Fig. 1
Representative images of mouse brain following systemic injection of AAV9 CB GFP. GFP immunofluorescence shows extensive GFP expression in neurons throughout multiple structures including the cortex (A), hippocampus (B) and cerebellum following injection into neonate mice. Sections from the cortex (D) and medulla (E) show that brains of animals that received intravenous AAV9 injections as adults have GFP expression primarily in astrocytes. Scale bars A-C = 50μm; D-E = 100μm
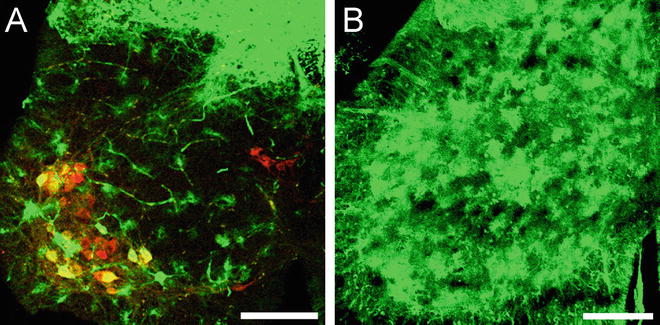
Fig. 2
Representative images of mouse lumbar spinal cord following systemic injection of AAV9 CB GFP. GFP immunofluorescence from lumbar spinal cord sections following intravenous injections into neonate (A) or adult (B) mice show differences in viral transgene expression. A) Injection into neonate mice results in abundant motor neuron (ChAT, red) and dorsal root ganglia cell transduction. GFP expression is also observed in endothelial cells and astrocytes. B) Intravenous injection in adult mice results in robust GFP expression within spinal grey matter astrocytes. GFP expression in motor neurons and dorsal root ganglia is reduced. All scale bars = 200μm
Our experience is with the systemic delivery of adeno-associated virus es (AAV ) in mice and non-human primates, though others have shown that similar crossing of the blood–brain barrier exists in rats and cats as well [7, 10, 21, 22]. Systemic delivery for targeting the CNS has also been reported in neonate mice using lenti-, retro-, and adenovirus vectors [23–26]. Currently AAV9 is the most widely used, but newer AAV serotypes are emerging that show similar properties [11]. Further characterization remains to determine the niche each new serotype can fill. Clinically, systemic AAV gene therapy for treatment of CNS disease has not yet been tested in humans, but the US Food and Drug Administration recently granted investigational new drug approval for IV delivery of AAV9 in 0- to 9-month-old children with the lower motor neuron disease, spinal muscular atrophy. A phase 1 clinical trial is slated to begin in the second half of 2014. Here, we describe the techniques we use to systemically deliver adeno-associated viruses to neonate and adult mice.
2 Materials
2.1 Neonate Injections
1.
Container of wet ice.
2.
Mouse pups on their first or second postnatal day.
3.
Thinpro Insulin Syringe (Terumo, Sommerset, NJ, USA), 3/10 cc, 3/8″ needle, 30 g, 1 per mouse.
4.
Cotton Tipped Applicators.
5.
Fiber Optic Light Source.
6.
Dissecting Microscope.
7.
For training, 1 % Evans Blue Dye solution, made with 1× Phosphate Buffered Saline.
8.
For experiments, viral solution.
2.2 Adult Injections
1.
Mouse tail vein injection device (Braintree Scientific, Braintree, MA, USA).
2.
Mice, >21 days of age.
3.
Thinpro Insulin Syringe (Terumo, Sommerset, NJ, USA), 3/10 cc, 3/8″ needle, 30 g, 1 per mouse.
4.
For training, 1× Phosphate Buffered Saline.
5.
For experiments, viral solution.
6.
Alcohol wipes to clean the tail.
3 Methods
3.1 Neonate Injections
1.
Gather wet ice to anesthetize the mouse pups, an empty cage to segregate the dam from the litter, a dissecting microscope, a light source that can be positioned at an angle to the injection (use of a light source at a 90° angle to the injection site obscures the vein), a clean surface to place the animal for the injection, cotton swabs, 3/10 cc insulin syringe with 3/8″ 30 g needle (one per animal) and 1 % Evans Blue Dye (made with phosphate buffered saline (PBS)) solution for training (see Note 1 ).
3.
Place a single pup directly on the wet ice for 30–60 s to anesthetize the animal.
4.
While the animal is on ice, load your syringe with 30 μl of Evans Blue Dye.
5.
When the animal is fully anesthetized, confirmed by lack of movement on the ice while still breathing, move it under the microscope. For a right-handed injection, face the animal’s muzzle to the right. Place your left index finger on the muzzle and your left middle finger caudal to the ear bud. The ear bud should be between the index and middle fingers (see Note 3 ).
6.
Examine just anterior to the ear bud for a superficial capillary that moves when the skin is manipulated. This capillary is NOT the target, however identification is important for temporal vein identification. Next, locate a dark, shadowy vein inferior to the capillary that remains fixed regardless of skin position. The temporal vein appears shadowy, runs dorsal to ventral, and feeds into the jugular vein.
7.
Enter the temporal vein with the needle bevel up. The needle bevel will fill with blood through the skin. Then depress the plunger slowly and note blanching of the vein down the side of the face.
8.
Allow the needle to remain within the vein for an added 10–15 s to prevent backflow in the injectant.
9.
After a proper injection, the pup should turn blue almost immediately. Remove the needle and apply a cotton swab to the injection site until the blood clots.
10.
Monitor the pup for signs of distress. Allow the pup 2–3 min to recover and re-warm, recognized when the pup is conscious, upright and moving, before returning to the cage. Pups are often cupped in the investigator’s gloved hands to provide appropriate warmth to aid in recovery.
11.
Place the pup back into the home cage and ensure the pup is coated with bedding and/or nestlet to ensure reacceptance by the dam.
12.
A new syringe and cotton swab should be used for each pup to maintain sterility.
13.
Return the mother to the cage with the pups.
3.2 Adult Tail Vein Injections
1.
Load the syringe with the viral solution.
2.
Secure the animal into the restrainer.
3.
Clean the tail with the alcohol wipe.
4.
Warm the tail with warm water (if not using the Braintree restrainer) or the trough in the mouse tail illuminator.
5.
Target the vein starting closer to the tip of the tail. Enter the vein. If you miss the vein, move closer to the base of the tail and try again.
7.
Return the animal to its housing.
4 Notes
1.




Evans blue dye is only used in training. Viral solutions are not mixed with dye. Injection volume in neonate mice is restricted to 50 μl. For neonate injections of self-complementary AAV viruses, viral doses range from 1 to 5 × 1011 vg for efficient CNS targeting.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree







