Fig. 1
Skull surface sutures and reference points. Stereotaxic surgery utilizes reference points created by the intersection of skull sutures to pinpoint the location of neuroanatomical structures in the brain . The most common reference points used are Bregma and Lambda. Bregma is the point at which the coronal suture intersects the sagittal suture. Lambda is the point that is in line with both the sagittal suture and the interaural line. Alternatively, True Lambda is the point at the intersection of the lambdoid suture and the sagittal suture. Shown is a diagram of the sutures and reference points found on the rat skull as well as a representative picture of a rat skull (corresponding to the area in the box on the diagram to the right) with the locations of the skull sutures and reference points demarcated
Combining the precision of stereotaxic surgery with the highly efficacious viral vectors now available, researchers can easily manipulate gene expression within discrete neural nuclei. Specifically, recombinant adeno-associated virus (rAAV ) vectors are favored for their high biosafety profile, low immunogenicity, and ability to transduce and achieve long-term gene expression in non-dividing cells of adult animals. Using rAAV, transduction can be achieved in structures as large as the striatum [2] or limited to structures as small as nuclei of the hypothalamus (Fig. 2) [3, 4]. Transduction kinetics and tropism can be optimized using particular rAAV pseudotypes and cell-specific promoters, to achieve tightly controlled transgene expression solely in cells of interest (Fig. 3) [2, 5–7].
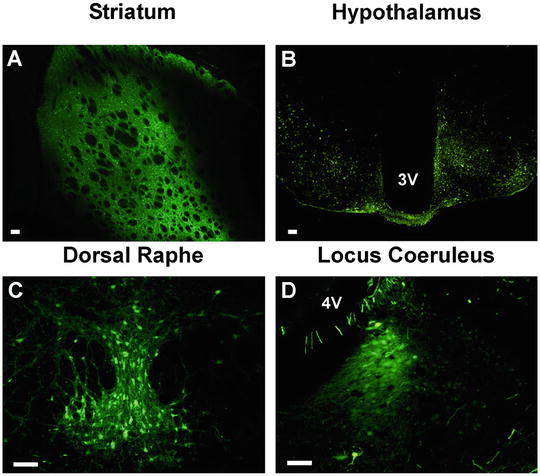
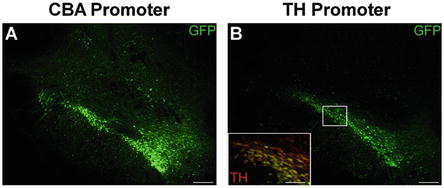

Fig. 2
Stereotaxic delivery of rAAV to discrete neural nuclei. rAAV 2/5 expressing GFP under control of the cytomegalovirus/chicken β-actin promoter was stereotaxically injected into various brain regions of the rat or mouse to demonstrate gene delivery and expression in discrete nuclei of the brain. Successful viral delivery, transduction and transgene expression is demonstrated in the rat striatum (a), the mouse mediobasal hypothalamus (b), the rat dorsal raphe nucleus (c) and the rat locus coeruleus (d). All coordinates, volumes of injection and flow rates for the shown stereotaxic injections are available in Table 1 (except the mouse hypothalamus which were bilateral, 250 nl injections performed at a 10° lateral angle at −1.75 anterior–posterior, ±1.25 mediolateral, and −6.2 dorsal ventral). Scale bars in all panels represent 100 μm. Abbreviations: 3 V third ventricle, 4 V fourth ventricle

Fig. 3
Cell-specific promoters increase specificity of transgene expression following stereotaxic delivery of rAAV. The precision of stereotaxic gene delivery can be improved by using cell-specific promoters to limit transgene expression to cells with known transcriptional profiles. Adult rats received unilateral stereotaxic injection of rAAV 2/5 into the midbrain. The expression cassette consisted of a GFP transgene under the control of either: (a) the ubiquitously expressed cytomegalovirus/chicken β-actin (CBA) hybrid promoter, or (b) the tyrosine hydroxylase (TH) promoter, which is active solely in catecholaminergic neurons. Under the control of the CBA promoter, GFP expression is observed throughout the majority of the midbrain, within many different cell types. In contrast, GFP expression under the control of the TH promoter is limited solely to the dopaminergic neurons of the substantia nigra and ventral tegmental area. Yellow cells in the inset of panel b represent the restricted expression of GFP (green) within TH immunoreactive neurons (red). Scale bars in panels a and b represent 200 μm. Scale bar within inset in panel b represents 50 μm
This chapter describes a protocol for attaining accurate and consistent delivery of rAAV vectors to the brain . Provided herein are detailed instructions for placing an animal in a stereotaxic frame, determination of skull surface reference points, localization of stereotaxic coordinates, and injection of viral vectors using a motorized micropump. Finally, due to the complications associated with utilizing viral vectors, special consideration will be given to the proper handling of virus in order to maximize transduction potential.
2 Materials
3.
Stereotaxic atlas (e.g., Paxinos and Watson).
4.
Isoflurane vaporizer with induction box and nose cone adapters.
5.
Isoflurane (Abbott Animal Health, Abbott Park, IL, USA).
6.
Programmable motorized micropump (e.g., QSI by Stoelting or equivalent).
7.
10 μL Hamilton syringe with 26-gauge needle (Hamilton, Reno, NV, USA).
8.
50 μL microcapillary pipette (Drummond Scientific, Broomall, PA, USA).
9.
Surgical drill (e.g., Osada electric Xl-30 W or equivalent).
11.
Intramedic polyethylene tubing (I.D. × O.D.: 0.588 × 0.965 mm; Clay Adams Brand, Sparks, MD, USA).
12.
Sigmacote siliconizing reagent (Sigma-Aldrich, St. Louis, MO, USA).
14.
Buprenorphine.
15.
Betadine surgical scrub (Thermo Fisher Scientific, Waltham, MA, USA).
16.
70 % ethanol.
17.
Sterile adhesive plastic surgical drape (e.g., Bioclusive by Johnson & Johnson, or equivalent).
18.
Sterile surgical gauze (Thermo Fisher Scientific, Waltham, MA, USA).
19.
Long cotton swabs.
20.
Scalpel with # 10 blade.
21.
11.5 cm straight fine scissors (F.S.T., Foster City, CA, USA).
22.
Fine curved forceps (e.g., Medical #7S forceps; F.S.T., Foster City, CA, USA).
23.
Serrated bulldog serrefines (F.S.T., Foster City, CA, USA).
24.
Autoclip stapler (F.S.T., Foster City, CA, USA).
26.
Lidocaine.
27.
Puralube vet ophthalmic ointment (Dechra Veterinary Products, Overland Park, KS, USA).
28.
Hair clippers.
29.
Isothermal heating pad.
30.
Parafilm (Neenah, WI, USA).
31.
Pipette and pipette tips (p20 and p200).
32.
Balanced salt solution (Alcon Laboratories, Fort Worth, TX, USA).
33.
1 and 10 mL syringe.
34.
25-gauge needle.
35.
1.5 mL Eppendorf tubes.
36.
3 % hydrogen peroxide.
37.
Sterile 0.9 % Saline.
38.
Calculator.
39.
Timer.
40.
Paper towel.
3 Methods
3.1 Preoperative Preparation
1.
Determine stereotaxic coordinates, volume of injection, and flow rate for the brain region to be injected (see Note 6 ). Table 1 shows a list of confirmed coordinates, volumes, and flow rates for various brain regions.
Table 1
Established stereotaxic coordinates of various neural nuclei
Brain region | Reference point | Anterior–posterior | Medio-lateral | Dorsal-ventral | Injection volume | Flow rate (μL/min) | Notes |
|---|---|---|---|---|---|---|---|
Striatum (single injection) | Bregma | 0 | +3.0 | −4 | 2 μL | 0.5 | Lower needle to site, inject immediately (4 min), wait 1 min retract needle 1 mm, wait 4 min before full retraction = total 9 min |
Striatum (double injection)a | Bregma | First site: 0 | First site: +3.0 | First site: −4.0 | 2 μL per injection | 0.5 | Lower needle to site, inject immediately (4 min), wait 1 min retract needle 1 mm, wait 4 min before full retraction = total 9 min |
Second site: −1.6 | Second site: +4.5 | Second site: −5.5 | |||||
Substantia nigra | Bregma | −5.4 | +2.0 | −7.2 from dura | 2 μL | 0.5 | Lower needle to site, inject immediately (4 min), wait 5 min retract needle = total 9 min |
Hippocampus | Bregma | −3.8 | +2.0 | −2.6 from dura | 2 μL | 0.5 | Lower needle to site, inject immediately (4 min), wait 5 min before full retraction = total 9 min |
Dorsal raphe | Bregma | −7.8 | −3.1 | −7.5 from skull | 2 μL | 0.5 | Stereotaxic arm at a 30° lateral angle. Lower needle to site, inject immediately (4 min), wait 5 min before full retraction = total 9 min |
Locus coeruleus | True Lambda | −3.7 | −1.2 | −6.1 from skull | 1.5 μL | 0.25 | Stereotaxic arm at a 12° angle in the anterior-posterior direction. Lower to site, inject immediately (6 min), wait 5 min before full retraction = total 11 min |
Hypothalamus | Bregma | −1.8 | +1.0 | −9.0 from Dura | 0.25 μL | 0.125 | Lower needle to site, inject immediately (2 min), wait 5 min before full retraction = total 7 min |
Motor cortex | Bregma | +1.6 | −2.6 | −1.6 from dura | 1.5 μL | 0.25 | Lower needle to site, inject immediately (6 min), wait 5 min before full retraction = total 11 min |
2.




Siliconize microcapillary pipettes, Eppendorf tubes, and pipette tips. To siliconize the microcapillary pipettes, place the pipettes in a beaker and use a handheld pipettor or transfer pipette to drip Sigmacote into the microcapillaries. Be sure fill the entire microcapillary with Sigmacote. Repeat process until the entire interior surface of the microcapillary is thoroughly coated with Sigmacote. To siliconize Eppendorf tubes, drip Sigmacote down all interior surfaces of the Eppendorf. Repeat until surface is thoroughly coated with Sigmacote. To siliconize pipette tips, infuse enough Sigmacote to fill the entire pipette tip, then expel contents. Repeat until entire interior surface of pipette tip is coated with Sigmacote. Drain any excess Sigmacote from microcapillary pipettes, Eppendorf tubes, and pipette tips and air-dry overnight in a hood (see Note 7 ).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree






