Spinal Meningoceles
Spinal meningoceles are defined as congenital herniations of dura and arachnoid through a bony defect in the spine. Occasionally, nerve roots may be contained within the sac, but the spinal cord is usually normal and is not contained within the sac. There are multiple classifications for spinal dysraphism,1 the most common of which separates closed lesions from open lesions. Meningoceles are considered by some to be a form of closed spinal dysraphism, consisting of skin-covered defects in the vertebral arch. Closed spinal dysraphism is further divided into lesions with a subcutaneous mass and those without a mass. Meningoceles usually fall into the former category, along with lipomyelomeningoceles and terminal myelocystoceles. Anterior and lateral meningoceles usually do not have an associated subcutaneous mass because of their location. In contrast to closed spinal dysraphism, open spinal dysraphism includes cystic lesions, which are not entirely skin-covered, such as myelomeningoceles, myeloceles, and rachischisis.
The true prevalence of spinal meningoceles is hard to measure as meningoceles are often grouped with myelomeningoceles.2,3 The total incidence of spina bifida in the United States is 0.3 to 0.4 per 1,000 live births.4 Spinal meningoceles occur in 1 per 15,000 to 20,000 births and are less common than myelomeningoceles, with less than 1 meningocele for every 20 myelomeningoceles.5 The majority (95%) of meningoceles occur in the lumbar spine, and most are skin-covered. Associated hydrocephalus is seen in fewer than 5% of cases. Many meningoceles contain associated nerve roots within the sac and are therefore not technically true meningoceles as the definition would suggest; rather, they represent an outward pouching of dura associated with a normal spinal cord and nerve roots. Microscopic evaluation can show ganglion cells and glial nodules as an extension from the central canal that may also be contained within the sac.3
23.1 Pathophysiology
The pathophysiology of meningoceles depends upon the location and position within the spine. Posterior lumbar and posterior sacral meningoceles are thought to occur secondary to defects in secondary neurulation.5 Anterior and lateral meningoceles occur in association with generalized mesenchymal abnormalities, as may be seen with neurofibromatosis type 1 (NF-1) and Marfan syndrome. In addition, there may be a ball valve effect that results in enlargement of the meningocele sac overtime.
23.2 Classification
The term meningocele encompasses a heterogeneous group of mostly congenital lesions that occur adjacent to the spinal column. In the case of posterior spinal meningoceles, there is a congenital herniation of dura and cerebrospinal fluid (CSF) through a bony defect in the posterior arch. Posterior meningoceles occur most commonly in the lumbar and sacral spine, but they may also occur in the cervical and thoracic spine.6 Anterior and lateral spinal meningoceles result from generalized mesenchymal abnormalities, and although most are congenital, they may progress and be detected postnatally. Posttraumatic or post-procedural meningoceles can occur after disruption of the dura but are considered pseudomeningoceles. We focus on congenital meningoceles in this chapter. For purposes of this discussion, spinal meningoceles are categorized based on a posterior or anterior/lateral location as well as on level of the spine (cervical, thoracic, lumbar, or sacral).
23.3 Posterior Meningoceles
23.3.1 Lumbar and Sacral Meningoceles
Presentation
Patients with meningoceles usually present after recognition of a skin-covered dorsal mass (▶ Fig. 23.1) and (▶ Fig. 23.2). Meningoceles may also be diagnosed on prenatal ultrasound, although the detection rate is less than that for open spinal dysraphic lesions because there are no indirect signs such as Chiari 2 malformation (banana sign) and frontal bossing (lemon sign), which may occur with myelomeningoceles.7 Alpha-fetoprotein levels should be normal with the skin-covered defect, in contrast to the elevated levels present with open spinal dysraphism. The neurologic examination is often normal because most meningoceles contain only spinal fluid without neural tissue. However, when they are associated with other anomalies, such as tight filum terminale, tethered cord, hydromyelia, split-cord malformation, and neurenteric cyst, there may be neurologic compromise at presentation.8,9 Spinal meningoceles may be associated with additional spinal anomalies, so it is important to obtain magnetic resonance (MR) imaging of the entire spine. MR imaging will show a dorsal fluid-filled sac that is hyperintense on T2 sequences (▶ Fig. 23.3). The entire spine should be evaluated because occult spinal lesions may be present in up to 81% of patients with meningoceles.9
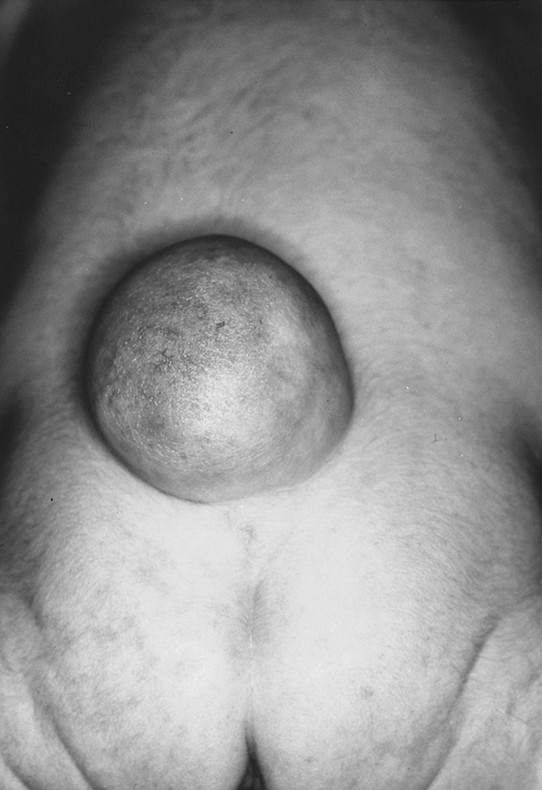
Fig. 23.1 Typical lumbosacral meningocele in a newborn. Overlying skin is normal. The neonate had no neurologic deficit. The meningocele was repaired, and the patient has remained completely normal.
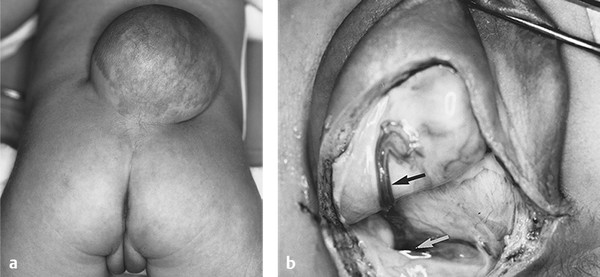
Fig. 23.2 (a) Newborn with a larger, more pedunculated meningocele in the lumbosacral region. The skin is fully epithelialized, although mildly dysplastic. The patient’s neurologic examination was normal and has remained so. (b) After the meningocele dome had been opened, a fibroglial nodule, densely adherent to the meningocele sac, was noted (black arrow) and resected flush with the spinal cord to minimize subsequent tethering. The white arrow points to the pedicle where the meningocele connected to the lumbar subarachnoid space.
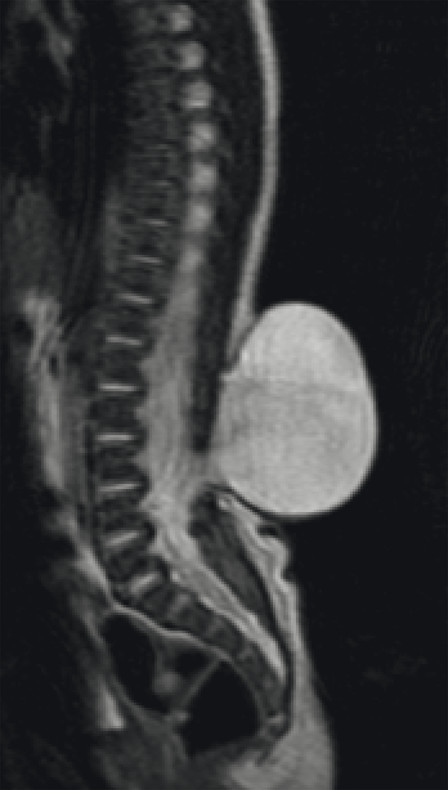
Fig. 23.3 Sagittal T2-weighted magnetic resonance image of a lumbar meningocele.
Differential Diagnosis
The differential diagnosis of a posterior skin-covered cystic lesion includes meningocele, lipomyelomeningocele, myelocystocele, sacrococcygeal teratoma, and pseudomeningocele. Meningoceles, in contrast to lipomyelomeningoceles, may transilluminate. Sacrococcygeal teratomas are located at the level of the coccyx, more caudally than most meningoceles. A pseudomeningocele usually presents after prior surgery or trauma and occurs through an iatrogenic defect rather than a congenital one.
Treatment
Repair of a posterior meningocele is usually recommended at the time of presentation to prevent subsequent tethering, infection, growth of the sac, and CSF leak. Treatment of a meningocele consists of removal of the sac and repair of the defect. Although more than 90% are not associated with neurologic deficits, it is important to perform an intradural exploration and evaluate for any cord tethering before the stalk is amputated9 because there may be intradural fibrous bands extending from the spinal cord into the sac. It is occasionally necessary to perform laminectomies and explore one to two levels above and below the lesion for tethering bands. The sac may be ligated and amputated if no neural tissue is present.
Follow-up
If there is clinical concern, patients who undergo surgery for spinal meningoceles may be followed for recurrence of the meningocele or signs and symptoms of subsequent tethering, which may result in weakness, urologic changes, or scoliosis. In some cases, follow-up with MR imaging may be performed.
23.3.2 Cervical Meningoceles
Cervical meningoceles are thought to arise from incomplete fusion of the posterior aspect of the neural tube when the cutaneous ectoderm fails to separate from the neuroectoderm.10 This results in an abnormal connection of the neural tube to the skin. Cervical meningoceles, myelomeningoceles, and myelocystoceles are often grouped together as cervical spinal dysraphism and account for 4 to 8% of patients with spina bifida cystica.11 Cervical meningoceles represent 2% of all meningoceles.12 There are several theories regarding the embryologic origin of these three entities, with one theory implicating limited dorsal myeloschisis as the underlying abnormality in both meningoceles and myelocystoceles10 (▶ Fig. 23.4). In both cases, limited dorsal myeloschisis results in an abnormal attachment of the neuroectoderm to the cutaneous ectoderm. If the central canal does not dilate, it regresses, leaving behind a band of tissue that extends to the dorsal aspect of the meningocele cavity. This band of tissue may contain glial, ependymal, or neural tissue, and its course may be either directly dorsal or lateral to one side of the meningocele sac. If the central canal remains dilated, an associated hydromyelic cavity is created that results in a myelocystocele, which distends and displaces the surrounding subarachnoid meningocele.
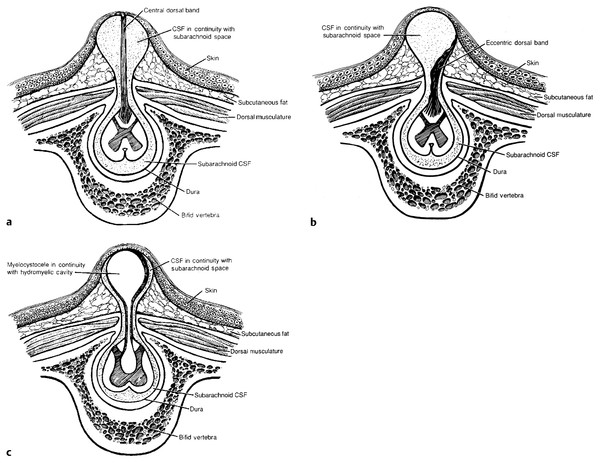
Fig. 23.4 (a) Cervical meningocele with a fibroglial band extending from the dorsal surface of the spinal cord to the dome of the meningocele. (b) Cervical meningocele with a fibroglial stalk eccentrically located and extending from the dorsal surface of the spinal cord to the lateral aspect of the meningocele sac. (c) Cervical meningocele with a hydromyelic central canal herniating from the dorsal surface of the spinal cord to produce a myelocystocele. CSF, cerebrospinal fluid.
(Reproduced with permission from Steinbok P, Cochrane D. Cervical meningoceles and myelocystoceles: a unifying hypothesis. Pediatr Neurosurg 1995;23:320–321)
A more recent classification has been proposed, and as in previous work published on cervical meningoceles, it emphasizes the presence of an intradural stalk in stratifying cervical meningoceles.13 This classification separates lesions with a fibrovascular or neuroglial stalk from those without a stalk. Lesions without a stalk are classified as true meningoceles. Therefore, depending on the definition and classification system used, the term cervical meningocele may encompass several entities—a true meningocele sac devoid of any neural tissue or a meningocele sac containing a stalk.
Cervical meningoceles usually present as a noticeable midline dorsal cervical mass (▶ Fig. 23.5) and (▶ Fig. 23.6). These lesions are usually skin-covered and rarely leak CSF.14 In contrast to lumbosacral meningoceles, cervical meningoceles may be associated with additional anomalies, such as hydrocephalus, Chiari 1 malformation, hydromyelia, lipomyelomeningocele, split-cord malformation, and Klippel-Feil syndrome.8,15,16 This association may reflect the relative heterogeneity of the term cervical meningocele because studies that separate meningoceles with hydromyelia (myelocystoceles) from meningoceles without hydromyelia suggest that true meningoceles are rarely associated with additional anomalies, such as hydrocephalus and Chiari 2 malformation.14,17 In addition, there are fewer associated anomalies with a true meningocele, according to the alternative classification of Salomão et al.13 Nevertheless, because of the association with additional abnormalities of the neuraxis, MR imaging of the brain and entire spine is indicated.8,15,18
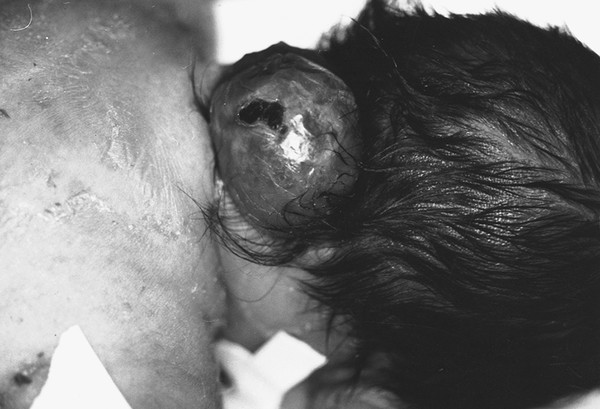
Fig. 23.5 Neonate with a cervical meningocele that has a wide base and is covered almost entirely by dysplastic skin. An eschar is noted at its dome (dark area).
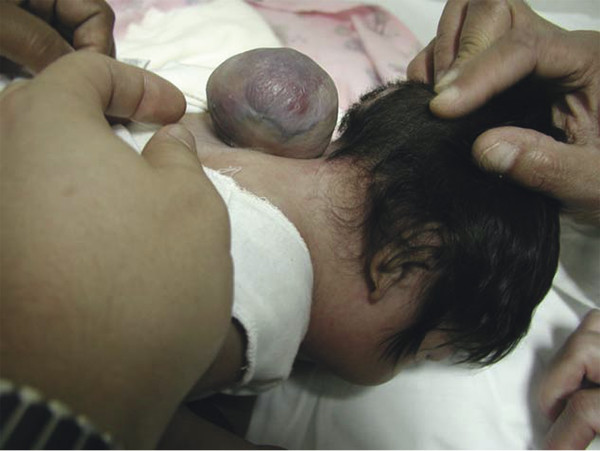
Fig. 23.6 Posterior cervical meningocele in an infant.
It is necessary to repair cervical meningoceles at the time of presentation because tethering by fibrous tissue extending to the dorsal aspect of the meningocele sac may lead to progressive neurologic symptoms over time.19 Operative repair involves opening the sac and performing intradural exploration for any points of tethering by fibrous bands. Goals of surgery include untethering, resection of the meningocele, cosmesis, creation of a watertight dural closure, and prevention of infection.19 Long-term follow-up was assessed in one study of four patients with cervical meningoceles.11 Among the three patients with an isolated cervical meningocele, there were no long-term neurologic or urologic abnormalities. Mild orthopedic abnormalities were noted in three patients, none of which required bracing or surgery.
23.3.3 Thoracic Meningoceles
Posterior thoracic meningoceles are rare and less common than lumbar or sacral meningoceles.22 In a series of 22 patients presenting with spinal meningoceles, 18% of them were in the thoracic region and 77% were in the lumbosacral region.9 Patients may initially present because of a skin-covered dorsal mass or present after birth with radicular pain. There are usually no neurologic deficits. Surgical treatment is similar to that for dorsal meningoceles that occur in other regions of the spine, with the primary goal of evaluating for intradural tethering bands23 and resecting the meningocele sac.
23.4 Anterior and Lateral Meningoceles
23.4.1 Cervical, Thoracic, and Lumbar Meningoceles
Meningoceles that occur at the anterior or lateral aspect of the spinal column result from a different mechanism than those that occur posteriorly. Anterior and lateral meningoceles are rare and are thought to result from mesenchymal disruptions, as often occurs in NF-1 or Marfan syndrome.24,25 These lesions have also been associated with lateral meningocele syndrome and Currarino triad (see later section on anterior sacral meningoceles). Lateral meningoceles that are not associated with either Marfan syndrome or NF-1 may be part of lateral meningocele syndrome,24 which has autosomal-dominant inheritance and is associated with craniofacial abnormalities and connective tissue dysplasia.26 The majority of anterior and lateral meningoceles associated with Marfan syndrome occur in the thoracic or lumbar spine, whereas those associated with NF-1 most often occur in the thoracic spine.24 In the setting of Marfan syndrome, the development of dural ectasia and meningoceles is thought to occur from progressive dilation of the dura secondary to CSF pulsations distending abnormal dura.27 In NF-1, meningoceles may occur secondary to trauma, dural ectasia, cystic degeneration of a neurofibroma, trauma, or congenital herniation of dura.24,28Their location in the thoracic spine may be secondary to a relatively high pressure differential between the CSF and the thoracic cavity that is due to weak paravertebral muscles.32 Anterior and lateral meningoceles are rare in the cervical spine.24
Unlike posterior meningoceles, anterior and lateral lesions do not have associated skin abnormalities and may present throughout childhood and into adulthood. Growth over time may occur through the intervertebral foramen or anteriorly after erosion of the surrounding vertebral body.33 Anterior growth may also cause respiratory failure by compression of the lung or trachea. Presentation may occur at any age and may be associated with radicular pain, weakness, respiratory compromise, or neck pain.24 These lesions are often asymptomatic and may be incidental findings on imaging performed for other reasons. MR imaging should always be performed to delineate the lesion and to evaluate for associated compression or other spinal anomalies, such as associated arachnoid cysts, tethering bands, and lipoma. In the case of NF-1, MR imaging may identify a neuroma associated with the meningocele.25 X-ray or computed tomography (CT) may demonstrate widened intervertebral foramina and erosion or scalloping of the vertebral bodies. There is usually no dysraphic laminar defect, as occurs with posterior lesions. On CT, the meningocele will appear hypodense, similar to CSF, and CT myelography will show a communication between the CSF spaces and the meningocele.
Treatment is indicated if a lesion is symptomatic (e.g., neurologic deficit or respiratory compromise). Asymptomatic lesions may be treated conservatively. Anterior or posterior approaches may be used, depending on the location, size, and presence of an associated neuroma.24,25 For most laterally directed meningoceles, a posterior approach with intradural repair is preferred. For larger lesions, an extradural lateral extracavitary approach is appropriate. For anterior thoracic lesions, an anterolateral transthoracic approach is recommended to access the lesion. Video-assisted thoracoscopy may also be used. A transthoracic–transdiaphragmatic approach or transpleural–retroperitoneal approach may be used for lesions at the thoracolumbar junction. Other treatments may include a cystopleural shunt.33
23.4.2 Anterior Sacral Meningoceles
Anterior sacral meningoceles are rare and occur as a CSF-filled dural sac in the presacral space that connects with the spinal canal through an anterior sacral bony defect.34 Most are congenital, and a female predominance has been noted.35,36 They are thought to arise from the agenesis of anterior sacral elements.37 Although most often sporadic, anterior sacral meningoceles may display an autosomal-dominant inheritance pattern with variable penetrance.38,39 They may also be associated with Currarino syndrome—the triad of anorectal stenosis, sacral bony defect, and presacral mass.40 The presacral mass is often a meningocele, teratoma, or epidermoid or dermoid cyst. A recent review of the Currarino triad noted five patients with tethered cord or anterior myelomeningocele,41 and it is possible that some anterior sacral lesions thought to be meningoceles actually contain nerve roots or neural structures.
Anterior sacral meningoceles may present earlier than anterior and lateral meningoceles in the thoracic spine. A presentation with constipation may be due to the direct compression of pelvic structures or secondary to tethering of nerve roots or the spinal cord. Other presenting signs include headaches resulting from intracranial hypotension secondary to CSF accumulation in the meningocele.24 Urinary dysfunction and back or radicular pain are also possible. Female patients may present during pregnancy with dystocia, and in the past, this was a cause of significant mortality due to rupture of the meningocele during delivery.36 Prenatal diagnosis is rare.42 There is no cutaneous mass or abnormality; however, a mass may be palpated during an abdominal, a rectal, or a vaginal examination. In any newborn presenting with constipation, a thorough rectal examination should be performed. X-rays may show the classic scimitar sign of the pelvis; this results from a lack of pelvic development on one side, which leaves a contralateral area of bone in the shape of scimitar (▶ Fig. 23.7). Despite the large bony defect, the neck of the meningocele is often much smaller.34,35 MR imaging will differentiate other presacral masses, such as teratomas and epidermoid or dermoid cysts, and will also show a tethered cord if present (▶ Fig. 23.8). On CT, the meningocele will appear hypodense, similar to CSF. CT myelography will show a communication between the CSF spaces and the meningocele (▶ Fig. 23.9). As anterior sacral meningoceles may be inherited, some recommend screening immediate family members with a lumbosacral X-ray.38
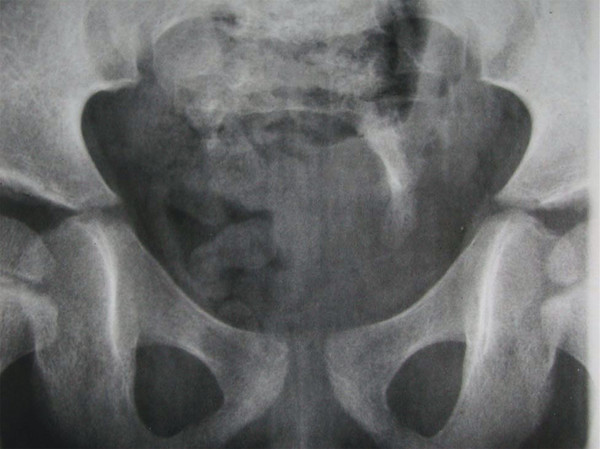
Fig. 23.7 Characteristic scimitar deformity of the sacrum seen in anterior sacral meningoceles.









