Functional Anatomy of the Visual System
Vascular Supply of Visual Pathways
Functional Anatomy of the Ocular Motor System
Supranuclear Control of Eye Movements
Associated Neurologic Abnormalities
Disorders of the Visual System
Nonarteritic Anterior Ischemic Optic Neuropathy
Giant Cell (Temporal) Arteritis
Oculomotor (III) Nerve Lesions
APPROACH TO DIAGNOSIS
Disorders that affect the ocular muscles, ocular motor (III, IV, and VI) cranial nerves, or visual or ocular motor pathways in the brain produce a wide variety of neuro-ophthalmic disturbances. Because the anatomic pathways of the visual and ocular motor systems traverse major portions of the brainstem and cerebral hemispheres, neuro-ophthalmic symptoms and signs are often valuable in the anatomic localization and diagnosis of neurologic disease.
FUNCTIONAL ANATOMY OF THE VISUAL SYSTEM
VISUAL INPUT
Visual information enters the nervous system when light, refracted and focused by the lens, creates a visual image on the retina at the back of the eye (Figure 7-1). The action of the lens causes this image to be reversed in the horizontal and vertical planes. Thus the superior portion of the visual image falls on the inferior retina and vice versa, and the temporal (lateral) and nasal (medial) fields are likewise reversed (Figure 7-2). The center of the visual field is focused at the fovea, where the retina’s perceptual sensitivity is greatest. Within the retina, photoreceptor cells (rods and cones) transduce incident light into neuronal impulses, which are transmitted by retinal neurons to the optic (II) nerve. At this and all other levels of the visual system, the topographic relations of the visual field are preserved.
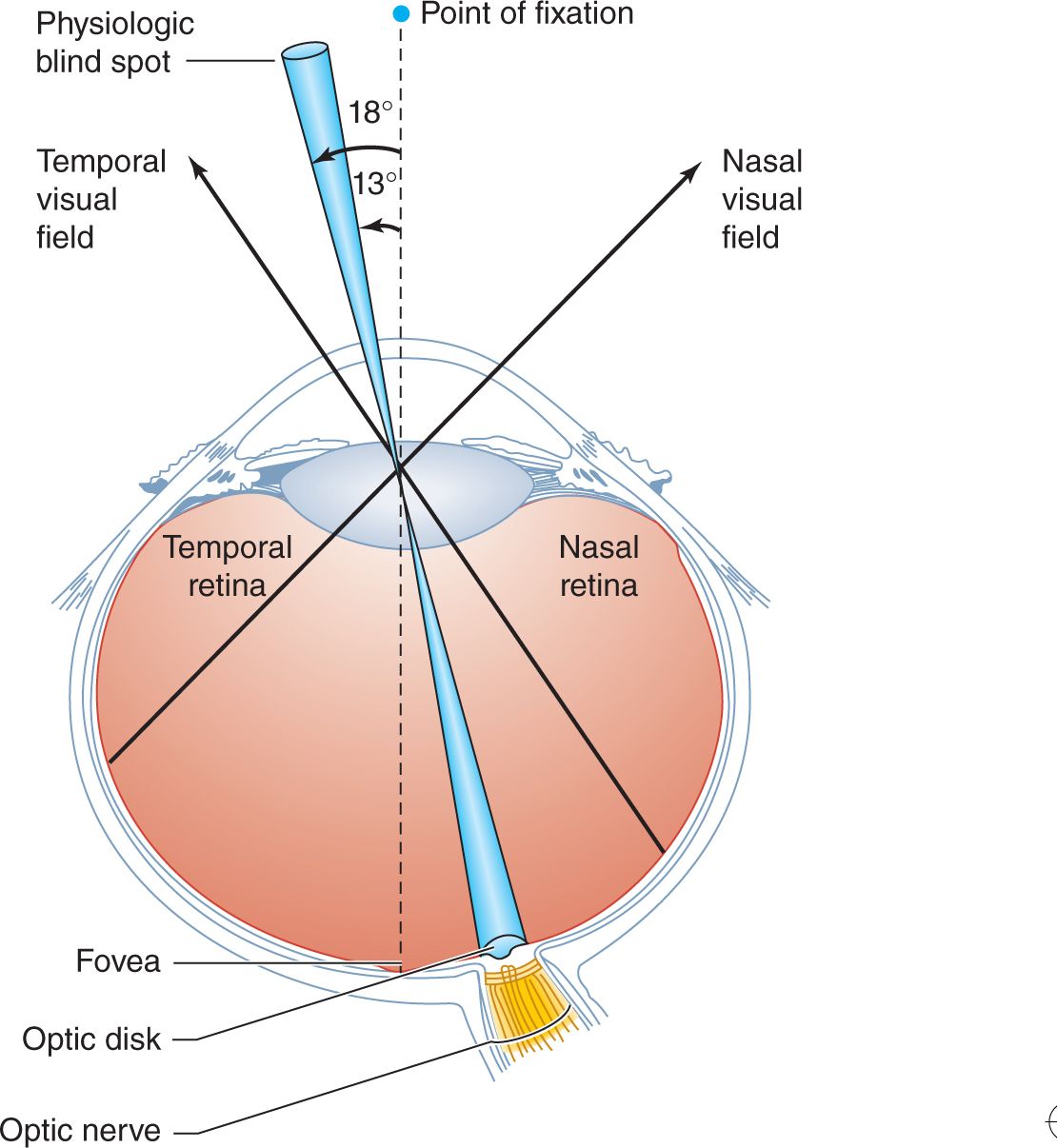
![]() Figure 7-1. Representation of the visual field at the level of the retina. The point of fixation is focused on the fovea, the physiologic blind spot on the optic disk, the temporal half of the visual field on the nasal side of the retina, and the nasal half of the visual field on the temporal side of the retina.
Figure 7-1. Representation of the visual field at the level of the retina. The point of fixation is focused on the fovea, the physiologic blind spot on the optic disk, the temporal half of the visual field on the nasal side of the retina, and the nasal half of the visual field on the temporal side of the retina.
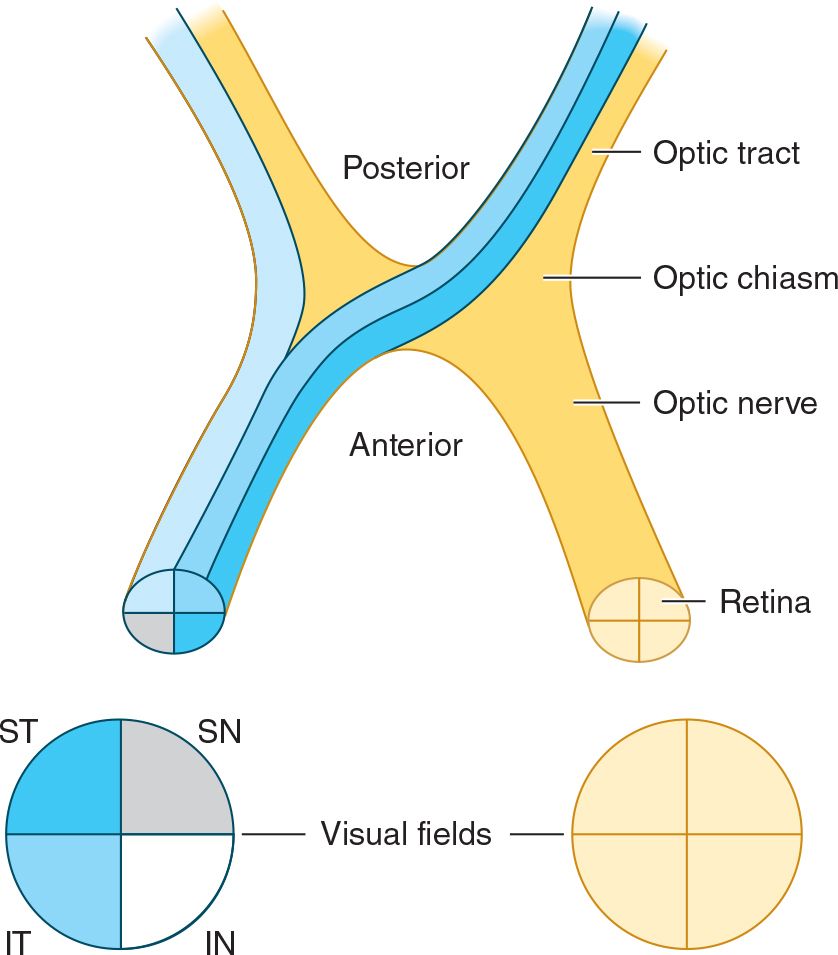
![]() Figure 7-2. Representation of the visual field at the level of the optic nerve, chiasm, and tract. Quadrants of the visual field are designated ST (superior temporal), IT (inferior temporal), SN (superior nasal), and IN (inferior nasal).
Figure 7-2. Representation of the visual field at the level of the optic nerve, chiasm, and tract. Quadrants of the visual field are designated ST (superior temporal), IT (inferior temporal), SN (superior nasal), and IN (inferior nasal).
PERIPHERAL VISUAL PATHWAYS
Each optic nerve contains fibers from one eye, but the nasal (medial) fibers, conveying information from the temporal (lateral) visual fields, cross in the optic chiasm (see Figures 1-13 and 7-2). As a result, each optic tract contains fibers not from one eye, but from one-half of the visual fields. Because of this arrangement, prechiasmal lesions affect vision in the ipsilateral eye and retrochiasmal lesions produce defects in the contralateral half of the visual field of both eyes (see Figure 1-13).
CENTRAL VISUAL PATHWAYS
The optic tracts terminate in the lateral geniculate nuclei, synapsing on neurons that project through the optic radiations to the primary visual or calcarine cortex (area 17), located near the posterior poles of the occipital lobes, and visual association areas (areas 18 and 19). Here, too, the visual image is represented in such a way that its topographic organization is preserved (Figure 7-3). The central region of the visual field (macula) is represented in the most posterior portion of the visual cortex, whereas the inferior and superior parts of the field (superior and inferior retina) are represented above and below the calcarine fissure, respectively.
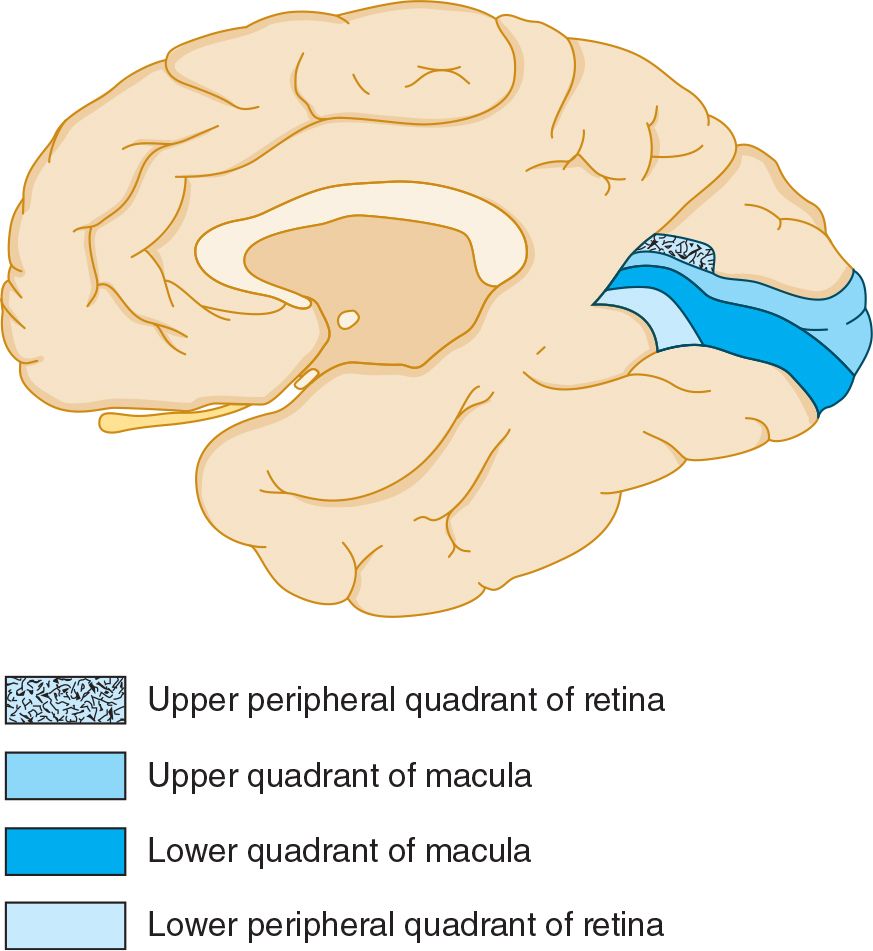
![]() Figure 7-3. Representation of the visual field at the level of the primary visual cortex, midsagittal view, shows the medial surface of the right occipital lobe, which receives visual input from the left side of the visual field of both eyes.
Figure 7-3. Representation of the visual field at the level of the primary visual cortex, midsagittal view, shows the medial surface of the right occipital lobe, which receives visual input from the left side of the visual field of both eyes.
VASCULAR SUPPLY OF VISUAL PATHWAYS
The vascular supply of the visual system is derived from the ophthalmic, middle cerebral, and posterior cerebral arteries (Figure 7-4); ischemia or infarction in the territory of any of these vessels can produce visual field defects.
1. Retina—The retina is supplied by the central retinal artery, a branch of the ophthalmic artery, which is itself a branch of the internal carotid artery. Because the central retinal artery subsequently divides into superior and inferior retinal branches, vascular disease of the retina tends to produce altitudinal (ie, superior or inferior) visual field deficits.
2. Optic nerve—The optic nerve receives arterial blood primarily from the ophthalmic artery and its branches.
3. Optic radiations—As the optic radiations course backward toward the visual cortex, they are supplied by branches of the middle cerebral artery. Ischemia or infarction in the distribution of the middle cerebral artery may thus cause loss of vision in the contralateral visual field (see Figure 1-13).
4. Primary visual cortex—The primary visual cortex is supplied principally by the posterior cerebral artery. Occlusion of one posterior cerebral artery produces blindness in the contralateral visual field, although the dual (middle and posterior cerebral) arterial supply to the macular region of the visual cortex may spare central (macular) vision. Because the posterior cerebral arteries arise together from the basilar artery, occlusion at the tip of the basilar artery can cause bilateral occipital infarction and complete cortical blindness—although, in some cases, macular vision is spared.
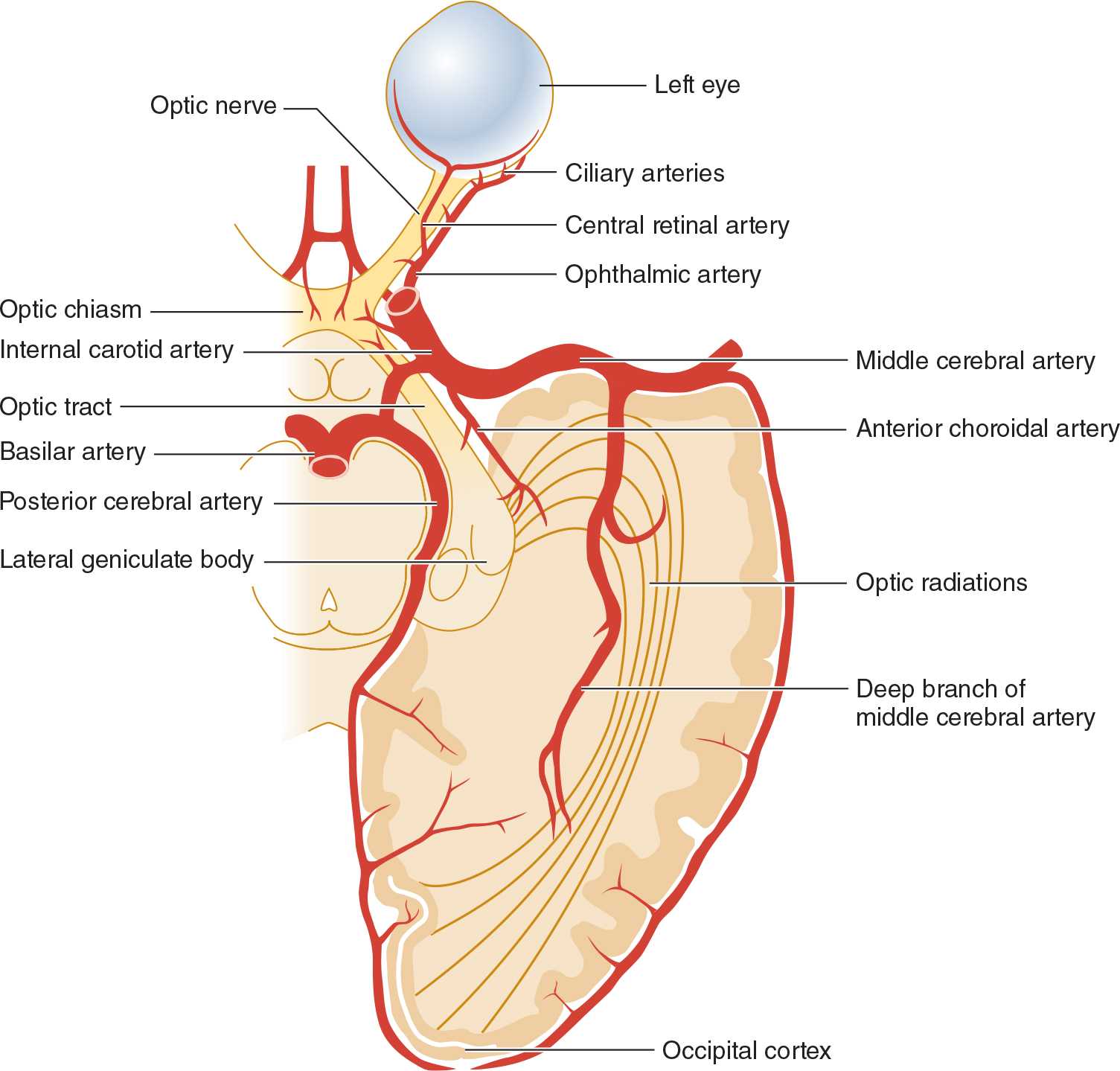
![]() Figure 7-4. Arterial supply of the visual system, viewed from below.
Figure 7-4. Arterial supply of the visual system, viewed from below.
FUNCTIONAL ANATOMY OF THE OCULAR MOTOR SYSTEM
EXTRAOCULAR MUSCLES
Movement of the eyes is accomplished by the action of six muscles attached to each globe (Figure 7-5). These muscles act to move the eye into each of six cardinal positions of gaze. Equal and opposed actions of these six muscles in the resting state place the eye in mid- or primary position, that is, looking directly forward. When the function of one extraocular muscle is disrupted, the eye is unable to move in the direction of action of the affected muscle (ophthalmoplegia) and may deviate in the opposite direction because of the unopposed action of other extraocular muscles. When the eyes are thus misaligned, visual images of perceived objects fall on a different region of each retina, creating the illusion of double vision, or diplopia.
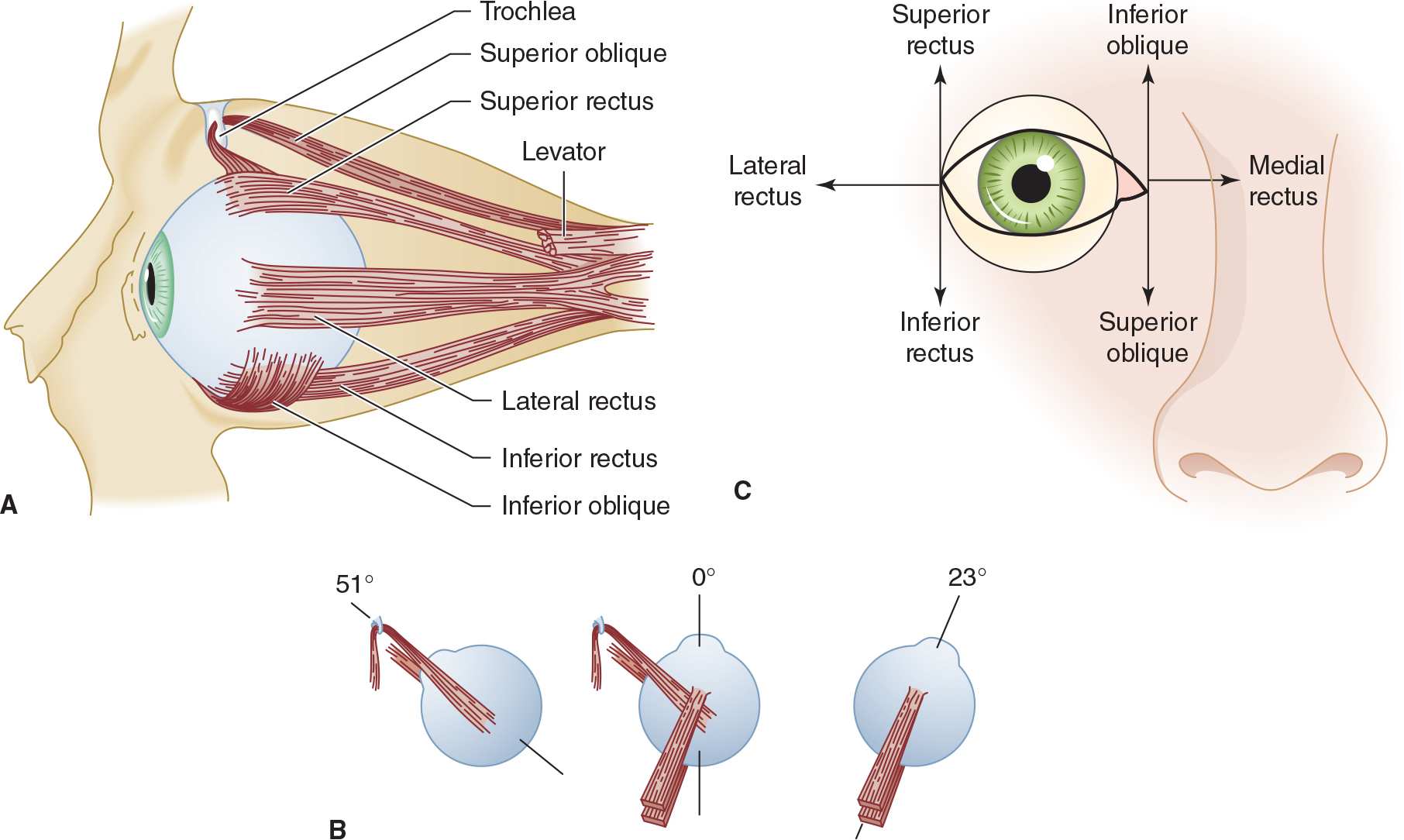
![]() Figure 7-5. Anatomy and function of extraocular muscles. (A) Extraocular muscles in the left orbit (lateral view). (B) An illustration of the right eye viewed from above in the primary position (center figure) showing the angle of attachment of the superior and inferior rectus muscles and the superior and inferior oblique muscles. With the eye directed to the right, the superior and inferior rectus muscles can now be examined as pure elevators and depressors of the globe (right image), and with the eye deviated to the left, the oblique muscles can now be examined as pure elevators and depressors of the globe as illustrated in C. (C) The six cardinal positions of gaze for testing eye movement. The eye is adducted by the medial rectus and abducted by the lateral rectus. The adducted eye is elevated by the inferior oblique and depressed by the superior oblique; the abducted eye is elevated by the superior rectus and depressed by the inferior rectus.
Figure 7-5. Anatomy and function of extraocular muscles. (A) Extraocular muscles in the left orbit (lateral view). (B) An illustration of the right eye viewed from above in the primary position (center figure) showing the angle of attachment of the superior and inferior rectus muscles and the superior and inferior oblique muscles. With the eye directed to the right, the superior and inferior rectus muscles can now be examined as pure elevators and depressors of the globe (right image), and with the eye deviated to the left, the oblique muscles can now be examined as pure elevators and depressors of the globe as illustrated in C. (C) The six cardinal positions of gaze for testing eye movement. The eye is adducted by the medial rectus and abducted by the lateral rectus. The adducted eye is elevated by the inferior oblique and depressed by the superior oblique; the abducted eye is elevated by the superior rectus and depressed by the inferior rectus.
CRANIAL NERVES
The extraocular muscles are innervated by the oculomotor (III), trochlear (IV), and abducens (VI) nerves. Because of this differential innervation of the ocular muscles, the pattern of their involvement in pathologic conditions can help to distinguish a disorder of the ocular muscles from a disorder that affects a cranial nerve. Cranial nerves that control eye movement traverse long distances to pass from the brainstem to the eye; they are thereby rendered vulnerable to injury by a variety of pathologic processes.
1. Oculomotor (III) nerve—The oculomotor nerve supplies the medial rectus, superior and inferior rectus, and inferior oblique muscles and carries fibers to the levator palpebrae (which raises the eyelid). It also supplies the parasympathetic fibers responsible for pupillary constriction. With a complete nerve III lesion, the eye is partially abducted, and cannot be adducted elevated, and depressed; the eyelid droops (ptosis), and the pupil is nonreactive.
2. Trochlear (IV) nerve—The trochlear nerve innervates the superior oblique muscle. Lesions of this nerve result in defective depression of the adducted eye.
3. Abducens (VI) nerve—Lesions of the abducens nerve cause lateral rectus palsy, with impaired abduction of the affected eye.
CRANIAL NERVE NUCLEI
The nuclei of the oculomotor (III) and trochlear (IV) nerves are located in the dorsal midbrain, ventral to the cerebral aqueduct (of Sylvius), whereas the abducens (VI) nerve nucleus occupies a similarly dorsal and periventricular position in the pons. Lesions involving these nuclei give rise to clinical abnormalities similar to those produced by involvement of their respective cranial nerves; in some cases, nuclear and nerve lesions can be distinguished.
1. Oculomotor (III) nerve nucleus—Although each oculomotor nerve supplies muscles of the ipsilateral eye only, fibers to the superior rectus originate in the contralateral oculomotor nerve nucleus, and the levator palpebrae receives bilateral nuclear innervation. Thus ophthalmoplegia affecting only one eye with ipsilateral ptosis or superior rectus palsy suggests oculomotor nerve disease, whereas ophthalmoplegia with by bilateral ptosis or a contralateral superior rectus palsy is probably due to a nuclear lesion.
2. Trochlear (IV) nerve nucleus—It is not possible to distinguish clinically between lesions of the trochlear nerve (see earlier) and those of its nucleus.
3. Abducens (VI) nerve nucleus—In disorders affecting the abducens nerve nucleus (rather than the nerve itself), lateral rectus paresis is often associated with facial weakness, paresis of ipsilateral conjugate gaze, or a depressed level of consciousness. This is because of the proximity of the abducens nerve nucleus to the facial (VII) nerve fasciculus, pontine lateral gaze center, and ascending reticular activating system, respectively. When a Horner syndrome (miosis of the pupil, ptosis, and sometimes segmental anhidrosis) accompanies an abducens nerve palsy, the lesion is in the cavernous sinus.
SUPRANUCLEAR CONTROL OF EYE MOVEMENTS
Supranuclear control of eye movements enables the two eyes to act in concert to produce version (conjugate gaze) or vergence (convergence and divergence) movements.
1. Brainstem gaze centers—Centers that control horizontal (lateral) and vertical gaze are located in the pons and pretectal region of the midbrain, respectively, and receive descending inputs from the cerebral cortex that allow voluntary control of gaze (Figure 7-6). Each lateral gaze center, located in the paramedian pontine reticular formation (PPRF) adjacent to the abducens nerve nucleus, mediates ipsilateral, conjugate, horizontal gaze via its connections to the ipsilateral abducens (VI) and contralateral oculomotor (III) nerve nucleus. A lesion in the pons affecting the PPRF therefore produces a gaze preference away from the side of the lesion and toward the side of an associated hemiparesis, if present. Disorders of vertical gaze, typically impaired upgaze, may result from mass lesions that exert downward pressure on the dorsal midbrain, such as pineal tumors (Parinaud syndrome).
2. Cortical input—The PPRF receives cortical input from the contralateral frontal lobe, which regulates rapid eye movements (saccades), and from the ipsilateral parietooccipital lobe, which regulates slow eye movements (pursuits). Therefore, a destructive lesion affecting the frontal cortex interferes with the mechanism for contralateral horizontal gaze and may result in a gaze preference toward the side of the lesion (and away from the side of associated hemiparesis). By contrast, an irritative (seizure) focus in the frontal lobe may cause gaze away from the side of the focus.
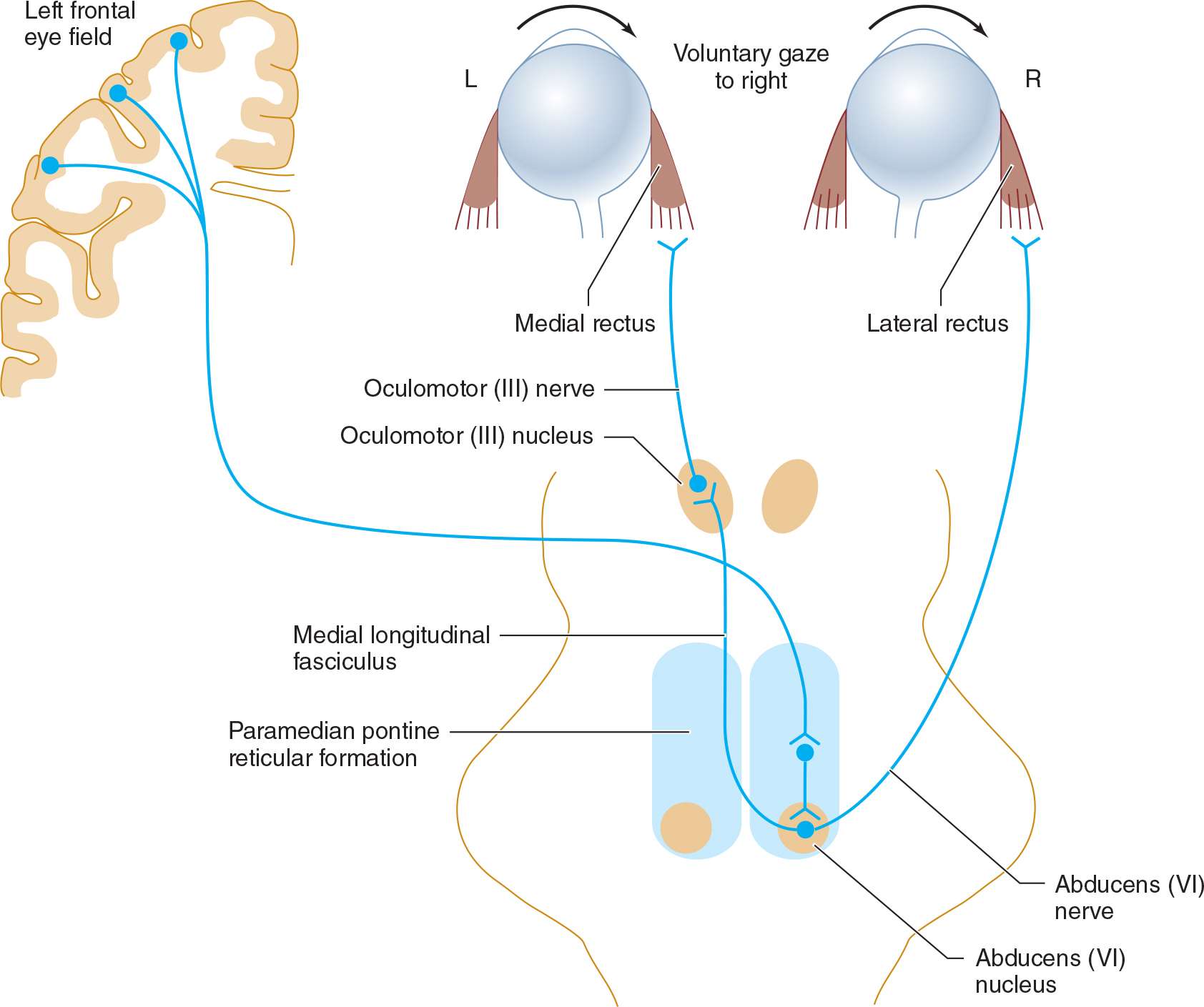
![]() Figure 7-6. Neuronal pathways involved in horizontal gaze.
Figure 7-6. Neuronal pathways involved in horizontal gaze.
HISTORY
NATURE OF COMPLAINT
The first step is to obtain a clear description of the complaint. Patients may only complain of vague symptoms, such as blurred vision, which provide little diagnostic information. Therefore, it is important to determine whether the patient means to describe decreased visual acuity in one or both eyes, loss of vision in part of the visual field, diplopia, an unstable visual image, pain in or about the eye, or some other problem.
TEMPORAL PATTERN OF SYMPTOMS
Once the nature of the complaint has been established, inquiries regarding its temporal pattern can provide clues to the underlying pathologic process.
1. Sudden onset—Vascular disorders that affect the eye or its connections in the brain tend to produce symptoms of sudden onset.
2. Slow onset—With inflammatory or neoplastic disease, symptoms usually evolve over a longer period.
3. Transient, recurrent symptoms—Transient and recurrent symptoms suggest a select group of pathologic processes, including intermittent ischemia, multiple sclerosis, and myasthenia gravis.
ASSOCIATED NEUROLOGIC ABNORMALITIES
The nature of any associated neurologic abnormalities, such as impaired facial sensation, weakness, ataxia, or aphasia, can help in localizing the site of involvement.
MEDICAL HISTORY
The history should be scrutinized for conditions that predispose the patient to neuro-ophthalmic problems.
1. Multiple sclerosis often involves the optic nerve or brainstem, leading to a variety of neuro-ophthalmic disorders. A history of disturbances that also involve other parts of the central nervous system should suggest this diagnosis.
2. Atherosclerosis, hypertension, and diabetes can be complicated by vascular disorders of the eye, cranial nerves, or visual or ocular motor pathways in the brain.
3. Endocrine disorders (eg, hyperthyroidism) can cause ocular myopathy.
4. Connective tissue disease and systemic cancer can affect the visual and ocular motor systems at a variety of sites in the brain or subarachnoid space.
5. Nutritional deficiencies may present with neuro-ophthalmic symptoms, as in the amblyopia (decreased visual acuity) associated with malnutrition and the ophthalmoplegia of Wernicke encephalopathy (thiamine deficiency).
6. Drugs (eg, ethambutol, isoniazid, digitalis, clioquinol) may be toxic to the visual system, and others (sedative drugs, anticonvulsants) commonly produce ocular motor disorders.
NEURO-OPHTHALMIC EXAMINATION
VISUAL ACUITY
 Assessment
Assessment
To identify neuro-ophthalmic problems, vision should be tested under conditions that eliminate refractive errors. Therefore, patients should be examined wearing their spectacles (a pinhole can be substituted if the corrective lenses usually worn are not available at the time of testing). Visual acuity must be assessed for each eye separately. Distant vision is tested using a Snellen eye chart, with the patient 6 m (20 ft) away. Near vision is tested with a Rosenbaum pocket eye chart held approximately 36 cm (14 in) from the patient. In each case, the smallest line of print that can be read is noted.
 Recording
Recording
Visual acuity is expressed as a fraction (eg 20/20, 20/40, 20/200). The numerator is the distance (in feet) from the test figures at which the examination is performed, and the denominator is the distance (in feet) at which figures of a given size can be correctly identified by persons with normal vision. For example, if a patient standing 20 ft away from the eye chart is unable to identify figures that can normally be seen from that distance but can identify the larger figures that would be visible 40 ft away with normal acuity, the visual acuity is recorded as 20/40. If the patient can read most of a given line but makes some errors, acuity may be recorded as 20/40–1, for example, indicating that all but one letter on the 20/40 line were correctly identified. When visual acuity is markedly reduced, it can still be quantified, though less precisely, by the distance at which the patient can count fingers (CF), discern hand movement (HM), or perceive light. If an eye is totally blind, the examination will reveal no light perception (NLP).
 Red–Green Color Vision
Red–Green Color Vision
Red–green color vision is often disproportionately impaired in optic nerve lesions and can be tested with colored objects such as pens or hatpins or with color vision plates.
VISUAL FIELDS
Evaluating the visual fields can be a lengthy and tedious procedure if conducted in an undirected fashion. Familiarity with the common types of visual field defects is important if testing is to be reasonably rapid and yield useful information. The most common visual field abnormalities are illustrated in Figure 7-7.
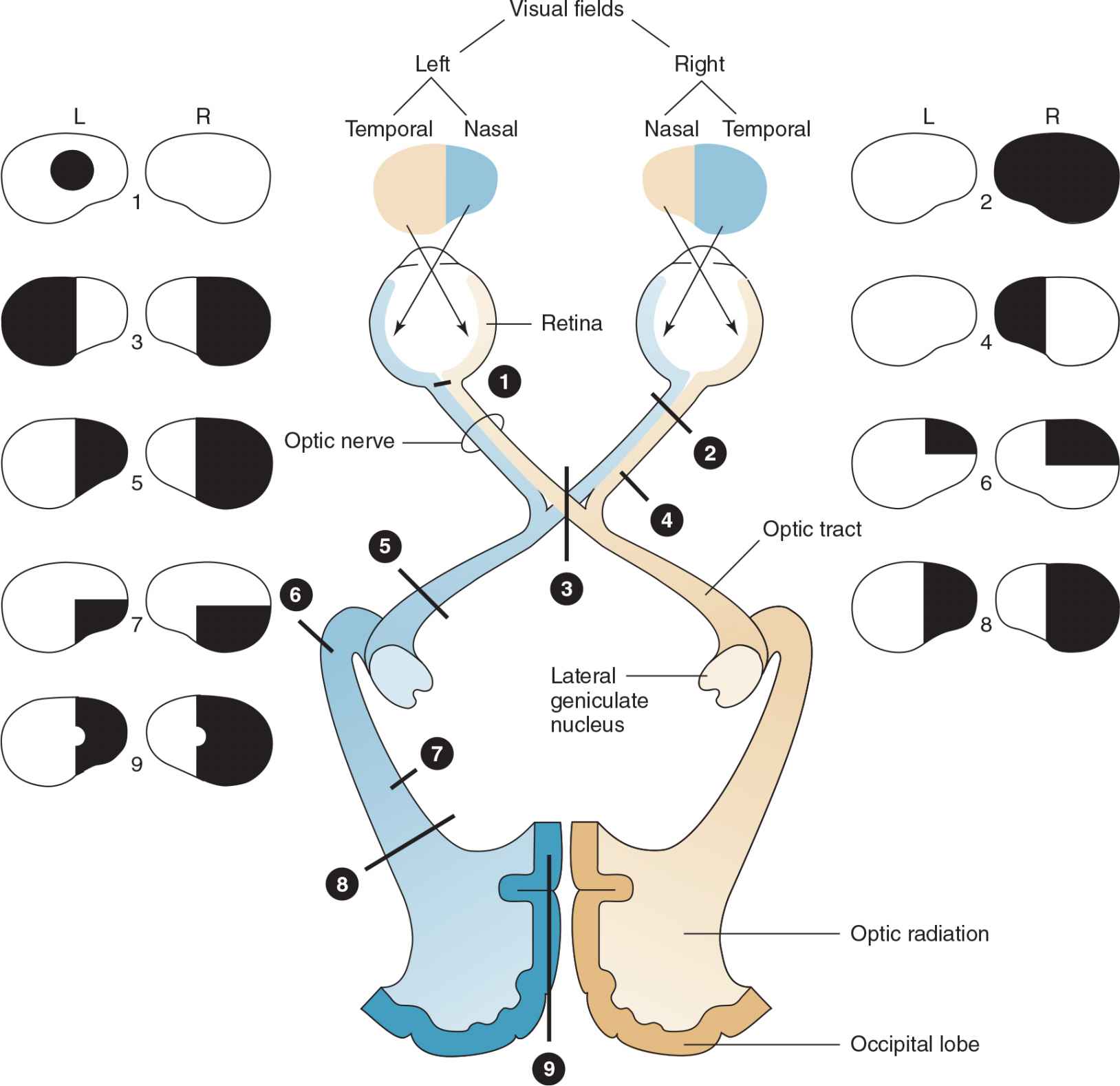
![]() Figure 7-7. Common visual field defects and their anatomic bases. 1. Central scotoma caused by inflammation of the optic disk (optic neuritis) or optic nerve (retrobulbar neuritis). 2. Total blindness of the right eye from a complete lesion of the right optic nerve. 3. Bitemporal hemianopia caused by pressure exerted on the optic chiasm by a pituitary tumor. 4. Right nasal hemianopia caused by a perichiasmal lesion (eg, calcified internal carotid artery). 5. Right homonymous hemianopia from a lesion of the left optic tract. 6. Right homonymous superior quadrantanopia caused by partial involvement of the optic radiation by a lesion in the left temporal lobe (Meyer loop). 7. Right homonymous inferior quadrantanopia caused by partial involvement of the optic radiation by a lesion in the left parietal lobe. 8. Right homonymous hemianopia from a complete lesion of the left optic radiation. (A similar defect may also result from lesion 9.) 9. Right homonymous hemianopia (with macular sparing) resulting from posterior cerebral artery occlusion.
Figure 7-7. Common visual field defects and their anatomic bases. 1. Central scotoma caused by inflammation of the optic disk (optic neuritis) or optic nerve (retrobulbar neuritis). 2. Total blindness of the right eye from a complete lesion of the right optic nerve. 3. Bitemporal hemianopia caused by pressure exerted on the optic chiasm by a pituitary tumor. 4. Right nasal hemianopia caused by a perichiasmal lesion (eg, calcified internal carotid artery). 5. Right homonymous hemianopia from a lesion of the left optic tract. 6. Right homonymous superior quadrantanopia caused by partial involvement of the optic radiation by a lesion in the left temporal lobe (Meyer loop). 7. Right homonymous inferior quadrantanopia caused by partial involvement of the optic radiation by a lesion in the left parietal lobe. 8. Right homonymous hemianopia from a complete lesion of the left optic radiation. (A similar defect may also result from lesion 9.) 9. Right homonymous hemianopia (with macular sparing) resulting from posterior cerebral artery occlusion.
 Extent of Visual Fields
Extent of Visual Fields
The normal monocular visual field subtends an angle of approximately 160 degrees in the horizontal plane and approximately 135 degrees in the vertical plane. With binocular vision, the horizontal range of vision exceeds 180 degrees.
 Physiologic Blind Spot
Physiologic Blind Spot
Within the normal field of each eye is a 5-degree blind spot, corresponding to the optic disk, which lacks receptor cells. The blind spot is located 15 degrees temporal to fixation in each eye.
 Measurement Techniques
Measurement Techniques
Like visual acuity, the visual field must be examined separately for each eye.
1. Confrontation (Figure 7-8) is the simplest method for visual field testing. The examiner stands at about arm’s length from the patient, with the eyes of both patient and examiner aligned in the horizontal plane. The eye not being tested is covered by the patient’s hand or an eye patch. The examiner closes the eye opposite the patient’s covered eye, and the patient is instructed to fix on the examiner’s open eye. Now the monocular fields of patient and examiner are superimposed, which allows comparison of the patient’s field with the examiner’s presumably normal field. The examiner uses the index fingers of either hand to locate the boundaries of the patient’s field, moving them slowly inward from the periphery in all directions until the patient detects them. The boundaries are then defined more carefully by determining the farthest peripheral sites at which the patient can detect slight movements of the fingertips or the white head of a pin. The patient’s blind spot can be located in the region of the examiner’s own blind spot, and the sizes of these spots can be compared using a pin with a white head as the target. The procedure is then repeated for the other eye.
2. Subtle field defects may be detected by asking the patient to compare the brightness of colored objects presented at different sites in the field or by measuring the fields using a pin with a red head as the target.
3. In young children, the fields may be assessed by standing behind the child and bringing an attention-getting object, such as a toy, forward around the child’s head in various directions until it is first noticed.
4. A gross indication of visual field abnormalities may be obtained in obtunded patients by determining whether they blink in response to a visual threat—typically the examiner’s finger—brought toward the patient’s eye in various regions of the field.
5. Although many visual field deficits are detectable by these screening procedures, more precise mapping of the fields requires the use of standard tangent screen testing or automated perimetry techniques.
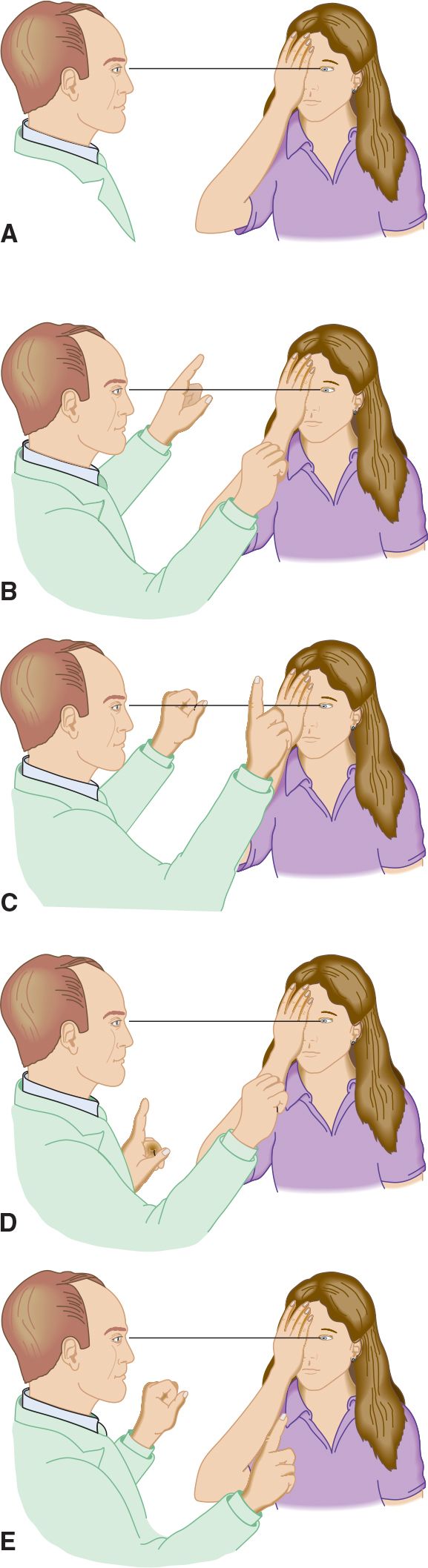
![]() Figure 7-8. Confrontation testing of the visual field. (A) The left eye of the patient and the right eye of the examiner are aligned. (B) Testing the superior nasal quadrant. (C) Testing the superior temporal quadrant. (D) Testing the inferior nasal quadrant. (E) Testing the inferior temporal quadrant. The procedure is then repeated for the patient’s other eye.
Figure 7-8. Confrontation testing of the visual field. (A) The left eye of the patient and the right eye of the examiner are aligned. (B) Testing the superior nasal quadrant. (C) Testing the superior temporal quadrant. (D) Testing the inferior nasal quadrant. (E) Testing the inferior temporal quadrant. The procedure is then repeated for the patient’s other eye.
OPHTHALMOSCOPY
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


