Fig. 13.1
Biodistribution of extracellular gadolinium-based contrast agents. Normal non-contrast T1 image (a) and corresponding post-contrast fat-suppressed T1 image (b) obtained with intravenous administration of MultiHance show expected enhancement within the venous structures (green arrow), basilar artery (red arrow), pituitary stalk (black arrow), mucosa (white arrow), extraocular muscles (yellow arrow), and lacrimal gland (blue arrow)
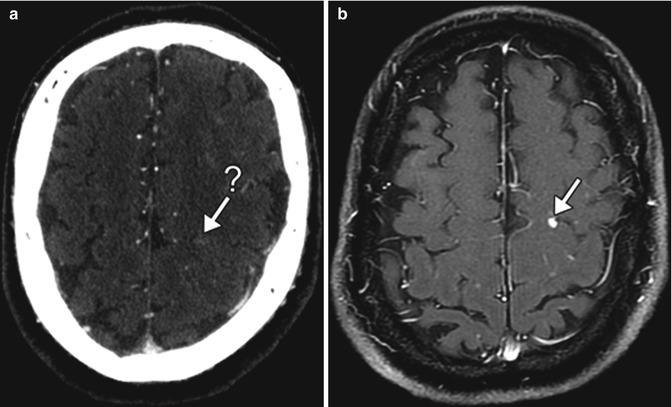
Fig. 13.2
Metastatic disease. Axial post-contrast CT (a) shows a very faintly enhancing lesion in the left perirolandic region (arrow-?). Post-contrast T1-weighted MRI (b) obtained shortly thereafter clearly shows the presence of a metastasis (arrow)
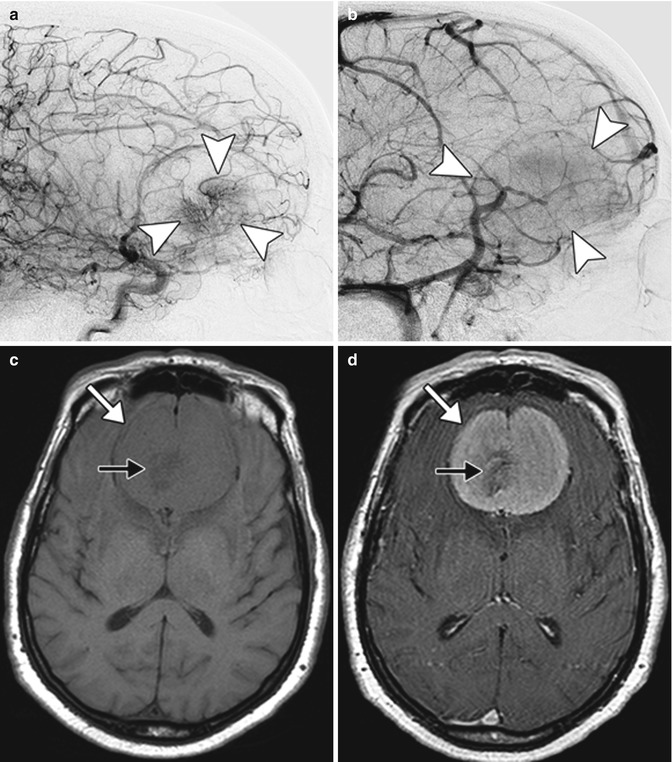
Fig. 13.3
Meningioma. Lateral digital subtraction angiogram images in early (a) and late (b) phases show a hypervascular mass in the frontal region (arrowheads). Axial T1 (c) and post-contrast T1 (d) images show a large anterior parafalcine mass (arrows) with delicate central flow voids (black arrows)
In general, GBCA are better tolerated than iodine-based contrast agents used in CT examinations, which can be explained by much smaller doses and biochemical characteristics. The incidence of adverse reactions ranged from 0.07 to 2.4 % when used in doses not exceeding 0.2 mmol/kg. The most common adverse reactions include nausea, headaches, dizziness, paresthesias, and coldness or itching at the injection site. True allergic type reactions are rare, with reported incidence of 0.004–0.7 %, and usually limited to urticaria or hives. Severe allergic reactions are extremely rare. Management of anaphylaxis and other contrast reaction is beyond the scope of this text.
A rare delayed complication of GBCA administration is nephrogenic systemic fibrosis (NSF). NSF occurs in patients with end-stage renal disease or severe acute renal failure in association with specific contrast agents (Omniscan, Magnevist, and OptiMARK) and is believed to be dose dependent. Development of NSF is related to in vivo release of gadolinium from chelating agents. The dissociation rate of macrocyclic agents is much lower than linear agents, and they are a safer alternative in patients with impaired renal function. NSF predominantly affects the liver, lungs, heart, and skin. In particular, the skin involvement in NSF typically manifests as bilateral symmetric epidermal atrophy and hair loss in the legs (Fig. 13.4). There can also be hyperpigmentation as well as skin hardening or wrinkling. No new cases have been reported in literature in recent years, likely due to increased awareness of the condition, leading to restricted use of GBCA in patients with low GFR.
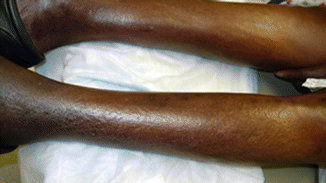

Fig. 13.4
Nephrogenic systemic fibrosis. Photograph show epidermal atrophy and hairlessness of the affected regions in the legs (Courtesy of Vesna Petronic-Rosic MD, University of Chicago)
Even in patients without renal failure, repeated contrast-enhanced MRI scans can lead to accumulation of gadolinium ions in the brain. This can manifest as high signal intensity in the dentate nucleus and globus pallidus on unenhanced T1-weighted images (Fig. 13.5).
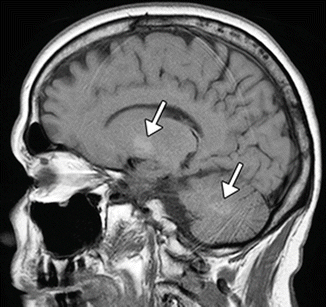





Fig. 13.5
Chronic gadolinium accumulation in the brain. The patient underwent 24 prior contrast-enhanced MRI exams. Sagittal T1 MRI shows patchy hyperintensity in the dentate nucleus and globus pallidus (arrows)
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








