20 to 30 minutes of its preparation, whereas ECD is stable for 4 to 6 hours. ECD is also rapidly excreted renally, decreasing radiation burden and allowing for administration of a larger amount of radiopharmaceutical with the same overall dose (5). In the experience of the authors, ECD produces a much better quality of image with much higher sensitivity.
In general, the patient is admitted for video EEG (vEEG) monitoring with ictal and interictal scans planned during the same admission. The patient (or their parents) is consented. The tracer is injected in the telemetry suite, with the radiopharmaceutical in a lead-shielded syringe containing the prescribed dose. A vial in a lead container with more of the radiopharmaceutical is available as well, so a sufficient amount of tracer to replace the decayed tracer can be withdrawn if necessary. A sheet is used for displaying an appropriate volume to be withdrawn to account for radioactive decay (5). Note that the compound must be discarded after 6 hours if the patient does not have a seizure, so frequently the radiopharmacy will have to send up to two doses during the day (13).
The exact times of tracer injection and EEG onset of seizure should be noted so one can determine if the injection is ictal, peri-ictal, or postictal (5). Seizures should last at least 5 to 10 seconds, and (ideally) the SPECT tracer should be injected less than 20 seconds (8) after the seizure starts, although 45 (13) to 90 seconds (10) is acceptable. Injection after the seizure—postictal SPECT—is less sensitive (13), as focal hyperperfusion becomes hypoperfusion. This postictal switch is generally complete at 100 seconds after the seizure ends (8). While ictal scans are generally considered superior, a postictal scan may retain some value, at least in temporal lobe lesions. One study showed a pattern of mesial hyperperfusion accompanied by lateral hypoperfusion having a 97% PPV; a postictal injection retains a 70% to 90% sensitivity in mesial TLE (5).
Imaging itself usually takes about 20 to 30 minutes (13). Children 1 to 4 years old (or older if uncooperative) are generally sedated, whereas children less than 1 year of age are fed and wrapped before the scan (5). In general, the patient should be sedated on the ward before arriving in the imaging department (5).
The interictal scan, conversely, is usually performed when the patient has been seizure-free for at least 30 minutes (usually on a different day). The patient is usually injected in the imaging department for this scan, with sedation if necessary for a pediatric patient. The interictal scan is less useful by itself, but aids the localization of the EF together with the ictal scan by visual assessment or subtraction (5).
Images can be reconstructed at any angle (13); however, data should be reconstructed after reorientation parallel to the anterior–posterior commissure line with another set of data reconstructed parallel to the long axis of the temporal lobe (5). A continuous color scale is recommended, to avoid overestimation of tiny asymmetries. A high-resolution or fan-beam collimator should be used (5).
Whole-body effective dose equivalent from a 20 mCi dose of Tc-99m HMPAO is 6.9 mSv, whereas that from 20 mCi of Tc-99m ECD is 5.7 mSv. This is about half the dose for a CT scan of the abdomen and pelvis, and 3 to 3.5 times that of a head CT scan (7).
Interpretation
In general, areas of abnormal activity are described as being hyper- or hypo-perfused, as compared with a reference set (cerebellum, if unaffected, or average cortical activity in the same slice). A SPECT study showing increased ictal relative cerebral perfusion (rCP) and decreased interictal rCP in the same area strongly suggests that a lesion is the cause of seizures (13); subtraction of the interictal from the ictal scan can help visualize this (2). If multiple “hot” areas are visible, the largest and “hottest” area of ictal hyperperfusion is assumed to represent the ictal-onset zone (8). Note that it takes 15 to 20 seconds for the tracer to reach the brain from intravenous injection, so areas of propagation are usually seen as well (8) (Figure 21.2).
Temporal Lobe
Interpreting the study should take into account time of injection. In cases of TLE, seizure activity tends to propagate to the contralateral temporal lobe and ipsilateral insula, basal ganglia, and frontal lobe (8). Specifically for mesial temporal sclerosis, one may see hyperperfusion in the whole temporal lobe and possibly orbital and frontal cortex within 15 seconds of injection. Slightly more delayed hyperperfusion may be seen in the lateral temporal, orbital, or motor cortex, basal ganglia, or both temporal lobes. Hypoperfusion of the lateral temporal lobe with persistent hyperperfusion of mesial structures occurs within 4 minutes after the end of the seizure. Finally, hypoperfusion of the whole area may be seen 15 minutes after the end of seizure (5).
Correlations between pathology and hyperperfusion patterns have been made. Mesial temporal lobe lesions tend to show well-localized hyperperfusion involving the mesial and lateral temporal lobes. Lateral temporal lobe lesions show more bilateral changes, possibly as a result of connections to the opposite amygdala via the anterior commissure. The changes are nonetheless more prominent on the ipsilateral side (5). MRI lesion-negative patients with good outcomes have shown a pattern of hyperperfusion restricted to ipsilateral antero-mesial temporal structures (5). However, due to issues of spatial resolution, differentiation between mesial and lateral temporal lobe lesions is difficult.
Extratemporal Lobe
The most common pathology in this case is focal cortical dysplasia, and, as with PET, one aims to confirm the importance of the visible lesion and to localize the area for subdural grid placement (5). A particular problem with extratemporal seizures is their short duration (at least 10 to 15 seconds is necessary for accurate localization). As perfusion changes do not persist past the seizure (unlike temporal lobe seizures), postictal injections are not useful (5).
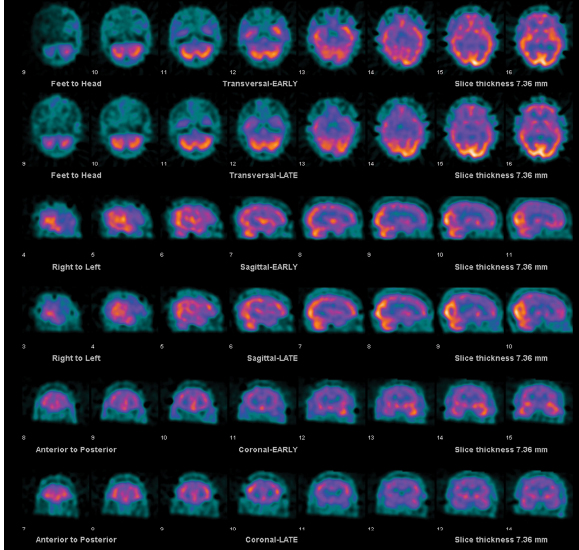
FIGURE 21.2 Ictal (upper rows-”EARLY”) and interictal (lower rows-”LATE”) SPECT, with focus of increased activity seen in left temporal lobe on ictal imaging, which is not present on interictal imaging. In this case where SPECT images are being interpreted without reference to an outside image, ictal and interictal series are lined up in each of the three reference planes, with manual adjustment of each series for better correspondence.
![]()
Patterns of hyperperfusion differ with time of injection, and include (ideally) a large and intense hyperperfused area at the lesion itself (in early injection), a less intensely hyperemic area at the actual lesion with the most intense uptake at a distance (at later injection times), and a complicated, multilobulated pattern (also at later injection times), seen in frontal lobe seizures with fast propagation. In frontal lobe lesions, the largest cluster of ictal hyperperfusion usually includes other brain regions as well as the ictal-onset zone (8).
Patterns of propagation are somewhat distinct for extratemporal epilepsies as well. Occipital seizures quickly spread to both temporal lobes, making early injection important. An anterior spread is often associated with sensory and motor manifestations (5). Frontal lobe epilepsy often demonstrates hyperperfusion in ipsilateral basal nuclei and contralateral cerebellar hypoperfusion (5). A larger area of hyperperfusion than the tumor itself may be seen in dysembryoplastic neuroepithelial tumour (DNET) (5). Focally increased uptake on ictal SPECT in tuberous sclerosis corresponds to sustained rhythmic fast activity on an ictal EEG (5). This may be useful in cases of multiple tubers (5).
Aids to Interpretation
With SPM, the ictal SPECT is compared to a series of normal scans, and each voxel is analyzed with statistical tests to identify regions of significant change. One advantage of this technique is that it may make interictal scans unnecessary (one can compare with the standard normal instead). However, there are issues of regional alterations in blood flow unrelated to seizure activity, and particularly in the pediatric population under 6, developmental stages of the brain may make the technique less accurate (15).
SISCOM (subtracted interictal SPECT coregistered to MRI) is a procedure where ictal and interictal SPECTs are subtracted and the result coregistered to an MRI (Figure 21.3). It may be useful in presurgical evaluation when MRI is normal or localizes discordantly with EEG, multiple epileptogenic lesions are present (such as in bilateral mesial temporal sclerosis or bilateral cortical dysplasias) or to identify a target for placement of intracranial EEG electrodes. SISCOM is particularly useful in malformations of cortical development, as the dysplastic areas are not definitely known to be epileptogenic yet (6,13), in tumors such as DNET where a surrounding area of dysplasia may be present (5), for extratemporal epilepsies, and cases of rapid seizure propagation (8). As with PET, coregistration with MRI may reveal previously occult abnormalities; however, SPECT is more commonly used to help choose intracranial electrode placement by providing information on secondary spread of ictal activity (2). Another aid is voxel-based analysis of studies—specifically, calculating the difference between ictal and interictal cerebral blood flow as a Z-score of the variation between scans using the mean and standard deviation of the differences in all brain voxels (2,8). In the case of SISCOM, careful quality control of registration (assessment of motion and registration errors) becomes important to avoid false negatives and positives. SISCOM assessment should be concordant with visual assessment (8).
Validity
Among the nuclear medicine methods, ictal SPECT is the most accurate, with a sensitivity of 96% reported in one meta-analysis (1). In the case of complex partial seizures in particular, ictal SPECT is 71% to 93% sensitive (over 90% for temporal lobe seizures) and has 95% PPV (13). Localization is correct in 80% to 90% of cases (5). Concordance with ultimate site of relief-producing resection is very high (10). Early postictal SPECT retains a sensitivity of 75% (8). Interictal SPECT is less sensitive, with sensitivity about 50% for temporal lobe seizures (6).
Neocortical epilepsy, including extratemporal limbic and extralimbic epilepsy, has a very low sensitivity for interictal SPECT (15% to 30%), but a reasonable sensitivity for ictal SPECT (>75%) (6,10) and good concordance (92%) (10). One reason for the relatively better performance of SPECT vis-à-vis PET in non-temporal-lobe epilepsy may be the preponderance of neuronal migration disorders, in which PET can have a variety of appearances (10). However, as extratemporal seizures are frequently brief, often a postictal SPECT is actually performed, which has a lower sensitivity of 20% to 50% (6). A comparison of PET and SPECT is presented in Table 21.1.
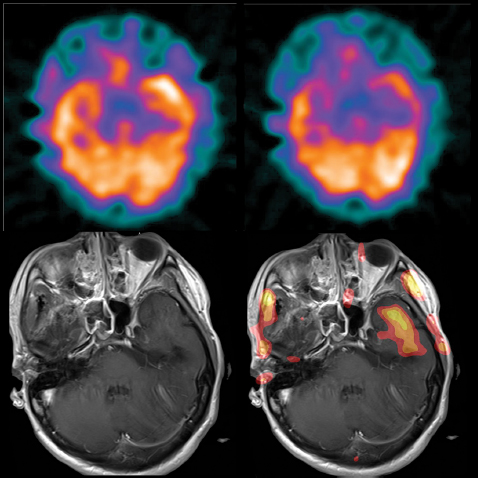
FIGURE 21.3 SISCOM. Ictal, interictal, MRI, and fused subtracted images (same patient as Figure 21.2). SISCOM projects the area of ictal over interictal increase over the MR, showing the area of greatest discrepancy (in favor of higher intensity in the ictal image) projecting over the left temporal lobe.
![]()