2 Recent Advances in Epidemiology of Brain Tumors
Refinements in Histological Categorization of Brain Tumors and Associated Developments
The International Classification of Diseases for Oncology, Third Edition (ICD-O-3) is the standard classification system for the registration of cancers in the United States (and most areas of the world) and contains widely-accepted histologic categories of brain tumors. Brain tumors are classified into the following major histologic groupings: tumors of neuroepithelial tissue (hereafter referred to as glioma, including astrocytoma [grade II], anaplastic astrocytoma [grade III], glioblastoma [grade IV], oligodendroglioma, and ependymoma), tumors of meninges (including meningioma and hemangioblastoma), germ cell tumors, and tumors of the sellar region (including pituitary tumors and craniopharyngioma). Glioma is the most common histological category, followed by meningioma, the category that includes the highest proportion of benign brain tumors (approximately 96% of meningiomas are considered benign).1 Criteria for classifying brain tumors have varied substantially across time and geographic region. Prior to the past decade, most epidemiologic studies presented results for crude categories such as “all central nervous system (CNS) tumors” or “brain tumors.” It is now well established that both the descriptive and analytic epidemiology of brain tumors varies considerably according to histologic grouping. A meaningful contribution of the Central Brain Tumor Registry of the United States (CBTRUS), which collects information on brain tumors occurring among residents of 19 U.S. states, has been the presentation of descriptive statistics according to categories detailed in the ICD-O-3.1
Progress in Understanding the Descriptive Epidemiology of Brain Tumors
Glioma incidence rates increased during the 1970s and 1980s (probably reflecting the use of new diagnostic imaging technologies2) and have remained relatively stable since the 1980s. Incidence rates for high grade gliomas among older age groups have increased over time from the late 1970s to the early 1990s, as have those for oligodendroglioma and mixed tumor histologies at the expense of less specific histologies.2 A similar description of American meningioma incidence rates over time is not possible, because benign brain tumors were only recently required to be reported to American central cancer registries. However, Klaeboe et al.3 report an increase in European meningioma incidence rates, which is also probably explained by new imaging technology introduced in the 1970s.
For all brain and CNS tumors combined, benign and malignant (brain tumors accounting for approximately 88%), the age-adjusted average annual (2000 to 2004) incidence rate for females (17.2 per 100,000 females) is greater than that for males (15.8 per 100,000 males).1 Table 2-1 shows age-adjusted average annual (2000 to 2004) incidence rates and median ages at diagnosis for the major histologic groupings and their selected common histologic subtypes of brain tumors.1 As shown in Table 2-1, men have higher incidence rates of gliomas, germ cell tumors and cysts, while women have a higher incidence rate of meningiomas. As described in a following section, results from analytic studies concerning reproductive and hormonal factors have suggested compelling possible explanations for the sex difference in glioma and meningioma risks.
TABLE 2-1 Number of Cases, Median Ages at Diagnosis and Age-Adjusted Average Annual (2000-2004) Incidence Ratesa of Primary Brain Tumors (Major Histologic Groupings and Selected Histologic Subtypes), According to Sex.
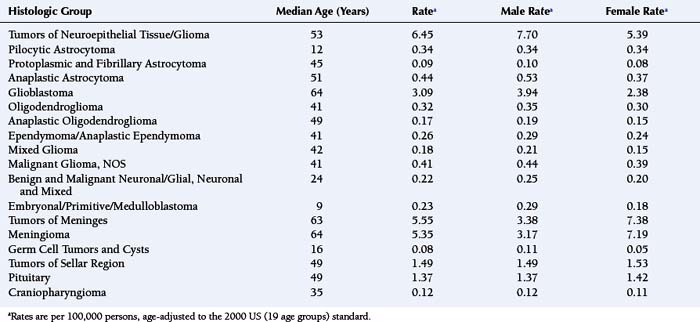
Newly diagnosed glioma is approximately two times more common among whites than among blacks, as are germ cell tumors. In contrast, meningioma incidence rates are similar between whites and blacks.1 Although there is no well-accepted mechanism for the race differences in glioma risk, differences in the distribution of human leukocyte antigens (HLA) among races, described later in the chapter, may explain it. In the United States, for diagnoses made between 2000 and 2004, the median age at diagnosis of a primary brain tumor was 57 years.1 For all major histologic groups except germ cell tumors and cysts, incidence rates increase with advancing age.1 Rates of cancers of nearly every anatomic site increase with advancing age; still, the reason for this increase is not known.
Brain tumor incidence rates vary moderately by geographic region in the United States and throughout the world.1,4 However, differences in diagnostic practices, completeness of reporting, access to and quality of health care, make geographic and especially international comparisons difficult to interpret.5,6
Brain tumor survival time varies greatly by histologic type and age at diagnosis. In every adult age group, the lowest relative 2-year survival is found for patients with glioblastoma multiforme (GBM), ranging from 30.4% among those aged 20 to 44 years to 1.3% among those aged 75 years and older.1 In general, within histologic types, survival time decreases with advancing age. The mechanisms for the strong and consistent inverse association between age and survival are poorly understood and deserve further exploration. Among adult (ages 20 years and older) patients diagnosed with primary malignant brain tumors between 2000 and 2005, only 32.5% and 23.7% survived 2 and 5 years, respectively, from the time of diagnosis.4 Although there has been little change in the poor survival rates for patients diagnosed with GBM, for adult (ages 20 years and older) patients diagnosed with all malignant brain tumors combined, 2-year survival has increased from 21.7% in 1975 to 31.9% in 2004.4 Among patients diagnosed with glioma, the largest improvements in survival occurred among patients with earlier stage glioma and those who were younger than 65 years of age. Population-based data from Norway and Finland suggest that survival for patients diagnosed with meningioma also improved between the 1950s and 1990s.7,8 McCarthy et al.9 estimate 5-year survival of 69% for meningioma, with those younger at diagnosis and those with benign meningioma having a more favorable prognosis.9 It is likely that improvements in imaging technology, which allow the earlier identification of tumors, explain the progress for both glioma and meningioma. GBM probably has only a brief preclinical period; therefore, there may be little opportunity for technology-associated improvement in GBM survival. In the past decade, there has been very little improvement in 2-year survival among adults diagnosed with a primary malignant brain tumor (1.4% difference between the 2-year survival among patients diagnosed in 1994 and those diagnosed in 2004). The only relatively important difference in length of survival following GBM diagnosis, in the past decade, has resulted from concomitant addition of, and maintenance with, the chemotherapeutic agent, Temodar, which has improved median survival time for GBM patients by only 2.5 months.10
Advances in Identifying Prognostic Factors for Brain Tumors
Previous research has shown that GBM prognosis is associated with the following factors: age, marital status, tumor size, Karnofsky Performance Scale (KPS) score, patient condition before radiation therapy, degree of necrosis, enhancement on preoperative magnetic resonance imaging studies, therapeutic approach including extent of resection and capacity for complete resection, volume of residual disease, location of tumor, patient deterioration, presurgical serum albumin level, and persistent hyperglycemia 1 to 3 months following surgical resection.11–16
Most recent advances in the search for prognostic factors related to GBM and other high-grade gliomas have focused on molecular or serologic factors or on inherited genetic variation. Loss of heterozygosity (LOH) on chromosome 10q has been associated with shorter duration of survival from GBM,17,18 and the combined LOH on 1p and 19q may afford a more favorable prognosis to GBM patients.18 For oligodendroglioma, it is now well established that the combined tumor loss of 1p and 19q confers a more favorable prognosis.19 Results submitted by Yang et al. showed that two genotypes associated with the 19q deletion region, GLTSCR1-exon-1 and ARCC2-exon-22, are independent predictors of glioma survival.20 Results reported in the past decade provide evidence for the following as prognostic indicators for GBM and other glioma subtypes: p53 mutation and expression,18,21–29 overexpression or amplification of epidermal growth factor receptor (EGFR),22,24,25,27–29 CDKN2A alterations and deletions,22,24,27 and MDM2 amplifications.21,24,27,29,30 For example, EGFR expression is associated with nearly three-fold poorer survival among anaplastic astrocytoma patients.29 In addition, Wrensch et al.29 reported that glutathione-S-transferase (GST) theta (T)1 deletion afforded a less favorable prognosis for glioma patients, while higher survival is afforded to glioma patients with the ERCC1 (a DNA excision repair gene) C8092A polymorphism.
Recently, we have learned that p53 protein expression probably decreases with advancing age,22,31 and investigators have reported interactions between age and molecular prognostic factors. For example, Simmons et al.31 showed that there was a shorter survival time among younger patients whose tumors overexpressed EFGR but had normal p53 immunohistochemistry.31 Age-dependent associations between GBM survival and 1p and CDKN2A have also been identified.22
GBM survival time varies with change in the MNS16A human telomerase polymorphism. Median survival time is 24.7 months for the SS-genotype, compared to 14.0 months and 13.1 months for the SL- and LL-genotypes respectively.32 These results suggest that MNS16A may be used as a biomarker of treatment success.
In order to develop and progress, brain tumors must evade anti-tumor immunity. Recently, several immunological factors have been implicated in glioma prognosis. GBM patients with elevated immunoglobulin (Ig) E live an average of 9 months longer than do patients with lower or normal IgE levels.29 Furthermore, amplification of interleukin (IL)-6, a cytokine which stimulates immune response, is significantly associated with decreased GBM survival time.33
Prognostic factors for meningioma patients have not been as thoroughly evaluated as those for glioma, perhaps because meningioma patients have more favorable prognoses. The recent requirement of central cancer registries to report benign brain tumors may increase our knowledge of demographic and geographic factors related to meningioma prognosis. Generally, younger meningioma patients survive longer than do older patients. A large study revealed that age, tumor size, and surgical and radiation treatments were associated with benign meningioma survival; however, only age and surgical and radiation treatments were associated with malignant meningioma survival.9 Loss of tumor suppressor in lung cancer-1 (TSLC1) protein and abnormalities of chromosome 14 have been associated with meningioma prognosis.34,35 We know little about the molecular and genetic factors that determine meningioma prognosis, but studies of such characteristics are being conducted at the present time.
Established Environmental Causal Factors for Brain Tumors
There are consistent and strong results from prospective studies of people exposed to ionizing radiation, providing unquestionable evidence of a linear dose-response association between ionizing radiation exposure and glioma risk.36 Exposure to ionizing radiation comes from therapeutic and diagnostic medical procedures, occupation, atmospheric testing of nuclear weapons, natural sources, industrial accidents, and atomic bomb explosions. Therapeutic doses of ionizing radiation probably contribute to the development of only a small proportion of brain tumors, as exposure to therapeutic levels of ionizing radiation is rare.37 Atomic bomb survivors have higher incidence rates of glioma and meningioma, as well as of schwannoma and pituitary tumors,38 and meningioma risk increases with estimated radiation dose to the brain.39 Ionizing radiation used to treat conditions such as tinea capitis in infants and children is associated with elevated relative risks for both glioma and meningioma, along with nerve sheath tumors and pituitary adenoma.38,40,41 There are mixed results pertaining to diagnostic and therapeutic x-rays of the head and neck.42–45 Second primary brain tumors occur more frequently than expected among patients previously treated for brain tumors with radiation therapy46; however, these results are confounded by the fact that people with higher grade tumors are more likely both to receive radiation and to have a recurrence. Future studies should consider the potential for underreporting of ionizing radiation exposure and imprecise estimates of age at first exposure.36 There may be interaction between ionizing radiation and both age at exposure and genetic variation that may mediate exposure; future studies should consider these possible interactions.
In addition to rare mutations in penetrant genes and ionizing radiation, exogenous hormone use among women is now an established risk factor for meningioma. In part because females have a greater meningioma risk than do males, investigators have examined factors associated with estrogen (menopausal status, ages at menarche and menopause, parity, and uses of oral contraceptives and hormone replacement therapy [HRT]). The ratio of the female-to-male meningioma incidence rate has increased in European countries over time with the increased use of hormone replacement therapy by perimenopausal and postmenopausal women.3 Several results suggest that meningioma risk is greatest among women during their reproductive years7,47,48; however, SEER statistics reveal that ratios of female-to-male meningioma incidence rates are greatest (greater than 2.5) during the ages 30 to 54 years.4 There is no consistent or convincing evidence that parity and oral contraceptive use are associated with meningioma risk.47–50 However, results from a population-based case-control study conducted by Wigertz et al.48 revealed elevated meningioma risks among women who had used long-acting hormonal contraceptives (odds ratio [OR] for at least 10 years of use = 2.7; 95% CI: 0.9-7.5) and postmenopausal women who had ever used HRT (OR = 1.7; 95% CI: 1.0-2.8). While exogenous female hormones probably play a role in the development of some meningiomas, our understanding of mechanisms governing their role is limited, perhaps in part because the menstrual and reproductive factors that have been examined are insufficient to accurately characterize lifetime estrogen or other hormonal exposure.
Probable Causal Factors for Brain Tumors
FAMILY HISTORY
The progressive accumulation of genetic and/or epigenetic alterations, permitting cells to evade normal regulatory mechanisms and/or escape destruction by the immune system, is thought to govern the development of glioma and meningioma, although these mechanisms have not yet been fully defined.51–53 Evidence for the presence of genetic involvement in the causal pathways of glioma and meningioma is demonstrated most simply by studies which have shown an increased risk of brain tumors in close relatives of brain tumor patients, especially those with gliomas. Although brain tumors clearly aggregate in families, it can be difficult to distinguish shared environmental exposures from inherited characteristics. In fact, Grossman et al.54 showed that brain tumors occur commonly in families with no known predisposing hereditary disease, and that the pattern of occurrence in many families suggests environmental, rather than genetic, causes. However, important results presented by Malmer et al.55 suggest that first-degree relatives, but not spouses, have a significantly increased brain tumor risk.
ALLERGIC AND ASSOCIATED IMMUNOLOGICAL CONDITIONS AND GLIOMA
There is consistent evidence for an association between allergy-related immune responses and glioma risk. Over the past two decades, results from ten case-control56–65 and one of two cohort studies66 show that self-reported allergies are inversely related to glioma risk. Linos et al.67 conducted a formal meta-analysis of a subset of these studies and found that self-reported allergies are related to a 40% reduction in the risk of glioma (OR = 0.61; 95% CI: 0.55-0.67). Further evidence for this inverse association was contributed by Wiemels et al.68 who found that total IgE levels were lower in glioma cases than in controls. Although mechanisms governing potential protection have not been identified, they may arise from the anti-inflammatory effects of IL- 4 and IL-13 cytokines involved in allergic and autoimmune disease69 or from increased tumor immunosurveillance among those with allergies and autoimmune disease.70 It is also possible that the inverse association results from immune suppression by a preclinical tumor.71
In addition to their role in allergic conditions, IL-4 and IL-13 cytokines also inhibit growth of glioma cell lines. IL-4 is strongly expressed during brain injury,72 where invading T cells may be a source of this cytokine,73 and IL-4 increases the number of T-cell precursors in GBM patients.74 Barna et al.73 found that three normal astrocytic, two low-grade astrocytoma, and three out of four GBM cell lines they evaluated expressed IL-4R alpha receptors. However, IL-4 suppressed DNA synthesis and cell proliferation only in the normal astrocytic and low-grade astrocytoma cell lines, not in the GBM cell lines. IL-4 could play a role in the inhibition of GBMs that arise from astrocytomas, but it may not be involved in de novo GBMs.75 In view of a possible role for IL-4 and IL-13 in both allergic conditions and glioma, Schwartzbaum et al.63 identified polymorphisms of the IL-4R alpha and IL-13 genes that increase allergic condition risk. Although these germline genetic variants are not sensitive indicators of the presence of allergic conditions, they do provide a measure of risk of these conditions free of recall bias. The working hypothesis was that individuals with IL-4R alpha or IL-13 polymorphisms that increase allergic condition risk would have a decreased risk of GBM. Using data from a small case-control study (111 GBM cases, 422 controls), the authors found results consistent with their hypothesis; each of the two IL-4R alpha and IL-13 single nucleotide polymorphisms (SNPs) associated with increased allergic condition risk were also related to decreased GBM risk. Wiemels et al.76 confirmed their finding for one of the IL-13 SNPs in a larger case-control study of glioma (456 glioma cases, 541 controls). Furthermore, they reported that this IL-13 SNP was inversely associated with IgE levels among controls (p = 0.04). However, they did not find associations between the IL-4R alpha SNPs and glioma as had Schwartzbaum et al. They did note a borderline association between an IL-4R alpha haplotype (OR = 1.5; 95% CI: 1.0-2.3) and glioma. They also saw that a rare IL-4 haplotype was associated with decreased glioma risk (OR = 0.23; 95% CI: 0.07-0.83).
A larger study of the original four IL-4R alpha and IL-13 genetic variants by Schwartzbaum et al.77 did not provide strong support for their original observations. Nonetheless, they found an IL-4R alpha haplotype associated with GBM (OR = 2.26; 95% CI: 1.13-4.52) and inversely related to self-report of hay fever or asthma among controls (OR= 0.39; 95% CI: 0.16-0.98). Although Wiemels et al. also found suggestive evidence for an association of between an IL-4R alpha haplotype and glioma, when they restricted their haplotype to the same IL-4R alpha SNPs that Schwartzbaum et al. examined, they observed no evidence for an association with glioma (OR = 1.13; 95% CI: 0.83-1.53).
Immunosuppressive regulatory T-cells and their associated cytokines TGF-beta and IL-10 repress effective anti-glioma reactions and may provide a conceptual and mechanistic framework to explain an indirect relationship between allergies and anti-glioma immune reactions. The unique architecture of the brain does not exclude glioma from immune system interaction, although immune responses may be attenuated compared to those found in other organs. In addition, there is now evidence for infiltration of T and B cells into the brains of brain tumor patients; the enhancement of such responses is likely to form the basis of future effective glioma therapies. In recent in vitro studies of glioma, human glioma cell lines were found to secrete immunosuppressive cytokines that can selectively recruit regulatory T cells into the tumor microenvironment.78 In addition, Chahlavi et al.79 demonstrated that glioma cell lines mediate immunosuppression by promoting T cell death through tumor-associated antigens and gangliosides. Two of the major immunosuppressive cytokines that are present in both the glioma microenvironment and the peripheral blood of glioma patients, IL-10 and TGF-beta, induce immune tolerance, thereby inhibiting allergy and asthma.80 Elevated IgE concentrations may therefore indicate low levels of immunosuppression and the resulting ability to conduct anti-tumor immunosurveillance against incipient glioma. Alternatively, the relative absence of allergies in glioma patients may merely show that these tumor-induced cytokines have suppressed the immune system.
Results pertaining to HLA—cell surface molecules that mediate interactions of tumor cells with the host immune response, in part by presenting antigenic peptides to T-lymphocytes—also suggest the importance of immunological responses in glioma development. Facoetti et al.81 found that HLA class I antigen loss was significantly (P<0.025) correlated with tumor grade: HLA class I antigens were lost in approximately half of GBM tumors, but only in 20% of grade II astrocytoma tumors; selective HLA-A2 antigen loss was observed in approximately 80% of GBM lesions and half of the grade II astrocytoma tumors. GBM is positively associated with the HLA genotype B*13 and the HLA haplotype B*07-Cw*07 (P=0.01 and P<0.001, respectively), and is inversely associated with the genotype Cw*01.82 Interestingly, these results could partially explain the increased GBM incidence among whites, because B*07 and B*07-Cw*07 are more common among whites than among nonwhites.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








