CHAPTER 51 GENETICS OF EPILEPSY
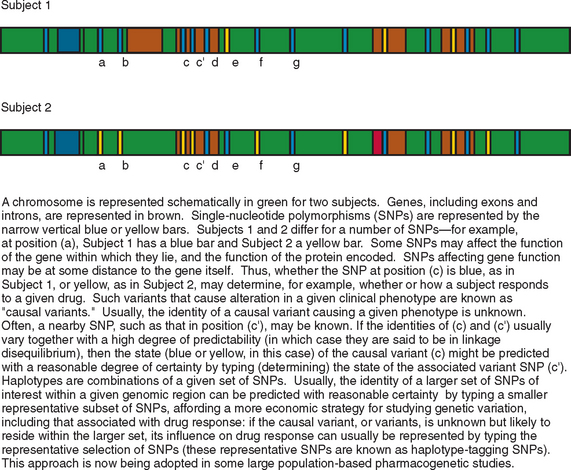
Epilepsy is a heterogeneous group of conditions, with a very broad range of possible causes, manifesting as recurrent, unprovoked, clinical seizures, sometimes with additional neurological or extraneural features. It is the most common serious neurological disorder, affecting 5 per every 1000 people, and is associated with increased rates of mortality and morbidity in almost every sphere of life for affected patients. The types of epilepsy can be subdivided in a number of ways; classification schemes focus variously on seizure types, natural history, or etiology. Although there are many causes of epilepsy, it is likely that an individual’s genetic makeup contributes to or modulates, to some degree, the risk of developing epilepsy in response to another insult to the brain, whatever the nature of the insult. Genetic differences between individuals take a number of forms: there are rare variants, called mutations, each of which occurs in less than 1% of the population and usually in a far smaller percentage; there are more common variants, the most common type of which is known as single-nucleotide polymorphism (SNP), a variation occurring at a single nucleotide and seen in greater than 1% (and usually greater than 5%) of the population (Fig. 51-1).
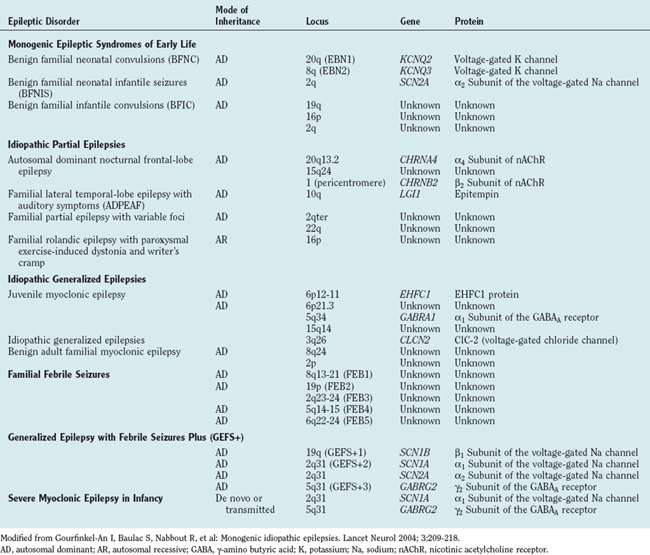
For most people with epilepsy, any genetic contribution to etiology is likely to arise from the additive, individually small effects of SNPs in a number of genes; understanding these effects requires analysis of large numbers of homogeneous groupings of patients and constitutes the field of population genetics. However, this area is only now beginning to be addressed by researchers in epilepsy. Most research to date on epilepsy genetics has concentrated on epilepsy caused by genetic mutations; almost by definition, such types of epilepsy are individually rare and even grouped together are unlikely to account for more than a small proportion of cases of epilepsy. The hope of research on these rare types of epilepsy caused by mendelian genetics is not only to inform disease management in affected individuals but also potentially to cast light on the genetics, pathophysiology, and management of epilepsies more broadly. In fact, this latter aim has yet to be realized, but certainly many epilepsies caused by single gene mutations are now known (Table 51-1) and account for a majority of the disorders covered in this chapter. Most of the genes implicated encode neuronal ion channels that directly affect neuronal excitability, or they function through alteration in ion flux; the finding that channelopathies cause seizures was a surprise initially, but it stands to reason because excitation and inhibition are fundamental neuronal properties, and seizures are generally held to arise from a systems imbalance between excitation and inhibition.
There are more than 200 genetically mediated conditions in which epilepsy is part of a broader phenotype. These conditions are not considered here; this chapter illustrates concepts of epilepsy genetics, drawing from genetic disorders in which the major and often sole manifestation is epilepsy. A glossary is given in Table 51-2.
TABLE 51-2 Glossary of Epilepsy Genetics Terms
| SCN1A | α1 Subunit of the voltage-gated sodium channel |
| SCN1B | β1 Subunit of the voltage-gated sodium channel |
| SCN2A | α2 Subunit of the voltage-gated sodium channel |
| GABA | γ-Amino butyric acid |
| KCNQ | Voltage-gated potassium channel |
| CLCN | Voltage-gated chloride channel |
| CHRNA4 | α4 Subunit of the nicotinic acetylcholine receptor |
| LGI1 | Leucine-rich gene, glioma inactivated |
| Gene | A segment of DNA that normally specifies a functional polypeptide or gene product |
| Allele | One of several alternative forms of a gene or DNA sequence at a specific locus |
| SNP | Single-nucleotide polymorphism, where more than one variant (allele) occurs at a locus with a frequency greater than 1% |
| Mutation | Change within a gene or DNA sequence due to base substitution, deletion or insertion occurring at a frequency < 1% and usually much less |
| Autosome | Any chromosome other than the sex chromosomes X and Y |
| Locus | A unique chromosomal location |
| Linkage | The tendency of genes to be inherited together as a consequence of their physical proximity |
| GEFS+ | Generalized epilepsy with febrile seizures plus |
| SMEI | Severe myoclonic epilepsy of infancy |
| JME | Juvenile myoclonic epilepsy |
| BFNC | Benign familial neonatal convulsions |
| ICCA | Infantile convulsions and choreoathetosis |
| ADNFLE | Autosomal-dominant nocturnal frontal lobe epilepsy |
| BFNIS | Benign familial neonatal-infantile seizures |
| ADPEAF | Autosomal dominant familial lateral temporal lobe epilepsy with auditory features |
| ARFGEF2 | Adenosine diphosphate (ADP)–ribosylation factor guanine nucleotide exchange factor 2 |
| BIG2 | Brefeldin A (BFA): inhibited GEF2 protein |
| GPCR | G protein–coupled receptor |
MONOGENIC INHERITED EPILEPSIES
Generalized Epilepsies
Juvenile Myoclonic Epilepsy
This syndrome is characterized by the occurrence of myoclonic jerks, generalized tonic-clonic seizures, and absence seizures with onset in the early teens, normal magnetic resonance imaging findings, and a characteristic electroencephalographic pattern. Familial cases occur, and despite widespread acceptance that there is, even in sporadic cases, strong genetic susceptibility to juvenile myoclonic epilepsy (JME), only two responsible genes are known.1,2
Linkage has been described for JME in two regions of the short arm of chromosome 6 (6p12-11 and 6p21.3).3,4 Suzuki and associates (2004)2 examined 18 genes encoded in the linked 6p12-11 region. They discovered mutations in only one gene, EHFC1. They described five missense mutations in EHFC1, which encodes a protein with an EF-hand motif that cosegregated with epilepsy or electroencephalographic polyspike and wave activity in six unrelated families and was not detected in 382 control individuals. However, mutations were detected in only 6 of 44 families with JME examined. Although mutations in EHFC1 are the first described for JME, this study also highlights the underlying genetic heterogeneity of this condition.
Genetic heterogeneity may also explain the variable mode of inheritance for JME. Elmslie and colleagues described linkage of JME to 15q14.5 Cossette and associates1 described autosomal dominant inheritance in a French-Canadian family with linkage to 5q34. This region includes the genes for a number of γ-amino butyric acid (GABA) receptor subunits and has since been shown to contain a causative mutation in the GABRA1 gene.
Generalized Epilepsy with Febrile Seizures Plus (GEFS+)
Investigators have described families in which some members have febrile seizures persisting beyond the usually accepted upper age limit for the diagnosis of such seizures (6 years) and are thus denoted “febrile seizures plus”; other members of the same family may have generalized epilepsy. Within such families there can be wide phenotypic variability; febrile seizures, febrile seizures plus, generalized epilepsy, hemiconvulsive seizures, and temporal lobe or frontal lobe seizures may occur. Some members with generalized or partial epilepsy may not have had febrile seizures or suffer from a number of different seizure types, and some members may have febrile seizures but never suffer another seizure beyond the age of 6. Generalized epilepsy with febrile seizures plus (GEFS+) has an autosomal dominant pattern of inheritance with reduced (70% to 80%) penetrance. GEFS+ displays genetic heterogeneity, inasmuch as mutations in different ion channel genes can give rise to similar phenotypes. The most frequent site of mutation in patients with febrile seizures plus (57%) is the in the SCN1A gene6; some mutations probably result in defective fast inactivation of channel gating and neuronal hyperexcitability. Mutations causing GEFS+ have also been described in SCN1B, SCN2A, and the GABA receptor gene GABRG2 (see Table 51-1).
GABAA receptors contain an integral chloride channel that mediates synaptic inhibition. There are specific binding sites for GABA, barbiturates, benzodiazepines, and steroids. Bowser and associates reported altered kinetics and benzodiazepine sensitivity of a GABA receptor subunit mutation, GABRG2 R43Q, in a family with childhood absence epilepsy and febrile seizures.7
GEFS+ may possibly be more correctly termed autosomal dominant epilepsy with febrile seizures plus because of the large degree of phenotypic variability within families.8
Severe Myoclonic Epilepsy in Infancy (Dravet’s Syndrome)
Severe myoclonic epilepsy in infancy (SMEI), or Dravet’s syndrome, is rare. Individuals with previously normal development develop severe epilepsy in their first year of life, initially with myoclonic seizures, cognitive decline, and ataxia; subsequently, myoclonic seizures may abate and other seizure types predominate. Patients frequently suffer febrile seizures and status epilepticus. Mutations in SCN1A, accounting for 35% of sporadic cases9 and GABRG2 in only one familial case,10 have been discovered to underlie SMEI. Of interest is that different mutations in the same gene (SCN1A) can cause both familial epilepsy with a relatively mild phenotype and sporadic epilepsy with a devastating one.
Focal Epilepsies
Benign Familial Neonatal Convulsions
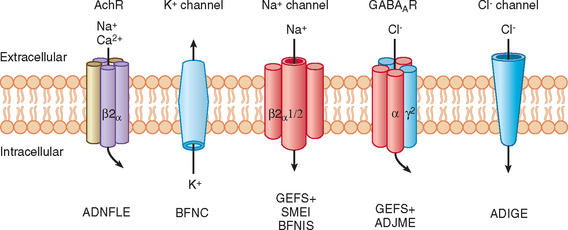
This is an autosomal dominant condition with high penetrance. It is characterized by the development of multiple seizure types on day 2 or 3 after birth, usually remitting spontaneously by 8 months of age, and is not associated with any psychomotor impairment. However, approximately 11% of these patients subsequently develop epilepsy. It was the first idiopathic epilepsy to be genetically linked. Causative mutations have been identified in voltage-gated potassium channel genes (KCNQ2 and KCNQ3) (Fig. 51-2).11–16 Of interest is that mutations in the homologous KCNQ1 gene result in the long QT and Jervell–Lange-Nielsen cardioauditory syndromes.17,18
< div class='tao-gold-member'>
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree