8
Disorders of Equilibrium
APPROACH TO DIAGNOSIS
Equilibrium is the ability to maintain orientation of the body and its parts in relation to external space. It depends on continuous visual, labyrinthine, and proprioceptive somatosensory input and its integration in the brainstem and cerebellum. Disorders of equilibrium result from diseases that affect central or peripheral vestibular pathways, the cerebellum, or sensory pathways involved in proprioception. Such disorders usually present with one of two clinical problems: vertigo or ataxia.
VERTIGO
Vertigo is the illusion of movement of the body or the environment. It may be associated with other symptoms, such as impulsion (a sensation that the body is being hurled or pulled in space), oscillopsia (a visual illusion of moving back and forth), nausea, vomiting, or gait ataxia.
Vertigo must be distinguished from nonvertiginous dizziness, which includes sensations of light-headedness, faintness, or giddiness not associated with an illusion of movement. In contrast to vertigo, these sensations are produced by conditions that deprive the brain of blood, oxygen, or glucose (eg, excessive vagal stimulation, orthostatic hypotension, cardiac arrhythmia, myocardial ischemia, hypoxia, or hypoglycemia) and may culminate in loss of consciousness (syncope; Chapter 12).
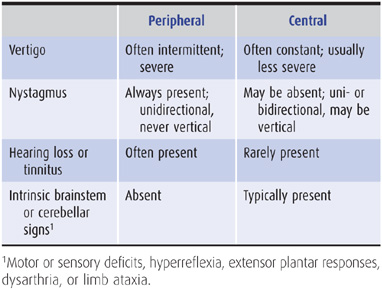
The first step in the differential diagnosis of vertigo is to localize the pathologic process to the peripheral or central vestibular pathways (Figure 8-1). Certain characteristics of vertigo, including the presence of any associated abnormalities, can help differentiate between peripheral and central causes (Table 8-1).
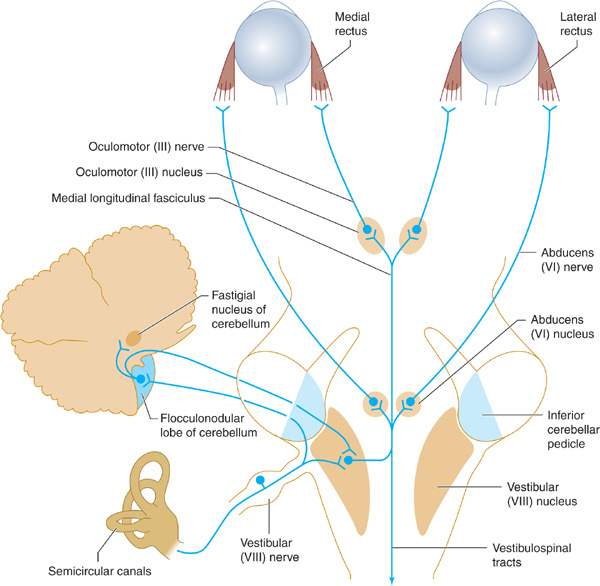
Figure 8-1. Peripheral and central vestibular pathways. The vestibular nerve terminates in the vestibular nucleus in the brainstem and midline cerebellar structures that project to the vestibular nucleus. From here, bilateral pathways in the medial longitudinal fasciculus ascend to the abducens (VI) and oculomotor (III) nuclei and descend to the spinal cord (vestibulospinal tracts).
Table 8-1 Characteristics of peripheral and central vertigo.
PERIPHERAL VERTIGO
Peripheral vestibular lesions affect the labyrinth of the inner ear or the vestibular division of the vestibulocochlear (VIII) nerve. Vertigo from peripheral lesions tends to be intermittent, lasts for briefer periods, and produces more distress than vertigo of central origin (see next section). Nystagmus (rhythmic oscillation of the eyeballs) is always present with peripheral vertigo; it is usually unidirectional and never vertical. Peripheral lesions commonly produce additional symptoms of inner ear or vestibulocochlear (VIII) nerve dysfunction, such as hearing loss and tinnitus (the illusion of hearing a nonexistent sound, such as ringing in the ears).
CENTRAL VERTIGO
Vertigo of central nervous system origin usually results from lesions that affect the brainstem vestibular nuclei or their connections; rarely, vertigo can be produced by a cerebral cortical lesion, such as when it occurs as a symptom of complex partial seizures (Chapter 12).
Central vertigo may occur with or without nystagmus; if nystagmus is present, it can be vertical, unidirectional, or multidirectional and may differ in character in the two eyes. (Vertical nystagmus is oscillation in a vertical plane; nystagmus produced by upgaze or downgaze is not necessarily in the vertical plane.) Vertigo from central lesions may be accompanied by intrinsic brainstem or cerebellar signs, such as motor or sensory deficits, hyperreflexia, extensor plantar responses, dysarthria, or limb ataxia.
ATAXIA
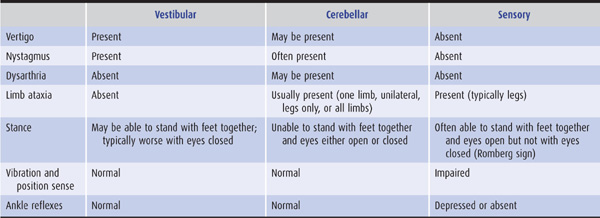
Ataxia is incoordination or clumsiness of movement that is not the result of muscular weakness. It can be caused by vestibular, cerebellar, or sensory (proprioceptive) disorders. Ataxia can affect eye movement, speech (producing dysarthria), individual limbs, the trunk, stance, or gait (Table 8-2).
Table 8-2 Characteristics of vestibular, cerebellar, and sensory ataxia.
VESTIBULAR ATAXIA
The same central and peripheral lesions that cause peripheral or central vertigo (see preceding section) can also produce vestibular ataxia. Nystagmus is frequently present and is typically unilateral and most pronounced on gaze away from the side of vestibular involvement. Dysarthria is not a component of vestibular ataxia. Vestibular ataxia is gravity-dependent: Incoordination of the limbs becomes apparent only when the patient attempts to stand or walk.
CEREBELLAR ATAXIA
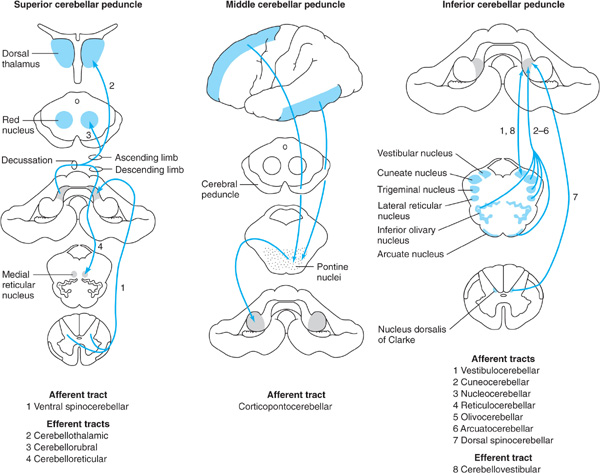
Cerebellar ataxia is produced by lesions of the cerebellum or its afferent or efferent connections in the cerebellar peduncles, red nucleus, pons, or spinal cord (Figure 8-2). Because of the crossed connection between the frontal cerebral cortex and the cerebellum, unilateral frontal disease can also occasionally mimic a disorder of the contralateral cerebellar hemisphere. The clinical manifestations of cerebellar ataxia consist of irregularities in the rate, rhythm, amplitude, and force of voluntary movements.
Figure 8-2. Cerebellar connections in the superior, middle, and inferior cerebellar peduncles. The peduncles are indicated by gray shading and the areas to and from which they project by blue shading.
Hypotonia
Cerebellar ataxia is commonly associated with hypotonia, which results in defective posture maintenance. Limbs are easily displaced by a relatively small force and, when shaken by the examiner, exhibit an increased range of excursion. The range of arm swing during walking may be similarly increased. Tendon reflexes take on a pendular quality, so that several oscillations of the limb may occur after the reflex is elicited, although neither the force nor the rate of the reflex is increased. When muscles are contracted against resistance that is then removed, the antagonist muscle fails to check the movement and compensatory muscular relaxation does not ensue promptly. This results in rebound movement of the limb.
Incoordination
In addition to hypotonia, cerebellar ataxia is associated with incoordination of voluntary movements. Simple movements are delayed in onset, and their rates of acceleration and deceleration are decreased. The rate, rhythm, amplitude, and force of movements fluctuate, producing a jerky appearance. Because these irregularities are most pronounced during initiation and termination of movement, their most obvious clinical manifestations include terminal dysmetria, or “overshoot” when the limb is directed at a target, and terminal intention tremor as the limb approaches the target. More complex movements tend to become decomposed into a succession of individual movements rather than a single smooth motor act (asynergia). Movements that involve rapid changes in direction or greater physiologic complexity, such as walking, are most severely affected.
Eye Movement Abnormalities
Because of the cerebellum’s prominent role in the control of eye movements, ocular abnormalities are a frequent consequence of cerebellar disease. These include nystagmus and related ocular oscillations, gaze paresis, and defective saccadic and pursuit movements.
Anatomic Basis of Clinical Signs
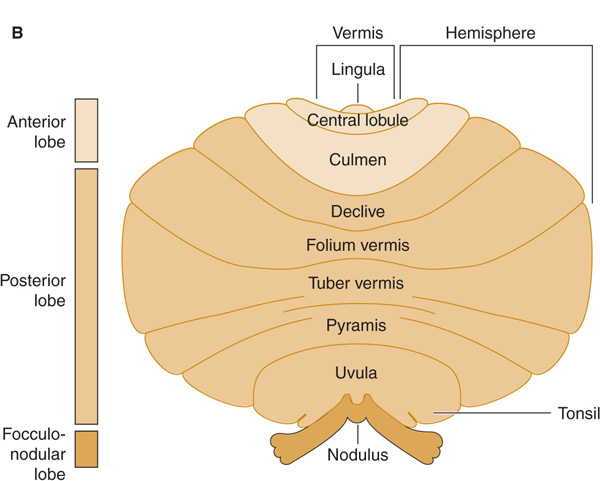
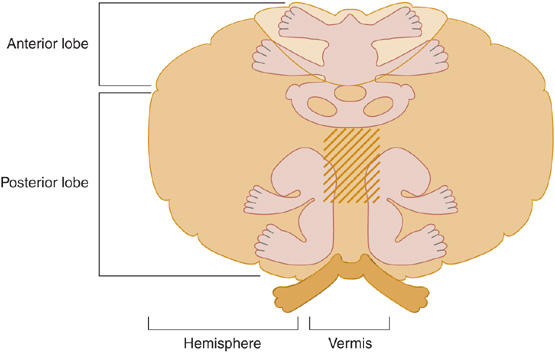
Various anatomic regions of the cerebellum (Figure 8-3) are functionally distinct, corresponding to the somatotopic organization of their motor, sensory, visual, and auditory connections (Figure 8-4).
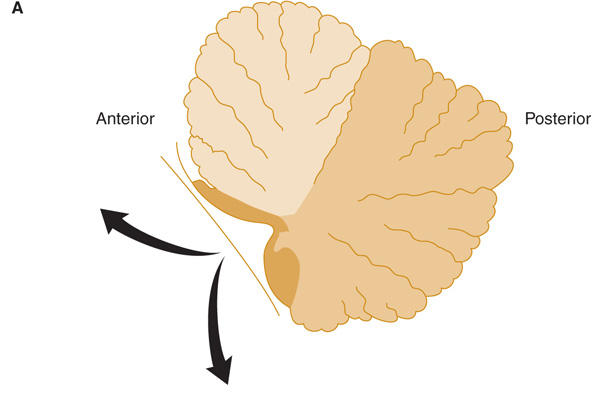
Figure 8-3. Anatomic divisions of the cerebellum in midsagittal view (A), or unfolded (arrows) and viewed from behind (B).
Figure 8-4. Functional organization of the cerebellum. The view is similar to that in Figure 8-3B but is of a monkey rather than a human cerebellum. The three cerebellar homunculi represent areas to which proprioceptive and tactile stimuli project, and the stripes represent areas to which auditory and visual stimuli project.
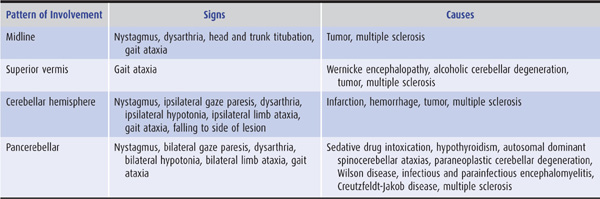
1. Midline lesions—The middle zone of the cerebellum—the vermis and flocculonodular lobe and their associated subcortical (fastigial) nuclei—is involved in the control of axial functions, including eye movements, head and trunk posture, stance, and gait. Midline cerebellar disease therefore results in a clinical syndrome characterized by nystagmus and other disorders of ocular motility, dysarthria, oscillation of the head and trunk (titubation), instability of stance, and gait ataxia (Table 8-3). Selective involvement of the superior cerebellar vermis, as commonly occurs in alcoholic cerebellar degeneration, produces exclusively or primarily ataxia of gait, as would be predicted from the somatotopic map of the cerebellum (Figure 8-4).
Table 8-3 Clinical patterns of cerebellar ataxia.
3. Diffuse disease—Many cerebellar disorders—typically toxic, metabolic, and degenerative conditions—affect the cerebellum diffusely. The clinical picture in such states combines the features of midline and bilateral hemisphere disease.
SENSORY ATAXIA
Sensory ataxia results from disorders that affect the prop-rioceptive pathways in peripheral sensory nerves, sensory roots, posterior columns of the spinal cord, or medial lemnisci. Thalamic and parietal lobe lesions are rare causes of contralateral sensory hemiataxia.
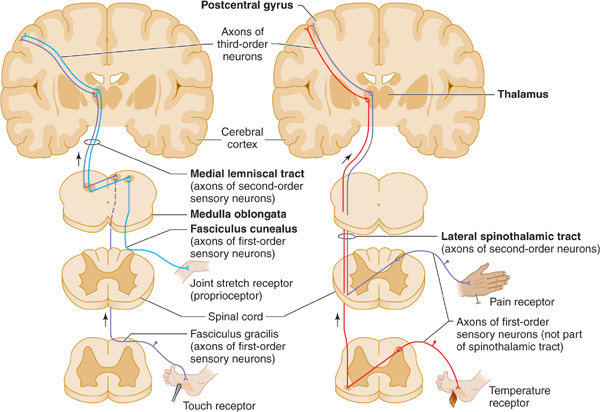
Sensations of joint position and movement (kinesthesis) originate in pacinian corpuscles and unencapsulated nerve endings in joint capsules, ligaments, muscle, and periosteum. Such sensations are transmitted via heavily myelinated A fibers of primary afferent neurons, which enter the dorsal horn of the spinal cord and ascend uncrossed in the posterior columns (Figure 8-5). Proprioceptive information from the legs is conveyed in the medially located fasciculus gracilis, and information from the arms is conveyed in the more laterally situated fasciculus cuneatus. These tracts synapse on second-order sensory neurons in the nucleus gracilis and nucleus cuneatus in the lower medulla. The second-order neurons decussate as internal arcuate fibers and ascend in the contralateral medial lemniscus. They terminate in the ventral posterior nucleus of the thalamus, from which third-order sensory neurons project to the parietal cortex.
Figure 8-5. Pathways mediating proprioception (left, light blue) and other somatic sensory modalities (left and right). (Reprinted with permission from Fox SI. Human Physiology. 10th ed. Boston, MA: McGraw-Hill; 2008.)
Sensory ataxia from polyneuropathy or posterior column lesions typically affects the gait and legs in symmetric fashion; the arms are involved to a lesser extent or are spared entirely. Examination reveals impaired sensation of joint position in the affected limbs, and vibration sense is also commonly disturbed. Vertigo, nystagmus, and dysarthria are characteristically absent.
HISTORY
SYMPTOMS & SIGNS
Vertigo
True vertigo must be distinguished from a light-headed or presyncopal sensation. Vertigo is typically described as spinning, rotating, or moving, but when the description is vague, the patient should be asked specifically if the symptom is associated with a sense of movement.
The circumstances under which symptoms occur may also be diagnostically helpful. Vertigo is often brought on by changes in head position. The occurrence of symptoms upon arising after prolonged recumbency is a common feature of orthostatic hypotension, which may be immediately relieved by sitting or lying down. Orthostatic hypotension and other cerebral hypoperfusion states can lead to loss of consciousness, which is rarely associated with true vertigo.
Symptoms associated with vertigo may help to localize the site of the causal lesion. Hearing loss or tinnitus strongly suggests a disorder of the peripheral vestibular apparatus (labyrinth or vestibulocochlear [VIII] nerve). Dysarthria, dysphagia, diplopia, or focal weakness or sensory loss affecting the face or limbs points to a likely central (brainstem) lesion.
Ataxia
Ataxia associated with vertigo suggests a vestibular disorder, whereas ataxia with numbness or tingling in the legs is common in patients with sensory ataxia. Because proprioceptive deficits may, to some extent, be compensated for by other sensory cues, patients with sensory ataxia may report that their balance is improved by watching their feet when they walk or by using a cane or the arm of a companion for support. They thus find that they are much more unsteady in the dark and may experience particular difficulty in descending stairs.
ONSET & TIME COURSE
The mode of onset and time course may help to identify the cause of a disorder of equilibrium.
Sudden
Sudden onset of disequilibrium occurs with infarcts and hemorrhages in the brainstem or cerebellum (eg, lateral medullary syndrome, cerebellar hemorrhage, or infarction).
Episodic
Episodic disequilibrium of acute onset suggests transient ischemic attacks in the basilar artery distribution, benign positional vertigo, or Ménière disease. Disequilibrium from transient ischemic attacks is usually accompanied by cranial nerve deficits, neurologic signs in the limbs, or both. Ménière disease is usually associated with progressive hearing loss and tinnitus as well as vertigo.
Chronic & Progressive
Chronic, progressive disequilibrium evolving over weeks to months is most suggestive of a toxic or nutritional disorder (eg, vitamin B12 or vitamin E deficiency, nitrous oxide exposure). Evolution over months to years is characteristic of an inherited spinocerebellar degeneration.
MEDICAL HISTORY
The medical history should be scrutinized for evidence of diseases that affect the sensory pathways (vitamin B12 deficiency, syphilis) or cerebellum (hypothyroidism, paraneoplastic syndromes, tumors) and drugs that produce disequilibrium by impairing vestibular or cerebellar function (ethanol, sedative drugs, phenytoin, aminoglycoside antibiotics, quinine, salicylates).
FAMILY HISTORY
A hereditary degenerative disorder may be the cause of chronic, progressive cerebellar ataxia. Such disorders include spinocerebellar degenerations, Friedreich ataxia, ataxia-telangiectasia, and Wilson disease.
GENERAL PHYSICAL EXAMINATION
Various features of the general physical examination may provide clues to the underlying disorder.
1. Orthostatic hypotension is associated with certain sensory disorders that produce ataxia (eg, tabes dorsalis, polyneuropathies) and with some cases of spinocerebellar degeneration.
2. Skin may show oculocutaneous telangiectasia (ataxia-telangiectasia), or it may be dry, with brittle hair (hypothyroidism) or have a lemon-yellow coloration (vitamin B12 deficiency).
3. Pigmented corneal (Kayser-Fleischer) rings are seen in Wilson disease (Chapter 11).
4. Skeletal abnormalities may be present. Kyphoscoliosis is typical in Friedreich ataxia, hypertrophic or hyperextensible joints are common in tabes dorsalis, and pes cavus is a feature of certain hereditary neuropathies. Abnormalities at the craniocervical junctions may be associated with Arnold-Chiari malformations or other congenital anomalies that involve the posterior fossa.
NEUROLOGIC EXAMINATION
MENTAL STATUS EXAMINATION
Acute confusional state with ataxia characterizes ethanol or sedative drug intoxication and Wernicke encephalopathy. Dementia with cerebellar ataxia is seen in Wilson disease, Creutzfeldt-Jakob disease, hypothyroidism, paraneoplastic syndromes, and some spinocerebellar degenerations. Dementia with sensory ataxia suggests syphilitic taboparesis or vitamin B12 deficiency. Amnesia and cerebellar ataxia are associated with chronic alcoholism (Korsakoff amnestic syndrome, Chapter 5).
STANCE & GAIT
Observation of stance and gait is helpful in distinguishing among vestibular, cerebellar, and sensory ataxia. In any ataxic patient, the stance and gait are wide-based and unsteady, often associated with reeling or lurching movements.
Stance
The ataxic patient asked to stand with the feet together may show great reluctance or an inability to do so. With persistent urging, the patient may gradually move the feet closer together but will leave some space between them.
1. Patients with sensory ataxia and some with vestibular ataxia are, nevertheless, ultimately able to stand with the feet together, compensating for the loss of one source of sensory input (proprioceptive or labyrinthine) with another (visual). This compensation is demonstrated when the patient closes the eyes, eliminating visual cues. With sensory or vestibular disorders, unsteadiness increases and may result in falling (Romberg sign). With a vestibular lesion, the tendency is to fall toward the side of the lesion.
2. Patients with cerebellar ataxia are unable to compensate for their deficit by using visual input and are unstable on their feet whether the eyes are open or closed.
Gait
1. The gait in cerebellar ataxia is wide-based, often with a staggering quality that might suggest drunkenness. Oscillation of the head or trunk (titubation) may be present. If a unilateral cerebellar hemisphere lesion is responsible, there is a tendency to deviate toward the side of the lesion when the patient attempts to walk in a straight line or circle or marches in place with eyes closed. Tandem (heel-to-toe) gait, which requires walking with an exaggerated narrow base, is always impaired.
2. In sensory ataxia, the gait is also wide-based and tandem gait is poor. In addition, walking is typically characterized by lifting the feet high off the ground and slapping them down heavily (steppage gait) because of impaired proprioception. Stability may be dramatically improved by letting the patient use a cane or lightly rest a hand on the examiner’s arm for support. If the patient is made to walk in the dark or with eyes closed, gait is much more impaired.
3. Gait ataxia may also be a manifestation of conversion disorder (conversion disorder with motor symptom or deficit) or malingering. Determining this can be particularly difficult, as isolated gait ataxia without ataxia of individual limbs can also be produced by diseases that affect the superior cerebellar vermis. The most helpful observation in identifying factitious gait ataxia is that such patients often exhibit wildly reeling or lurching movements from which they are able to recover without falling. In fact, recovery of balance from such awkward positions requires excellent equilibrium.
CRANIAL NERVES
Abnormalities of extraocular (III, IV, and VI) and vestibulocochlear (VIII) nerve function are typically present with vestibular disease and often present with lesions of the cerebellum.
Ocular Alignment
The eyes are examined in the primary position of gaze (looking directly forward) to detect misalignment in the horizontal or vertical plane.
Eye Movements
1. The patient is asked to turn the eyes in each of the cardinal directions of gaze (left, up and left, down and left, right, up and right, down and right; Chapter 1) to determine whether ocular nerve palsy or gaze paresis (impaired ability to move the two eyes coordinately in any of the cardinal directions of gaze) is present.
2. Nystagmus—an abnormal involuntary oscillation of the eyes—is characterized in terms of the positions of gaze in which it occurs (gaze-evoked nystagmus), its amplitude, and the direction of its fast phase. Pendular nystagmus, usually the result of visual impairment that begins in infancy, has the same velocity in both directions of eye movement; jerk nystagmus is characterized by both fast (vestibular-induced) and slow (cortical) phases. The direction of jerk nystagmus is defined by the direction of the fast component.
3. Fast voluntary eye movements (saccades) are elicited by having the patient rapidly shift gaze from one target to another placed in a different part of the visual field. Slow voluntary eye movements (pursuits) are assessed by having the patient track a slowly moving target such as the examiner’s finger.
4. Peripheral vestibular disorders produce unidirectional horizontal jerk nystagmus that is maximal on gaze away from the involved side.
5. Central vestibular disorders can cause unidirectional or bidirectional horizontal nystagmus, vertical nystagmus, or gaze paresis.
6. Cerebellar lesions are associated with a wide range of ocular abnormalities, including gaze pareses, defective saccades or pursuits, nystagmus in any or all directions, and ocular dysmetria (overshoot of visual targets during saccadic eye movements).
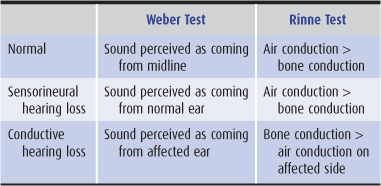
Hearing
Preliminary examination of the vestibulocochlear (VIII) nerve should include otoscopic inspection of the auditory canals and tympanic membranes, assessment of auditory acuity in each ear, and Weber and Rinne tests (Table 8-4) performed with a 256-Hz tuning fork.
Table 8-4 Assessment of hearing loss.
1. In the Weber test, unilateral sensorineural hearing loss (from lesions of the cochlea or vestibulocochlear nerve) causes the patient to perceive the sound produced by a vibrating tuning fork placed at the vertex of the skull as coming from the normal ear. With a conductive (external or middle ear) disorder, sound is localized to the abnormal ear.
2. The Rinne test may also distinguish between sensorineural and conductive defects in the affected ear. Air conduction (tested by holding the vibrating tuning fork next to the external auditory canal) normally produces a louder sound than does bone conduction (tested by placing the base of the tuning fork over the mastoid bone). This pattern also occurs with sensorineural hearing loss due to vestibulocochlear nerve lesions but is reversed in the case of conductive hearing loss.
Positional Tests
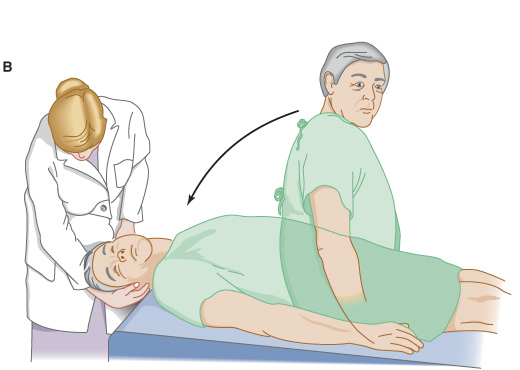
When patients indicate that vertigo occurs with a change in position, the Nylen-Bárány or Dix-Hallpike maneuver (Figure 8-6) is used to try to reproduce the precipitating circumstance. The head is rapidly lowered 30 degrees below horizontal. This process is repeated with the head and eyes turned first to the right and then to the left. The eyes are observed for nystagmus, and the patient is asked to note the onset, severity, and cessation of vertigo.
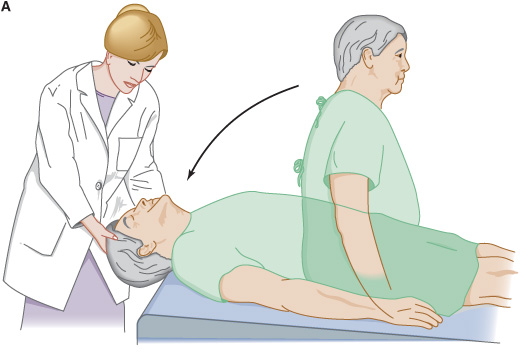
Figure 8-6. Test for positional vertigo and nystagmus. The patient is seated on a table with the head and eyes directed forward (A) and is then quickly lowered to a supine position with the head over the table edge, 45 degrees below horizontal. The patient’s eyes are then observed for nystagmus, and the patient is asked to report any vertigo. The test is repeated with the patient’s head and eyes turned 45 degrees to the right (B), and again with the head and eyes turned 45 degrees to the left (not shown).
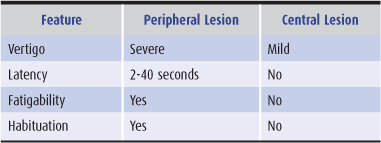
Positional nystagmus and vertigo are usually associated with peripheral vestibular lesions and are most often a feature of benign positional vertigo. This is typically characterized by severe distress, a latency of several seconds between assumption of the position and the onset of vertigo and nystagmus, a tendency for the response to remit spontaneously (fatigue) as the position is maintained, and attenuation of the response (habituation) as the offending position is repeatedly assumed (Table 8-5). Positional vertigo can also occur with central vestibular disease.
Table 8-5 Characteristics of positional nystagmus.
Caloric Testing
Disorders of the vestibuloocular pathways can be detected by caloric testing. Caloric testing should be preceded by careful otoscopic examination and should not be undertaken if the tympanic membrane is perforated. The patient is placed supine with the head elevated 30 degrees to bring the superficially situated lateral semicircular canal into the upright position. Each ear canal is irrigated in turn with cold (33°C) or warm (44°C) water for 40 seconds, with at least 5 minutes between tests. Warm water tends to produce less discomfort than cold.
1. In the normal, awake patient, cold-water caloric stimulation produces nystagmus with the slow phase toward and the fast phase away from the irrigated ear. Warm water irrigation produces the opposite response.
2. In patients with unilateral labyrinthine, vestibulocochlear (VIII) nerve, or vestibular nuclear dysfunction, irrigation of the affected side fails to cause nystagmus or elicits nystagmus that is later in onset or briefer in duration than on the normal side.
Other Cranial Nerves
Papilledema associated with disequilibrium suggests an intracranial mass lesion, usually in the posterior fossa, that is causing increased intracranial pressure. Optic neuropathy may be present in multiple sclerosis, neurosyphilis, or vitamin B12 deficiency. A depressed corneal reflex or facial palsy ipsilateral to the lesion (and the ataxia) can accompany cerebellopontine angle tumor. Weakness of the tongue or palate, hoarseness, or dysphagia results from lower brainstem disease.
MOTOR SYSTEM
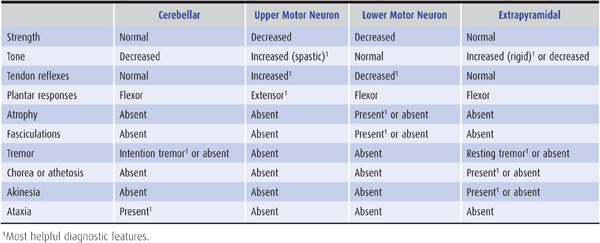
Examination of motor function in the patient with a disorder of equilibrium should disclose the pattern and severity of ataxia and any associated pyramidal, extrapyramidal, or lower motor neuron involvement that might suggest a cause. The clinical features that help distinguish cerebellar disease from disease involving these other motor systems are summarized in Table 8-6.
Table 8-6 Clinical features distinguishing cerebellar from other motor systems disorders.
Muscle Tone
1. Hypotonia is characteristic of cerebellar disorders; with unilateral cerebellar hemispheric lesions, the ipsilateral limbs are hypotonic.
2. Extrapyramidal hypertonia (rigidity) may occur in disorders that affect both the cerebellum and basal ganglia (eg, Wilson disease, acquired hepatocerebral degeneration, Creutzfeldt-Jakob disease, and some autosomal dominant spinocerebellar ataxias).
3. Ataxia with spasticity may be seen in conditions that affect both the cerebellum and upper motor neuron pathways (eg, multiple sclerosis, posterior fossa tumors, congenital anomalies, vertebrobasilar ischemia or infarction, some autosomal dominant spinocerebellar ataxias, Friedreich ataxia, neurosyphilis, Creutzfeldt-Jakob disease, and vitamin B12 deficiency).
Coordination
1. Truncal stability is assessed by examination of gait (discussed earlier) and by observing the patient as he or she sits unsupported. In addition to gait ataxia, patients with midline or pancerebellar disease may show oscillation of the head or trunk (titubation) and truncal ataxia while seated; in the latter case, they tend to fall when they try to remain sitting upright.
2. Movement of the patient’s arm is observed as his or her finger tracks back and forth between his or her own nose or chin and the examiner’s finger. With mild cerebellar ataxia, an intention tremor characteristically appears near the beginning and end of each such movement, and the patient may overshoot the target.
3. When the patient is asked to raise the arms rapidly to a given height—or when the arms, extended and outstretched in front of the patient, are displaced by a sudden force—there may be overshooting (rebound). Impaired ability to check the force of muscular contractions can also be demonstrated by having the patient forcefully flex the arm at the elbow against resistance—and then suddenly removing the resistance. If the limb is ataxic, continued contraction without resistance may cause the hand to strike the patient, whose head should be turned to the side to avoid being hit in the face.
4. Ataxia of the legs is demonstrated by the supine patient’s inability to run the heel of the foot smoothly up and down the opposite shin.
Weakness
1. Pure vestibular, cerebellar, or sensory disorders do not cause weakness. However, some diseases affect these systems in combination with motor pathways.
2. Distal weakness can be caused by disorders that also produce sensory ataxia, such as polyneuropathies and Friedreich ataxia.
3. Paraparesis
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree