Chapter 43 Interbody Cages
Anterior cervical discectomy and cervical corpectomy for decompression of degenerative disease, trauma, tumor, and infection are commonly performed spinal operations. Options for reconstruction of the ventral column include structural autografts and allografts, as well as an evolving genre of prosthetic devices. The objectives of reconstruction are to restore a stable load-bearing ventral column, to maintain intervertebral height, and to establish an anatomic cervical lordosis when possible. The ultimate goal is for the construct to become biologically integrated into the native spine and to be replaced by living bone over time. For simple discectomy and single-level corpectomy, these end points are usually achievable with tricortical autografts harvested from the iliac crest or commercially available prefabricated cadaveric bone grafts. Complications such as graft collapse or extrusion are occasionally encountered. The primary objection to the use of structural autografts, however, is the relatively high rate of morbidity at the donor site from chronic pain, numbness, infection, hematoma, and cosmetic deformity. Such complications are reported to occur in 10% to 25% of cases.1–4 A common observation among spine surgeons is that the morbidity associated with the bone graft harvest frequently exceeds the morbidity related to the primary procedure. Pain at the bone graft harvest site has traditionally been reported by patients to be worse than that at the primary surgical site. This high rate of donor site morbidity has spurred the search for alternative reconstructive possibilities. Cadaveric prefabricated allografts have gained popularity over the past several years because of concerns over this donor site morbidity. Recently, several authors have advocated use of donor site alternatives to iliac crest (e.g., manubrium)5; however, long-term follow-up after using such an alternative is lacking. Structural allografts eliminate donor site problems but are associated with a slightly increased risk of pseudarthrosis and graft resorption, especially in smokers and diabetics. Although highly processed, lingering patient concerns remain about potential risk for disease transmission from allografts.
Reconstruction of a multilevel corpectomy bed poses yet a greater challenge. In fact, in a recent study by Uribe et al., the rate of early hardware failure and pseudarthrosis was higher after cervical corpectomy than after anterior cervical discectomy, suggesting that in the absence of specific pathology requiring removal of a vertebral body, multilevel anterior cervical discectomy and fusion (ACDF) using interbody cages and autologous bone graft could result in lower morbidity.6 Iliac crest and fibula are the best options available for autograft, but harvest of suitably long struts contributes significantly to patient morbidity. It can also be very difficult to match the graft to the cross-sectional and longitudinal geometry of the recipient site. These obstacles have prompted many surgeons to substitute fibular allograft as a simpler expedient. Fibular allografts are very straight and have a much smaller diameter than does material from the cervical corpus. These features enable it to fit readily into the recipient bed, but may not indicate optimal load transfer characteristics. Fibular allograft is composed almost exclusively of hard cortical bone. The mismatch in cross-sectional diameters and physical characteristics between the graft and recipient bone contributes to “pistoning” of the fibular strut through the adjacent central end plates, a condition commonly observed as the reconstructed segment foreshortens during graft incorporation, with resultant loss of lordosis.7 An additional concern is the heightened risk of nonunion that accompanies long-segment allograft constructs.
Interbody Cages
A variety of prosthetic interbody cages are now available for use in the cervical spine, both for disc space arthrodesis and to bridge the larger voids created by single- or multilevel corpectomy. Current devices are fabricated, either from titanium alloy or polymer, and can be classified into four categories: (1) screw-in, (2) box-type, (3) interbody fusion cage with integrated ventral fixation, or (4) a cylindrical design.8,9 Devices approved by the U.S. Food and Drug Administration (FDA) include hydroxyapatite spacer grafts, bioabsorbable implants, and artificial discs; they are now widely used and are not the primary focus of this chapter.
Interbody cages are intended to confer immediate structural integrity to the ventral spine and to provide instant support through the instantaneous axis of rotation. Although some surgeons have placed these as naked implants,10 more typically, they are employed as carriers for osteoinductive or osteoconductive materials whose purpose is to secure long-term stability through biologic integration with the recipient spine. In our experience, interbody cages are usually loaded with morselized autograft obtained from the cervical spine itself or from cancellous harvest from the sternum or iliac crest with negligible added morbidity. Alternatives such as allograft bone, hydroxyapatite, and “biologics,” including recombinant human bone morphogenetic protein (rhBMP), may even obviate this need.
Devices designed for disc space arthrodesis come prefabricated in a variety of sizes for direct implantation. Cages used for vertebral body replacement are provided as stock material, which can be rapidly modified to match an individual patient’s unique anatomic need. Many of these implants remain in development and have not been released by the FDA for clinical application in the United States at this time. The literature that bears on these devices includes animal-testing data,11 biomechanical studies,8,12,13 and clinical reports2,10,14–29; it is comparatively sparse and is devoid of any class 1 evidentiary material.
Screw-in Devices
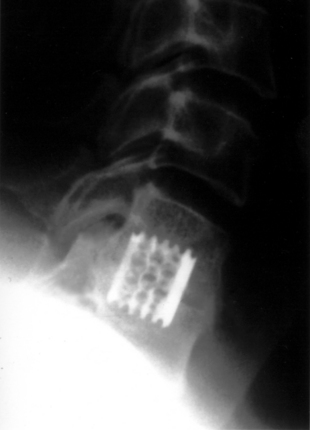
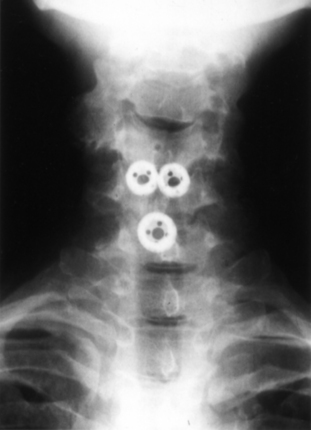
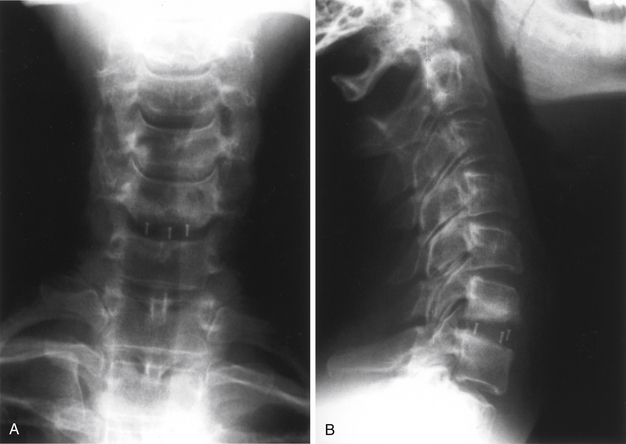
The Bagby and Kuslich cervical cage (BAK/C, Sulzer Spine-Tech, Minneapolis, MN) is the prototypic example of a screw-in design. The BAK/C cage is fundamentally an adaptation of a spinal instrumentation system already validated for disc space arthrodesis in the lumbar spine. After successful completion of a prospective randomized multicenter trial, it received FDA approval for use in the cervical spine.20 Similar to the larger lumbar devices, a BAK/C implant consists of a hollow, threaded cage with multiple side-wall fenestrations. The cages are manufactured from a titanium alloy and are provided in a 12-mm length, with a choice of 6-, 8-, 10-, and 12-mm diameters. Using a modification of the Cloward technique,30 it is usually inserted as a single, midline cage after reaming and tapping of the adjacent central end plates (Fig. 43-1). Alternatively, two smaller (6- or 8-mm) cages may be applied in a side-by-side fashion, although this necessarily limits the height of disc space distraction that can be achieved (Fig. 43-2). The cages are loaded with locally derived bone shavings, sometimes supplemented with allograft, according to volumetric needs. Sofamor Danek (Memphis, TN) has recently released the Affinity cage in North America. This is similar in concept to the BAK/C, but has a conically tapered configuration whose purpose is to help establish cervical lordosis.
In the BAK/C study a single-level “fusion rate” of nearly 100% was reported. No device-related failures or complications occurred, and other measures of clinical outcome appeared comparable to the noninstrumented ACDF “control” patients.20 Furthermore these outcomes were achieved without the morbidity associated with iliac crest bone graft harvest observed in the control group. However, certain concerns linger about the consequences of violating the central vertebral end plates, as is required for insertion of this device. Hacker observed a 20% incidence of postoperative “sagittal alignment abnormalities” at follow-up.2 Lordotic reaming and tapping techniques and a tapered expansile cage (Varilift, Advanced Spine, Irvine, CA) have been developed in an effort to address this issue.
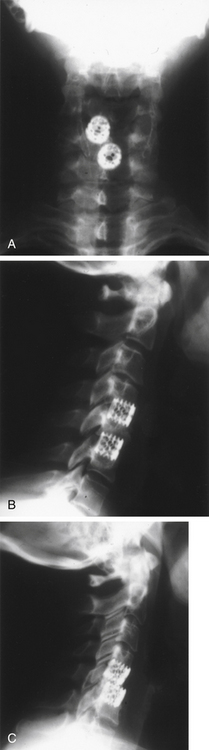
Because screw-in cages have a cylindrical cross section, the contact surface available for load transfer (and for fusion) is a comparatively narrow trough whose width is less than half of the cage diameter. This difference is mechanically disadvantageous in resisting lateral flexion, and the potential for subsidence of the implant with concomitant loss of cervical lordosis may be magnified if two or more adjacent segments are instrumented. Disc space restoration to an average height of 6 to 8 mm requires implantation of 8- to 10-mm diameter cages. When this is attempted at two or more adjacent levels, the resultant encroachment into the intervening body can be substantial and may necessitate offset placement of the devices and result in significant subsidence (Fig. 43-3). When Wang et al.31 studied 64 patients who underwent ventral decompression and interbody fusion with BAK/C, they found that the ventral height of intervertebral space decreased significantly after 1 year, when compared with the ventral height immediately after surgery. BAK/C subsidence was observed in nine patients, including five with single-level fusion, one with two-separated-level fusion, and three with double-adjacent-level fusion, according to the standard of loss of intervertebral height of more than 3 mm. BAK/C fusion was generally effective; however, neck pain tended to reoccur in the patients with cage subsidence and two of them needed revision surgery because of the recurrence of myelopathy with progressive neck pain. Although BAK/C was generally effective, the pitfalls in the design of the device have resulted in the observation of clinical subsidence, which, in some cases, required reoperation for the recurrence of symptoms.
Box Cages
In Smith-Robinson–type interbody fusions, the bony end plates are left intact, and a block-shaped structural graft with large, flat, superior and inferior contact surfaces is countersunk into the disc space.32 From a biomechanical standpoint this construct is superior because it retains the integrity of the end plates and provides a large surface area for load transfer and arthrodesis. Several box-type implant designs have been explored as alternatives to the more traditional tricortical autograft or structural allografts for Smith-Robinson fusion. These share a more or less rectangular configuration with a hollow core and fenestrations in their superior and inferior surfaces to allow the through growth of bone. Both titanium and polymer cages have been produced. The Rabea cage (Signus Medizintechnik, Alzenau, Germany), the Syncage (Synthes, Davos, Switzerland), and the Tibon cage (Biomet-Merck, Berlin, Germany) are representative of titanium box designs.
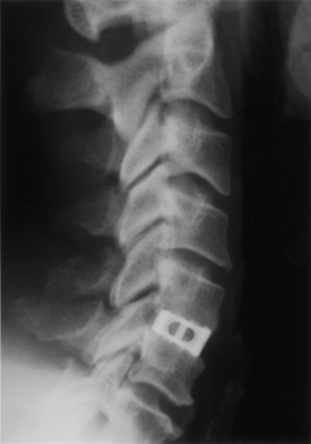
With FDA approval, the Rabea cage is currently available as a “cement restrictor.” It is made of forged titanium to ensure MRI compatibility and is prefabricated in a 12-mm width, 12- and 14-mm depths, and in heights ranging from 4 to 8 mm (Fig. 43-4). The Rabea is offered as a parallel end-plate design and as a “lordosed” version, with 5 degrees of divergence built in. The rostral and caudal surfaces also bear retentive serrations to engage the end plates and resist implant extrusion during neck flexion. Published experience with the Rabea cage is limited, but preliminary biomechanical studies and clinical reports are both favorable.10,12,15 As with other metallic devices, it is hard to judge fusion according to criteria of radiographically demonstrable bridging bone across the disc space and end plates. Alternative criteria including less than 2 degrees of angulation in dynamic radiographs and absence of any peri-implant bony lucency are substituted.
Carbon fiber box cages have found favor as intervertebral implants in the lumbar spine.33 These devices appear to be largely inert biologically; they exhibit good strength in all axes of applied stress, they are impact and fatigue resistant, their elastic modulus is purported to match well with that of the cortical bone of the recipient spine, and because they are radiolucent, they afford an opportunity for direct graft visualization and fusion assessment. Extending these advantages to a cervical interbody application, box-type polymer cages have been under active development abroad and are poised to enter the U.S. market. Most designs have been fabricated from polyetheretherketone (PEEK), some also including a carbon fiber component (Cervical IF Cage, DePuy AcroMed, Raynham, MA). The Rabea device is now offered as a PEEK cage, again with approval in the United States for use as a cement restrictor. Other major vendors, including Stryker Instruments (Kalamazoo, MI [Solis PEEK cage]), Medtronic Sofamor Danek (CornerStone-SR), and Synthes (CR), have polymer cages already in place in overseas markets (Fig. 43-5). Like the Rabea cage, they are offered in a variety of sizes and configurations, including parallel end-plates, lordotic, and superiorly convex designs to conform to individual disc space anatomic requirements. Shared features include a large central opening to contain graft material, ridged end plates to resist implant extrusion, and imbedded radiopaque markers to allow radiographic visualization of the device.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree