Fig. 1
Transduction of neurons and glia in the myenteric plexus of the ENS. Adult male rats received direct injection s of AAV9 (a–d) or AAV6 (e–h) expressing green fluorescent protein (GFP) into the wall of the descending colon and were sacrificed 1 month later. Tissue from the myenteric or submucosal plexus was stained for the neuronal marker HUc (red), and the glia marker, glial fibrillary acidic protein (GFAP; blue). AAV9-mediated neuronal transduction (a–d) in the myenteric plexus tissue is shown as colocalization of GFP (green) expression in HUc immunoreactive cells (colocalization appears yellow). Within the submucosal plexus, enteric glia form a sheath around neurons. AAV6-mediated transduction of glia (e–h) is shown as GFP expression within GFAP immunoreactive cells (colocalization appears turquoise). Scale bar in (d) and (h) is 100 μm and applies to all panels
2 Materials
3.
Isoflurane vaporizer with induction box and nose cone adapters.
4.
Isoflurane (Abbott Animal Health, Abbott Park, IL, USA).
5.
Programmable micro syringe pump with foot pedal control (e.g. Harvard Apparatus Pump 11 Elite or equivalent).
6.
50 μL Hamilton syringe with 26 or 31 gauge beveled tip needle (Hamilton, Reno, NV, USA; see Note 3 ).
7.
Sigmacote siliconizing reagent (Sigma-Aldrich, St. Louis, MO, USA).
8.
Heating pad.
9.
Hair clippers.
10.
Betadine surgical scrub (Thermo Fisher Scientific, Waltham, MA, USA).
11.
Sterile Surgical Gauze (Thermo Fisher Scientific, Waltham, MA, USA).
12.
Sterile saline (Aspen Veterinary Resources, Liberty, MO, USA).
13.
11.5 cm straight fine scissors (FST, Foster City, CA, USA).
14.
Two 12.5 cm serrated halstead hemostats (FST, Foster City, CA, USA).
15.
12 cm adson foreceps with rounded tip (FST, Foster City, CA, USA).
16.
1 and 20 mL syringes (BD, Franklin Lakes, NJ, USA).
17.
Sterile adhesive plastic surgical drape (e.g., Bioclusive by Johnson & Johnson, or equivalent).
18.
Black animal tattoo pigment and ultra-fine tip tattoo needle (AIMS, Hornell, NY, USA).
20.
Autoclip stapler (FST, Foster City, CA, USA).
22.
Ketofen (ketoprofen; Fort Dodge Animal Health, Fort Dodge, IA, USA).
23.
Timentin (ticarcillin disodium and clavulanate potassium; GlaxoSmithKline, Brentford, UK).
24.
3 % hydrogen peroxide.
3 Methods
3.1 Pre-operative Preparation
1.
Siliconize the Hamilton syringe using the Sigmacote siliconizing reagent (see Note 4 ). Fill an Eppendorf tube with approximately 500 μL of Sigmacote. Using the Hamilton syringe, withdraw Sigmacote to fill the syringe, allow to sit for 10 s, then expel the Sigmacote back into the Eppendorf tube. Repeat this process approximately 10 times to ensure adequate coating of the syringe. Dry overnight.
2.
All surgical tools should be autoclaved prior to use, including: Scissors, forceps, hemostats, autoclip stapler and staples, tattoo needle, and gauze. Thoroughly sterilize other surgical supplies that cannot be autoclaved. Disinfect and sterlilize surgical station (see Note 5 ).
3.
Prepare working concentrations of Ketofen and Timentin. Determine the total cumulative body weight (in kilograms) of all animals which will receive surgery. Add 10 % to this value to ensure adequate amount of drugs. This value is the total volume (in milliliters) of each respective drug you will need for all surgeries (based on the working concentrations described here). Prepare working dose of Ketofen by diluting Ketofen stock to a final working concentration of 5 mg/mL in the calculated total volume of sterile saline. Dilute Timentin stock to a final working concentration of 60 mg/mL in the calculated total volume of sterile saline (see Note 6 ).
4.
Prepare a 1.5 mL Eppendorf tube containing 3 % hydrogen peroxide and another containing sterile saline.
5.
Fill a 20 mL syringe with sterile saline.
6.
Set up the micro syringe pump and secure siliconized Hamilton syringe inside of syringe holder. Program the pump (for the specific syringe) to infuse a volume of 5 μL at a flow rate of 10 μL/min. Load the Hamilton with a total of 30 μL of virus .
3.2 Laparotomy and Viral Injection
1.
Turn on isoflurane vaporizer and set at 2 % isoflurane and oxygen set at 0.5 %. Allow the gas to fill the induction chamber (see Note 7 ).
4.
Place the animal on its back on top of the heating pad with the nose of the animal inside of the anesthesia nose cone (see Note 9 ).
5.
Apply betadine to the shaved surgical area and use the sterile surgical gauze to spread across the entire abdomen (Fig. 2a).
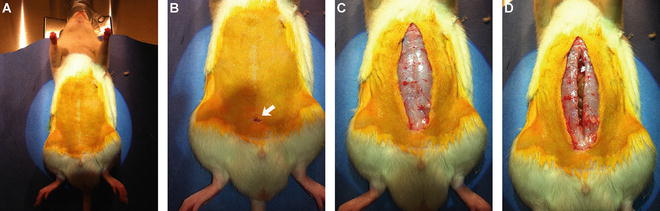

Fig. 2
Performing the laparatomy. Anesthetize the animal, thoroughly shave the abdomen and scrub with betadine to disinfect the surgical site. Place the animal on a heating pad with the abdomen facing up (a). Make a small initial incision in the skin at the approximate level of the pelvis (arrow in b). Continue the incision in the skin vertically up to the level of the sternum (c). Make a small initial incision in the abdominal muscle at the level of the pelvis and continue incision in the abdominal wall vertically up to the level of the sternum (d)
6.




Ensure that the animal is fully anesthetized again prior to making initial incision. Using the straight fine scissors cut a small entry incision in the skin overlaying the abdomen (Fig. 2b; see Note 10 ).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree






