7
What Can the EEG Tell Us?
Introduction
The electroencephalogram (EEG) measures the difference in voltage between pairs of electrodes placed in an array across the scalp (and, in some cases, directly upon or within the brain). During the long history of EEG – the first animal recording was performed by Canton in 1874 and the first human recording by Berger in 1924 – its role has evolved along with clinical and technical changes in neurology.
Electroencephalography has two general purposes. The first is the evaluation and treatment of epileptic seizures. The second is the assessment of encephalopathy. In these roles, the sensitivity and specificity of EEG diverges.
Electroencephalography carries a high specificity in epilepsy; the cardinal findings in the epileptic patient are difficult to mistake for others and are seldom seen in conditions outside of epilepsy. However, the sensitivity of EEG (technically, its positive predictive value) – its ability to confirm epilepsy in those who have it – is limited. Therefore, many techniques and technologies have arisen in EEG to help elicit epileptic abnormalities to help clinicians sort through the differential diagnosis of epilepsy.
Conversely, in encephalopathy, the EEG has high sensitivity and low specificity. The EEG is exquisitely sensitive in both detecting and reflecting the severity of encephalopathy but has little ability to distinguish among the myriad causes of encephalopathy.
The purpose of this chapter is to focus on the current clinical utility of the EEG applied to epilepsy (and its mimics) and encephalopathy.
 SCIENCE REVISITED
SCIENCE REVISITED- The primary source of the EEG lies in voltage fluctuations triggered by receptor-gated changes in the postsynaptic membrane of neurons. In other words, EEG voltage is the summation of excitatory and inhibitory postsynaptic potentials.
- The neurons most likely to be “seen” by the scalp EEG are large, radially oriented neurons in cortex close to the scalp.
- A large group of neurons is necessary to general potentials visible at the scalp; one estimate is that 10 cm2 of cortex must act simultaneously to generate findings important in epilepsy.
- Activities of groups of cortical neurons are organized by interactions with relay neurons of the thalamus; thalamocortical interactions create characteristic patterns denoting vigilance states (wakefulness, light/deep sleep).
 SCIENCE REVISITED
SCIENCE REVISITED- Each pair of electrodes on the scalp forms a channel; each channel represents the difference in voltage between the pair.
- The arrangement of channels on the EEG page is the montage. The two basic montage types are bipolar montages, in which adjacent electrodes are joined in longitudinal or transverse chains (analogous to sagittal or coronal slices of a brain MRI), and referential montages, in which each electrode on the scalp is paired with a common reference electrode(s), yielding what is analogous to an axial view of the brain.
- Ohm’s law dictates that in a simple electrical circuit, voltage is the product of electrical current and resistance. Impedance (the term used when resistance is encountered in a fluctuating current generated from a biological source) must be close among electrode pairs so that the same voltage recorded from the scalp is not artificially enhanced or diminished by virtue of a poor connection – high impedance – to the scalp (a practice called impedance matching). Good electrode care enables accurate EEG recording.
Epilepsy
Findings in EEG important in epilepsy can be divided into those that occur in between epileptic seizures – interictal discharges – and those that happen as part of an epileptic seizure – ictal discharges.
The interictal epileptiform discharge
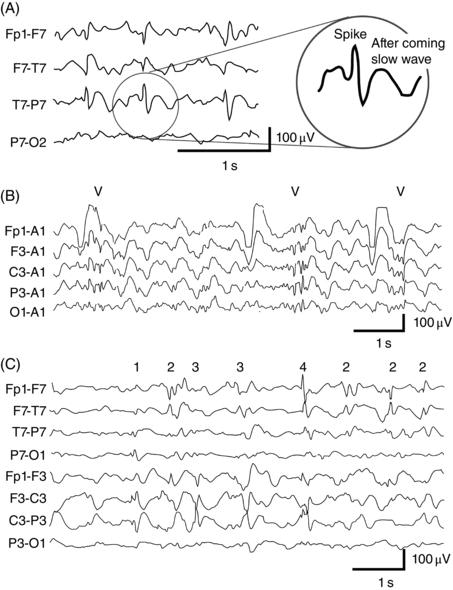
The interictal epileptiform discharge (IED), otherwise known as the spike or sharp wave, is the critical finding in EEG in the evaluation of possible epilepsy. The colloquial definition of an IED is also the most memorable: an IED is any waveform that would hurt if sat upon. A more official definition is that an IED is a waveform that has a duration of 20–200 ms, has a “field” (meaning its distribution across different EEG channels indicates a cerebral rather than artifact or non-brain origin), interrupts rather than forms ongoing activities, and frequently features an aftercoming slow wave with the same polarity (Figure 7.1A). IEDs can occur singly (Figure 7.1A), in a burst of polyspike–wave discharges (Figure 7.1B), or in more than one location (independent multifocal spikes, Figure 7.1C).
 CAUTION!
CAUTION!Figure 7.1. Examples of IEDs. (A) A train of spikes in the left temporal region. (B) Bursts (at marks) of generalized polyspike–wave discharges. (C) Independent multifocal spikes over the left (1) posterior temporal region, (2) anterior temporal region, (3) central region, and (4) frontopolar region.
The ictal discharge
The ictal discharge, in its broadest definition, is any paroxysmal burst of activity that (1) interrupts ongoing EEG activity, (2) evolves (gradually changing in morphology, spatial distribution, or frequency), and (3) involves activity that is often but not obligately sharp. Focal seizures arise from one specific region (Figure 7.3A), and generalized seizures from the entire head (Figure 7.3B). When the ictal discharge accompanies clinical behaviors such as staring, behavioral interruption, confusion, or falling and shaking, then the EEG and behavior are together diagnostic of an epileptic seizure.
Specificity and sensitivity in epilepsy
Unlike the capture of an epileptic seizure that confers diagnostic surety, IEDs provide diagnostic inference only on the evaluation of a patient with spells. Evaluating the specificity and sensitivity of the scalp EEG in the diagnosis of epilepsy is difficult because findings depend on the sample, the population from which it was selected, and technical details of the recordings.
Nevertheless, the probability of finding an IED during a single routine EEG in patients with known epilepsy is approximately 50%. Repeating routine studies increases sensitivity but with gradually diminishing returns, eventually attaining a ceiling of 80–85% after the fourth recording. At this point, even more prolonged recordings, such as overnight video–EEGs, may not be of further utility. This means that about one-fifth of patients with epilepsy will lack interictal evidence of epilepsy. The variable sensitivity of IEDs in patients indicates that a lack of IEDs should not be taken as definitive evidence against seizures.
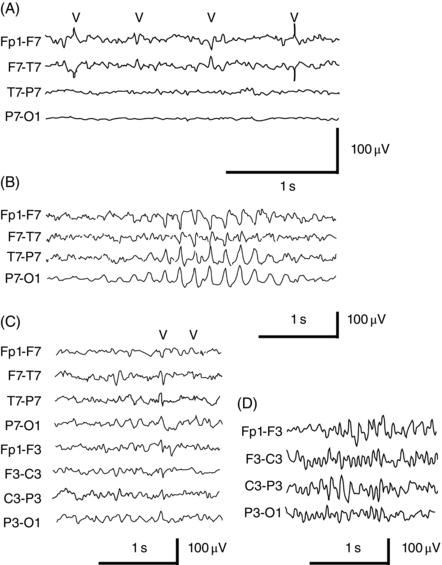
Figure 7.2. Examples of artifacts or other sharply contoured discharges that may mimic epileptiform activity. (A) Electrical artifacts from a poorly adherent F7 electrode (marks). (B) A burst of rhythmic, midtemporal sharp discharges occurring during drowsiness (RMTD), one of the more frequent benign sharp findings seen during drowsiness or light sleep. (C) BETS or small sharp spikes (marks). (D) Six- and fourteen-hertz spikes. Note that most benign epileptiform discharges usually occur during drowsiness or light sleep, and one of the tests is to observe whether the putative abnormalities persist into wakefulness or deeper sleep.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


