Chapter 42 Cervical Interbody Strut Techniques
Since the 1980s, extensive ventral decompression via corpectomy for cervical spondylotic myelopathy and spinal deformity has become routine.1 Although neurologic outcomes remain similar between multilevel anterior discectomy and corpectomy,2–5 certain clinical scenarios favor corpectomy (e.g., ossification of the posterior longitudinal ligament, trauma, osteomyelitis, neoplasms). Moreover, fusion rates after anterior decompression procedures across more than two disc levels may be higher for corpectomy than discectomy, particularly in uninstrumented cases.4,6,7 Therefore, spine surgeons must be comfortable with anterior decompression by corpectomy and also with the subsequent intervertebral strut grafting, the focus of this chapter.
Technologic advances now permit a wide variety of materials to be used as interbody devices; these newer products are covered elsewhere in this textbook (see Chapters 41 and 43). Furthermore, in the majority of clinical situations today, anterior corpectomy strut grafting is supplemented with anterior spinal plate instrumentation to reduce graft migration and enhance fusion rates.4,6–8 However, certain scenarios, for either clinical or logistical reasons, may dictate uninstrumented strut grafting. The techniques of interlocking bone grafting discussed in this chapter are most germane to the latter category of corpectomy cases. Even in instrumented strut grafts, however, some of the principles delineated here remain important for successful integration of the bone graft.
Fundamentals of Grafting
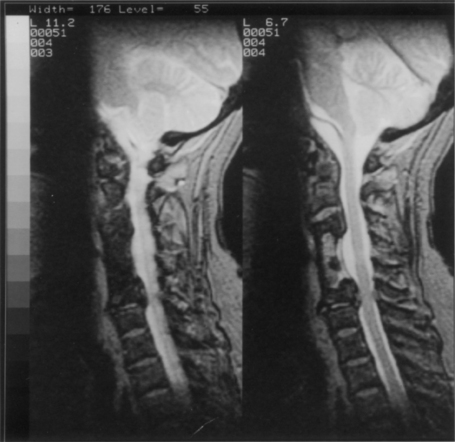
Three fundamental concepts need to be recognized for successful strut grafting. First is a clear understanding of the surgical objectives of the procedure in general. The primary goal for cervical spondylotic myelopathy typically is adequate and durable decompression of the neural elements. Although this generally would seem obvious, concerns over reconstruction can alter the operative plan and possibly subvert the primary goals of the surgery (Fig. 42-1). Ideally, the reconstruction must be fit to the decompression, and not vice versa.
The second essential component of strut grafting is an understanding of the factors affecting spinal stability (Box 42-1).9 An uninstrumented, unstable spine requires prolonged external bracing (e.g., halo brace or Minerva jacket). This is relatively independent of the surgical fusion technique. The stable spine reconstructed with a short-segment strut graft may be managed with a rigid cervical orthosis.
BOX 42-1 Factors Influencing Stability
From White AA, Panjabi MM: Clinical biomechanics of the spine, ed 2, Philadelphia, 1990, Lippincott-Raven, p 314.
Bone Graft
Both the origin of the graft material and its proper handling are important considerations in bone graft selection. Autogenous iliac crest tends to fuse rapidly, which is a distinct advantage. Its incorporation, however, can be compromised by suboptimal harvesting techniques (see Chapter 123), osteoporosis, and injudicious tailoring. Technical constraints typically limit its use to replacing two or three vertebral segments. In fashioning iliac crest to the bony defect, it is ideal to preserve at least two contiguous cortical surfaces from one end of the graft to the other to optimize axial loading strength. Surgeons must also keep in mind the real complications associated with iliac crest harvest, which fortunately only rarely result in long-term problems.
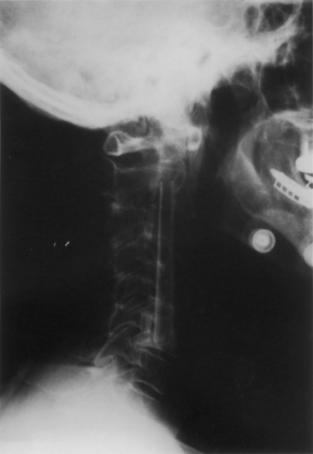
With a fibular implant, however, there are different characteristics to consider: (1) it is a strong, circumferential cortical strut with a higher modulus of elasticity than mixed cortical–cancellous implants, and as such must be used with caution in the osteoporotic spine; (2) it can be tailored to any needed length; and (3) it provides a central channel for the packing of autograft cancellous bone to enhance fusion. The disadvantage of fibula is the mismatch of the density with that of the vertebral body. As a general rule, the receiving vertebra will fail before the fibula graft does. This generally results in “pistoning,” in which the fibula penetrates through the vertebral body and can even enter the next motion segment. Some subsidence may be unavoidable, especially in osteoporosis, but is usually of no significant clinical consequence (Fig. 42-2). Subsidence may theoretically be limited by using minimal distraction during graft placement and by using an orthotic brace postoperatively to limit flexion. Too much graft loading and excessive neck flexion early in recovery predispose to graft pistoning. Minimal disruption of the vertebral body graft bed site is also important in maintaining the final height of the fusion. A fibula grafted to a partial corpectomy will almost invariably result in substantial subsidence and loss of height. If necessary, an additional vertebral level may need to be resected to preserve the resistance to subsidence at the graft site.
Allograft fibula is slower to incorporate than autologous iliac crest.8 Autograft fibula is less commonly used owing to the increased operative times and blood loss and significant complications associated with its harvest.10 One method to enhance fusion but attenuate graft harvest morbidity is to use allograft fibula packed with autograft cancellous bone, taken from the iliac crest or from the resected corpectomy bone itself.11 Autogenous cancellous bone may be accessed via the superficial surface of the iliac crest through a 3-cm skin incision. The medial and outer surfaces of the iliac crest are not disturbed, as would be needed for the harvest of tricortical grafts. This ideally reduces blood loss and postoperative pain. A 1-cm cortical defect is created in the iliac crest with a high-speed bur, and cancellous bone is taken with a large curette. This, in turn, is packed into the central canal of the allograft fibula with a 3-mm diameter rod. No bone need be placed around the outside of the fibula strut after insertion.
Despite the differences between iliac crest structural autograft and fibular allograft, a significant difference in pseudarthrosis rates has not been consistently demonstrated.2,5,12,13
Strut Graft
Preparation of Vertebral Defect for Strut Grafting
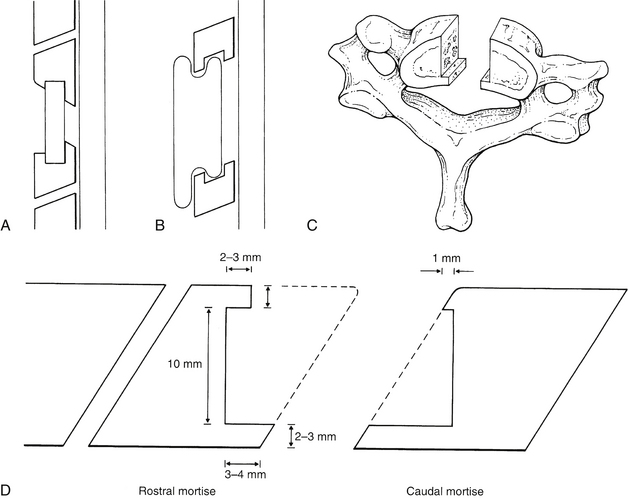
The paramount concern in preparing the vertebral end plates for arthrodesis is the prevention of graft displacement. Although plates and screws prevent graft displacement and improve graft incorporation, even instrumented grafts in rare cases can retropulse toward the spinal cord. The bed for the graft must be prepared in such a manner that the avenue toward the spinal canal is shorter or narrower than the graft itself. If graft migration were to occur, the direction should be away from the spinal cord. When anterior plating is used, deep slots or mortises in the vertebral body are limited by the need for adequate remaining vertebral body volume for screw purchase. When hardware insertion is not anticipated, spinal canal protection may be attained by one of four strategies (Figs. 42-3A–C): (1) the keystone mortise and tenon, (2) the dovetail technique, (3) the lateral step method, or (4) the anterior peg method of Niu et al.11 The primary focus here is on the keystone method.
Keystone Technique
The keystone graft method places the graft close to the middle column of the vertebral body. It is secured by means of mortises or slots in the opposing vertebral end plates (see Fig. 42-3A).
Proper preparation of the mortises in the keystone technique requires consideration of the angling of the cervical disc space (Fig. 42-3D). This disc space angling is the consequence of the ventral vertebral surface being slightly more caudal than the dorsal vertebral surface. The caudal mortise can be fashioned into the face of the vertebral end plate without removal of the anterior cortical corner of the vertebra. Thus, the sloping of this end plate away from the spinal canal provides the opportunity for creating the ideal mortise. The dorsal mortise lip is longer than its ventral counterpart. This ensures that any potential displacement of the graft occurs across the shallower ventral mortise lip. Because the caudal vertebral mortise can be readily fashioned with preservation of the cortical vertebral margins, this is the strongest mortise construct (see Fig. 42-3D).
Creation of the rostral mortise is more complex. Again, the critical consideration is the disc space angle. At the caudal end plate of the rostral mortise, the angle is such that to ensure a shorter ventral mortise lip, a portion of the anterior vertebral body must be resected. To avoid undue anterior resection while ensuring adequacy of the posterior mortise lip, appreciable resection of the dorsal vertebral margin in the decompression is precluded. Should any dorsal vertebral body decompression be pursued, the remaining vertebral body may be inadequate for proper mortising (see Fig. 42-3D).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree