2
Pediatric Febrile Seizures
In this chapter, we discuss the basics of febrile seizures and the controversies pertaining to them and provide insights into the understanding of these seizures from the Consequences of Prolonged Febrile Seizures in Childhood (FEBSTAT) study.
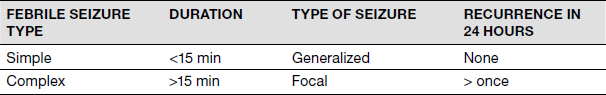
Febrile seizures are generally defined as seizures occurring in the presence of a fever higher than 100.4°F in the absence of central nervous system infection, metabolic disturbance, or a previous history of afebrile seizures (1). These are the most common types of convulsive events in infants and young children under 5 years of age and remain the most common childhood neurological emergency affecting 2% to 5% of children. The typical age of the occurrence is between 3 months and 5 years with a peak incidence at 9 to 20 months. The prevalence is 3% to 7% in children up to 7 years, with the above range due to the variability in definition and inclusion criteria in different settings (2). Febrile seizures are classified as simple or complex (Table 2.1) (3). Febrile status epilepticus is a febrile seizure lasting more than 30 minutes and is an extreme form of a complex febrile seizure. It constitutes 5% of febrile seizures (4) and contributes to 25% to 30% of pediatric status epilepticus (5).
ETIOLOGY
Febrile seizures are thought to be an age-specific phenomenon. These are genetically and environmentally modified (6). Febrile seizures are more common in monozygotic (7) than dizygotic twins (9%–22% versus 11%), and by linkage analysis, several loci have been associated with febrile seizures (FEB 1–FEB 11) (8). Some of these, among other loci, are on chromosome 8q13-q21 (FEB1), 19p (FEB2), 2q23-q24 (FEB3 or SCN1A,), 5q14-q15 (FEB4), 6q22-q24 (FEB5), 18p11 (FEB6), 21q22, 5q31.1-q33.1, 3p24.2-p23, and 19q13.1 (SCN1B). These genes encode for various proteins, which are involved in the electrical activity of neurons, such as sodium channels (SCN1A/SCN1B) or the receptors for the transmitter gamma-aminobutyric acid (GABRG2 in the region 5q34). These genes are also frequently affected in other febrile seizure–related epileptic syndromes, such as generalized epilepsy with febrile seizures plus (GEFS+) and severe infantile myoclonic epilepsy (Dravet syndrome) as described later.
TABLE 2.1 Classification of Febrile Seizures
As far as environmental influences are concerned, risk factors implicated include increasing temperature, fever mediators, genetic factors, and hyperthermia-induced hyperventilation with alkalosis. Although all rat/mouse models of seizures seize with hyperthermia, the threshold temperature needed to cause the seizure varies, with different strains implicating genetic factors. Febrile seizure susceptibility is modified by genes coding sodium channels, gamma-aminobutyric acid (GABA) A receptors, and interleukins. Fever and hyperthermia (after hot water baths or with anticholinergic medications) are both associated with the release of interleukin 1β in the brain (9), which then increases neuronal excitability via glutamate and GABA (10,11).
Fever induced by human herpes virus 6 (HHV6) infection is highly associated with febrile seizures (12). Hyperthermia-induced hyperventilation and therefore alkalosis seems to provoke neuronal excitability (9), contributing to seizure pathophysiology where latency between fever and seizure is greater than 30 minutes (9).
SHOULD CHILDREN WITH FEBRILE SEIZURES BE VACCINATED? DO VACCINATIONS CAUSE FEBRILE SEIZURES?
Vaccine administration is the second leading cause of febrile seizures. Febrile seizure occurrence in children is a serious concern because it leads to public apprehension of vaccinations (13). Seizures following vaccination are most likely associated with the febrile episode and not with vaccination itself. Vaccination causes febrile seizure onset in approximately one-third of the patients with Dravet syndrome (14). The condition of vaccine-associated encephalopathy is poorly defined. However it is thought to be a clinical state where a previously well infant develops seizures and encephalopathy soon after vaccination. In a seminal paper by Berkowicz et al., out of 14 patients with vaccine-associated encephalopathy, 11 were found to have mutations in their sodium channel and all 14 had a diagnosis of a specific epilepsy syndrome, suggesting that the concept of vaccine-associated encephalopathy may be more a myth than a fact (15). According to a recently published position statement by the Italian league against epilepsy (16), although it is apparent that vaccines for diphtheria-tetanus-pertussis (DTP) and measles-mumps-rubella (MMR) are at a higher risk for febrile seizures, the rate of febrile seizures is similar in children with and without a personal history of previous febrile seizures. Vaccine-induced febrile seizures are no more frequent than febrile seizures with any other cause of fever. The risk of nonfebrile seizures following vaccine-induced febrile seizures is not higher than in children who have not shown vaccine-induced febrile seizures.
EVALUATION OF FEBRILE SEIZURES
The main role of the physician responding to a child with a febrile seizure is to investigate for the cause of fever and rule out meningitis. The latest American Academy of Pediatrics (AAP) guideline (17) unfortunately focuses only on simple febrile seizures and states that in the evaluation of simple febrile seizures, serum electrolytes, complete blood count, calcium, and phosphorus should not be considered. A CT scan and an electroencephalogram (EEG) are not indicated in the evaluation of simple febrile seizures. A lumbar puncture is indicated in any child who appears ill or has signs of meningismus. In general, children under 12 months of age may not exhibit signs of meningitis and a low threshold should be maintained for doing a lumbar puncture. In children between the ages of 6 months and 18 months, lumbar puncture may be considered when pretreated with antibiotics and when children are deemed to be deficient in their immunizations for Haemophilus influenzae type B or Streptococcus pneumoniae or documentation for these vaccinations is incomplete.
No such guidelines exist for complex febrile seizures. The role of neuroimaging in complex febrile seizure (not status epilepticus) is limited. In a retrospective review by Teng et al. of 76 children with complex febrile seizures, none was found to have any intracranial pathology. On the other hand in a prospective study of 159 children with a first febrile seizure and a brain MRI completed within 7 days, Hesdorffer et al. found abnormalities in 20 of the 159 children. However children who had both a focal as well as a prolonged seizure were more likely to have an abnormality (n = 14) on MRI imaging such as focal cortical dysplasia or abnormality on white matter signal. This suggested that the MRI abnormality was perhaps predisposing the patient to seizures in the setting of a fever (18,19). In a more recent study evaluating the role of emergent new imaging (CT scan) in the emergency room (ER) after a complex febrile seizure (20), 268 patients with complex febrile seizures had emergent imaging completed. Of these 268 patients, only 4 patients had an abnormality: Two had intracranial hemorrhage, one had acute disseminated encephalomyelitis, and one patient had focal cerebral edema; three of these patients had obvious abnormalities like bruising suggesting nonaccidental trauma and then on neurological examination, had nystagmus or altered mental status.
The role of EEG in the workup of febrile seizures is also controversial. An EEG may remain slow for up to 7 days after a febrile seizure. In a recent case control study of 36 patients with febrile seizures who had abnormal EEG compared to 87 patients with normal EEG after febrile seizures (21), 9/36 with EEG abnormalities were more likely to have epilepsy. In a recent Cochrane review, no randomized controls trials were found to show evidence for the use of EEG after a complex febrile seizure. Guidelines do not exist to assist in the understanding of when an EEG may be valuable in these patients (22).
Lumbar puncture guidelines in complex febrile seizures are no different from those for simple febrile seizures, meaning that a lumbar puncture is indicated in any child who appears ill or has signs of meningismus or has incomplete or unclear documentation of immunization.
FEBRILE STATUS EPILEPTICUS
Febrile status epilepticus is defined as a febrile seizure lasting more than 30 minutes and may be conceived as an extreme form of a complex febrile seizure. Risk factors for developing febrile status epilepticus include younger age at onset of febrile seizure, lower temperature at onset of seizure, and longer duration of recognized temperature before onset of febrile seizure (23).
PROGNOSIS OF FEBRILE SEIZURES
After a single febrile seizure, the risk of recurrence is 30%. After two or more febrile seizures, the risk increases to 50%. Other risk factors for recurrence include young age at onset, a history of febrile seizures in a first-degree relative, low degree of fever while in the emergency department, and a brief duration between the onset of fever and the initial seizure (24,25). Risk of future epilepsy after simple febrile seizures is negligible at 1% to 2.4% and that after a complex febrile seizure is 4% to 6%. Risk factors for future epilepsy include complex febrile seizure, developmental delay, and family history of epilepsy. These risk factors are different from the risk factors for febrile seizure recurrence (younger age at onset, lower temperature at time of febrile seizure, and family history of febrile seizures) (26–28).
MANAGEMENT OF SIMPLE AND COMPLEX FEBRILE SEIZURES
Parental education is critical in further management of febrile seizures. Parents should be educated about seizure first aid and also be trained to administer rescue medication at home for seizures lasting 5 minutes. Medications used in the prehospital termination of seizures include rectal diazepam (0.3–0.5 mg/kg/dose) and buccal or intranasal midazolam (0.2–0.3 mg/kg/dose). Emergency seizure management will be the same in patients with febrile seizures as patients with afebrile seizures (29). After securing airway, breathing, and circulation in a patient still actively seizing, an intravenous benzodiazepine is administered as the first line of treatment followed by an intravenous load of a second-line drug—which may vary from hospital to hospital (at our institution we use intravenous fosphenytoin given as 20–30 phenytoin equivalents/kg/dose). Although prophylactic antipyretics may give a patient symptomatic comfort, they do not prevent the occurrence of febrile seizures (30). In addition, prophylactic anticonvulsant medications are not indicated in the treatment of febrile seizures (31).
FEBRILE SEIZURES AS THE PRESENTING FEATURE OF OTHER EPILEPSIES
Febrile seizures may be the presenting feature of Dravet syndrome, GEFS+, and PCDH19 (Table 2.2) and may also be seen in a subset of patients who develop future mesial temporal sclerosis (2,32,33). The association of febrile seizures and future hippocampal sclerosis remains a topic of much debate. Febrile seizures in school-aged children may be seen in association with a recently recognized entity called febrile infection–related epilepsy syndrome (FIRES).
FEBSTAT STUDY
Febrile status epilepticus occurs in 5% of patients with febrile seizures. The FEBSTAT study was designed to prospectively examine the association between prolonged febrile seizures and the development of hippocampal sclerosis and associated temporal lobe epilepsy, one of the most controversial issues in epilepsy. FEBSTAT is a prospective, multicenter study. Inclusion criteria are children, aged 1 month to 6 years of age, presenting with a febrile seizure lasting 30 minutes or longer, based on ambulance, emergency department, and hospital records, and parental interview. At baseline, procedures include an MRI study and EEG recording done within 72 hours of status, and a detailed history and neurologic examination. Baseline development and behavior are assessed at 1 month. The baseline assessment is repeated, with age-appropriate developmental testing at 1 and 5 years after enrollment as well as at the development of epilepsy and 1 year after that. Telephone calls every 3 months document additional seizures. Two other groups of children are included: a “control” group consisting of children with a first febrile seizure ascertained at Columbia University and with almost identical baseline and 1-year follow-up examinations and a pilot cohort of febrile status epilepticus from Duke University (24). Initial results of the study have been published and these results are described in the following text.
TABLE 2.2 Epilepsy Syndromes That May Start With Fever-Induced Seizures
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree






