Chapter 24 Formation, Modification, and Repair of Neuronal Connections
Once the matching process is complete, plasticity is reduced but not abolished; ongoing adjustments of connections and synaptic strength underlie processes such as learning and memory. This relative stability of the mature nervous system carries a complementary peril: a limited ability to repair itself after disease or injury. However, experimental methods of enhancing the remaining plasticity, or of reactivating the processes at work during development, are beginning to offer promising approaches to treatment.
Both Neurons and Connections Are Produced in Excess during Development
The central nervous system (CNS) develops from the neural tube as a series of modules, each supplying the neurons and connections needed for particular functions. The result is most apparent in the spinal cord, where individual segments contain the motor neurons, interneurons, and primary afferent connections for specific regions of the body (see Figs. 10-1 and 10-4). Comparable modules come into play in other parts of the CNS, including brainstem segments with particular sets of cranial nerve nuclei and forebrain zones that provide particular sets of neurons.
The final number of neurons, and the number and pattern of connections of these neurons, varies from one module to another. Again, this is most apparent in the spinal cord, where different segments wind up with, for example, different numbers of lower motor neurons or different populations of autonomic motor neurons (see Fig. 10-8). The initially surprising mechanism accounting for these level-to-level differences is that each module produces an excess of neurons, and each of these neurons makes more widespread connections than are necessary. The matching process is one of winnowing the surplus neurons and retracting inappropriate connections.
Neurotrophic Factors Ensure That Adequate Numbers of Neurons Survive
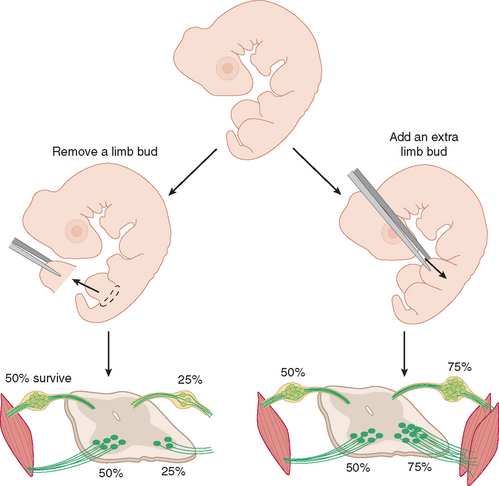
Early experiments with chicks showed that the number of embryonically produced dorsal root ganglion cells, lower motor neurons, and autonomic ganglion cells that survive into adulthood depends on the amount of target tissue with which these neurons interact during development (Fig. 24-1). The basis of this effect is neurotrophic factors* that are produced in limited quantities by target tissues, taken up by peripheral nerve endings, and transported retrogradely to neuronal cell bodies. Neurons compete for these factors during development, and those that acquire sufficient quantities survive. One result of normal development is that thoracic spinal segments, with less tissue to innervate and a smaller supply of trophic factors, lose more motor neurons and other neurons than do cervical segments. Trophic interactions are not limited to neurons with processes in peripheral nerves (Fig. 24-2A), and throughout the nervous system, roughly half of all newborn neurons die.
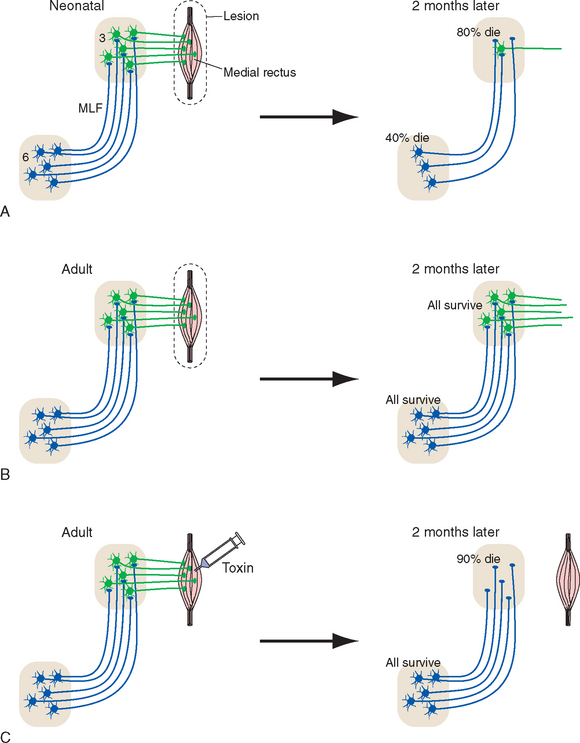
Figure 24-2 The precise and nearly exclusive projection of internuclear neurons in the abducens nucleus (6) to medial rectus motor neurons in the oculomotor nucleus (3; see Fig. 12-9) provides a convenient means to test the dependence of these internuclear neurons on their postsynaptic targets. A, Unilateral removal of the medial rectus (together with the terminals of medial rectus motor neurons) from a neonatal rat results in death of most medial rectus motor neurons. This in turn reduces the supply of neurotrophic factors to internuclear neurons, and many of them die. B, Adult neurons are less dependent on trophic factors for their survival, so the same medial rectus removal from an adult rat does not result in the death of any motor neurons (although they go through a period of metabolic changes in the weeks following the lesion; see Fig. 24-27). Internuclear neurons also survive, but this could be due to survival of motor neurons or to decreased dependence on trophic factors. C, Injection of an adult medial rectus with a cytotoxic lectin (ricin) results in death of almost all medial rectus motor neurons. Internuclear neurons still survive, consistent with decreased dependence on trophic factors. MLF, medial longitudinal fasciculus.
(Based on Morcuende S et al: Exp Neurol 195:244, 2005.)
A host of neurotrophic factors have now been characterized. Nerve growth factor (NGF), the first discovered and most famous, is part of a family of small proteins called neurotrophins. Other members include brain-derived neurotrophic factor (BDNF), neurotrophic factor-3 (NT-3), and neurotrophic factor-4/5 (NT-4/5). The neurotrophins act primarily on neurons, but many other neurotrophic factors are active in both the nervous system and other parts of the body. Different types of peripheral nervous system (PNS) neurons depend on particular neurotrophins or combinations of neurotrophins for their early survival. For example, sympathetic ganglion cells and dorsal root ganglion cells with free nerve endings are especially sensitive to NGF, vestibular ganglion neurons to BDNF, and spiral ganglion neurons to NT-3. Most CNS neurons are less selective and respond to multiple neurotrophic factors, although there are exceptions (e.g., the cholinergic neurons of the basal nucleus [see Fig. 11-28] are especially sensitive to NGF).
Axonal Branches Are Pruned to Match Functional Requirements
Later in development, neurotrophic factors cease to be essential for the survival of many neurons (Fig. 24-2B and C) but continue to have a major role in determining which dendrites and axon branches flourish and which wither and retract. Immature neurons are considerably more promiscuous in both receiving and making synaptic connections than are adult neurons. Some are as seemingly peculiar as neurons in the occipital lobe that project to the spinal cord, or other cortical neurons with one branch that traverses the corpus callosum and another that enters the internal capsule. In a series of activity-dependent processes that mostly begin after the period of cell death and extend well beyond birth, connections that contribute less effectively to function are withdrawn; for the most part, connections at which there is correlated presynaptic and postsynaptic activity are retained, and others are pruned away. (This is not to say that the total number of synapses decreases. The surviving connections become more complex and elaborate, and the total number of synapses increases.)
One well-studied example is the innervation of skeletal muscle fibers by motor neurons (Fig. 24-3). The neuromuscular junction of each skeletal muscle fiber is initially innervated during fetal life by terminal branches of two or more motor neurons. Starting at about midgestation (after the period of naturally occurring motor neuron death is over), some of these terminal branches begin to withdraw, and the remaining branches expand to take over the vacated territory. Near the time of birth, each muscle fiber is left with a singly innervated neuromuscular junction.
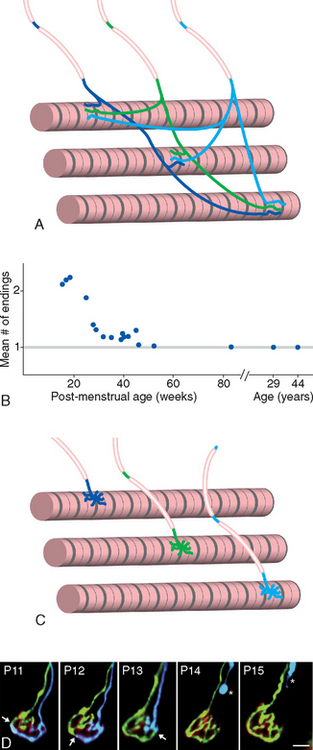
Similar elimination of axonal branches is widespread in the CNS. Two thirds of the axons in the corpus callosum of a newborn monkey, for example, die off during the first few months of life; those that survive acquire myelin sheaths and permanent connections. Something comparable undoubtedly happens in the human corpus callosum, although it probably extends over several years. Likewise, there is an excess of corticospinal axons at birth, including large numbers of uncrossed projections that retract over a period of years (Fig. 24-4); most of these are probably branches of neurons whose functionally most important connections lie elsewhere, as in the temporary occipital corticospinal projections referred to earlier. The process leading to innervation of individual cerebellar Purkinje cells in adults by single climbing fibers (see Figs. 20-13, 20-14, and 20-20) provides another example (Fig. 24-5), this one analogous to the development of singly innervated neuromuscular junctions. Each immature Purkinje cell receives limited synaptic inputs from multiple climbing fibers; all but one retract, and the remaining climbing fiber extends over the proximal dendrites and forms a much more powerful distributed synapse.
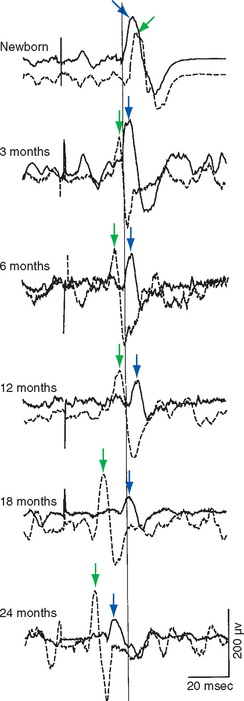
Figure 24-4 Mass electrical activity (electromyogram) of the right (dashed traces, green arrows) and left (solid traces, blue arrows) biceps in response to transcranial magnetic stimulation (see Box 22-4) of left motor cortex. All recordings were made from the same subject at different ages. The response of the biceps ipsilateral to the stimulated cortex slowly becomes smaller over a period of years, while the response of the contralateral biceps becomes larger and faster.
(From Eyre JA et al: Neurology 57:1543, 2001.)
Pruning of Neuronal Connections Occurs during Critical Periods
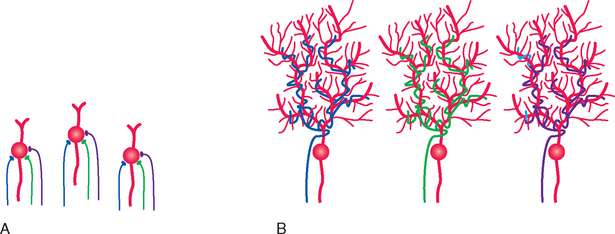
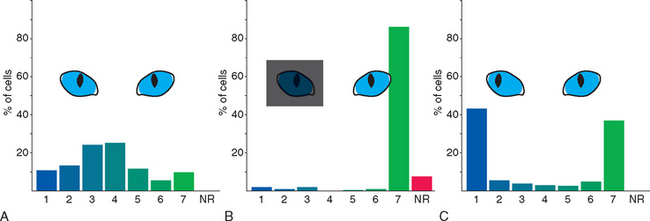
The time available for matching CNS connections to the environment is normally limited to a series of critical periods during which plasticity is maximal; the wiring patterns achieved during these periods are more or less permanent. This has been studied most extensively in primary visual cortex, where changes in the systematic organization of hypercolumns (see Fig. 17-35) can be examined in detail. Lateral geniculate neurons receive input from just one eye (see Fig. 17-27C) and pass it along to neurons in layer IV (see Fig. 17-35C). Outputs from layer IV then converge on neurons in other layers, with the result that most of these neurons respond, at least to some extent, to inputs from both eyes (Fig. 24-6A); this is the beginning of binocular vision and depth perception. Covering one eye of an adult cat for long periods causes no change in these properties of cortical neurons. In striking contrast, however, covering one eye for the first 2 to 3 months of life results in nearly all cells being responsive only to the eye that had not been covered (Fig. 24-6B). This is a permanent effect associated specifically with early deprivation; leaving the previously deprived eye uncovered does not restore its ability to activate cortical neurons. Clear functional deficits accompany these physiological changes. Although the pupillary light reflex is normal, the animal behaves as though blind when using the previously covered eye. Segregated monocular inputs to layer IV are present at birth in humans and monkeys, and these compete with each other for synaptic sites (much like the competition for space at neuromuscular junctions). If one eye is occluded, as by an infantile cataract, inputs from the other eye expand in layer IV to take over its territory; the result is amblyopia (Greek for “dim vision”), in which vision is poor in that eye despite a normal retina.
Competition between the two eyes is also revealed if the two eyes are not normally aligned early in life (Fig. 24-6C), thus disrupting the correlated visual information that ordinarily reaches cortical neurons from the two eyes. The input from one eye or the other then takes over at almost all cortical neurons; binocular vision and depth perception are impaired permanently.
Experience-dependent plasticity during critical periods is also found in the auditory and somatosensory systems. Similarly, prevention of limb use during early life (presumably analogous to covering an eye) leads to abnormal development of corticospinal terminals in the spinal cord (Fig. 24-7). Normal terminals fail to develop later in life, and fine movement control continues to be impaired.
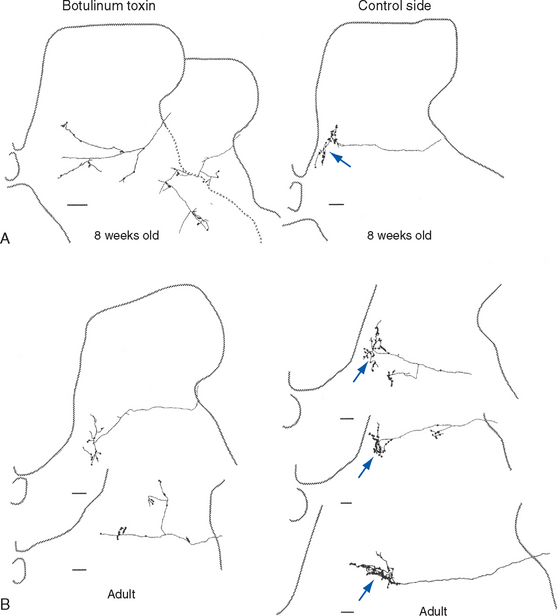
Figure 24-7 Critical period for the development of fine motor control. Use of one forelimb was prevented during weeks 3 to 7 of a kitten’s life—the time when some corticospinal branches are retracted and others become much more elaborate—by injecting botulinum toxin (see Box 8-1) into the elbow flexors and extensors and the wrist flexors. Single corticospinal axons were then examined in the C8 segment. A, At 8 weeks, a week after the botulinum toxin had worn off, corticospinal axons on the previously paralyzed side had failed to develop the elaborate sprays of synaptic endings (arrow) seen on the normal side. B, The immature corticospinal endings persisted into adulthood. Scale bars = 100 μm.
(From Martin JH et al: J Neurosci 24:2122, 2004.)
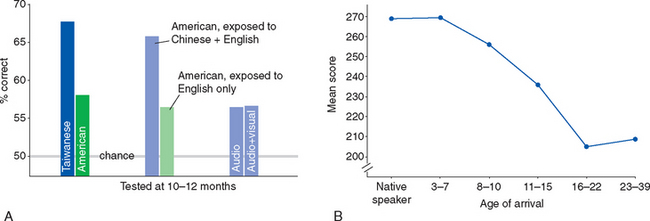
Critical periods extend to the acquisition of more complex skills as well. For example, children learn languages much more easily and successfully than adults do. This is a complicated process that starts early in life and extends into the teenage years. Very young infants are able to discriminate among the speech sounds of all human languages. Starting at about 6 months of age, they get better at detecting different sounds heard from caregivers in their native language and start to lose the ability to discriminate among speech sounds they have not heard, that is, some sounds of nonnative languages (Fig. 24-8A). Much of this process is complete by 1 year of age and presumably corresponds to the refinement of synaptic connections someplace in auditory association cortex. Acquisition of more subtle aspects of language continues for years after this but does not go on indefinitely; learning a new language fluently is considerably more difficult after about age 16 (Fig. 24-8B). Comparable phenomena are found with other complex skills. Six-month-old infants, for example, can distinguish equally well between a familiar face and a novel face, whether it belongs to a human or a monkey. Nine-month-old infants, like adults, are much better at distinguishing between human faces than monkey faces. There are at least hints of critical-period effects on virtually every human cognitive ability.
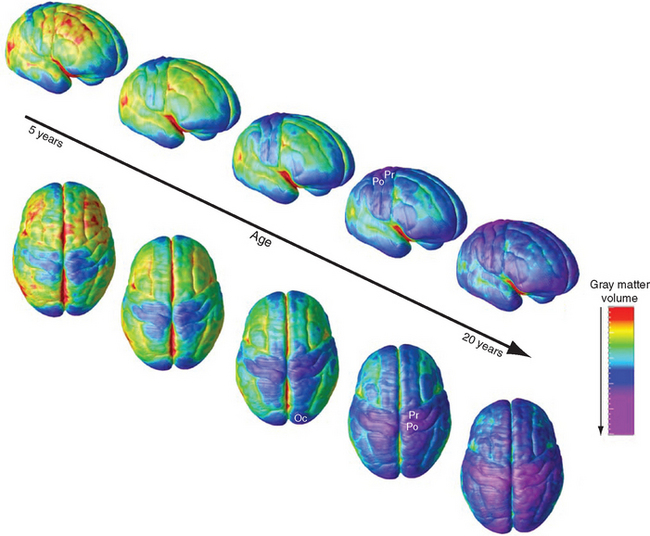
Critical periods draw to a close at different times in different parts of the brain (Fig. 24-9), in part due to the onset of processes that restrict the ability of the adult CNS to respond effectively to damage; these are described later in the chapter. Not surprisingly, the refinement of synapses in multimodal areas, which depend on inputs from unimodal areas, takes longest.
Synaptic Connections Are Adjusted throughout Life
There Are Short-Term and Long-Term Adjustments of Synaptic Strength
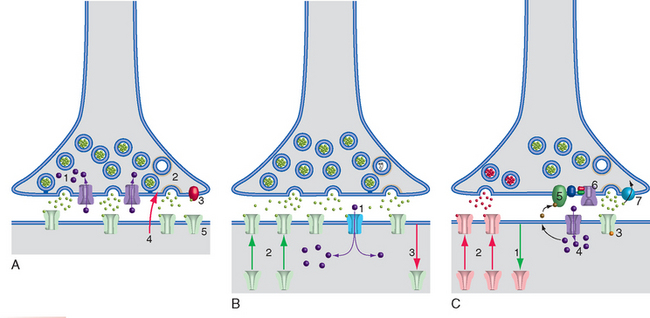
The voltage-gated Ca2+ channels essential for chemical synaptic transmission (see Fig. 8-6) let in Ca2+ faster than it can be sequestered. The result is a brief increase in presynaptic Ca2+ concentrations, of a size dictated by the frequency of invading action potentials. This in turn facilitates transmitter release and causes potentiation of transmission at that synapse—an increase in transmitter release in response to subsequent action potentials that abates within a few minutes. Other changes accompanying routine synaptic transmission can cause complementary short-term depression, also lasting minutes or less, through both presynaptic and postsynaptic mechanisms (Fig. 24-10A). High-frequency stimulation can cause depletion of vesicles and smaller subsequent releases; transmitter can bind to presynaptic autoreceptors, usually decreasing transmission; continued binding of transmitter generally makes postsynaptic receptors less sensitive for a little while (much as sensory receptors adapt to continued stimulation); and retrograde signals such as nitric oxide act briefly at many synapses.
More prolonged effects on synaptic strength, collectively called long-term potentiation (LTP) and long-term depression (LTD), can act through almost any part of a synapse (Fig. 24-10B and C); these are the basis of the lasting changes that occur during critical periods and in learning and memory. The best-known examples of each occur at the postsynaptic membranes of glutamate synapses on pyramidal and many other neurons, where two different kinds of ionotropic receptors usually coexist. AMPA receptors* are typical ligand-gated channels that cause depolarization by allowing nonselective passage of Na+ and K+ ions. NMDA receptors are also ligand gated but have the additional property of being voltage sensitive (see Fig. 8-20); they open only if they bind glutamate and the membrane is already depolarized. In addition, NMDA receptors allow passage of not just Na+ and K+ but also Ca2+. Collectively, these properties make NMDA receptors eminently suited to initiate activity-dependent changes in synaptic strength. In the presence of background depolarization—caused, for example, by high-frequency inputs, correlated activity at nearby synapses, or changes in modulatory inputs during increased attention—glutamate release results in not only further depolarization but also Ca2+ influx. Ca2+ influx in turn sets in motion intracellular signaling pathways that result in the insertion or removal of AMPA receptors at that synapse. More AMPA receptors make the synapse more sensitive to subsequent glutamate release (LTP); fewer receptors make it less sensitive (LTD). Early stages of LTP and LTD, lasting hours, are based on the insertion or removal of AMPA receptors already present in the presynaptic ending. The changes can be stabilized and persist for years through later stages that involve communication with the nucleus and protein synthesis.
Multiple Memory Systems Depend on Adjustments of Synaptic Strength
Although we tend to think of memory as a unitary function and to associate it with remembering facts and events, there are in fact multiple kinds of memory, each depending on different sets of CNS structures (Fig. 24-11). Memory of events and facts—remembering what you had for dinner last night (episodic memory), or remembering that Yankee Stadium is in the Bronx or the meaning of a word (semantic memory)—is called declarative (or explicit) memory, indicating that these items are accessible to consciousness and can be declared as remembered events or facts. Memory that manifests more as subconsciously generated responses to events or stimuli is called nondeclarative (or implicit) memory. Nondeclarative memory is more varied and includes memories of skills and procedures—how to play handball or pinochle—as well as conditioned reflexes and seemingly automatic emotional reactions to certain situations. Just as we normally combine the activities of multiple eye-movement control systems in real-life situations, we combine different kinds of memory in most learning tasks. The knowledge of how to get to school or work, for example, is a mixture of learned habits and procedures and remembered facts.
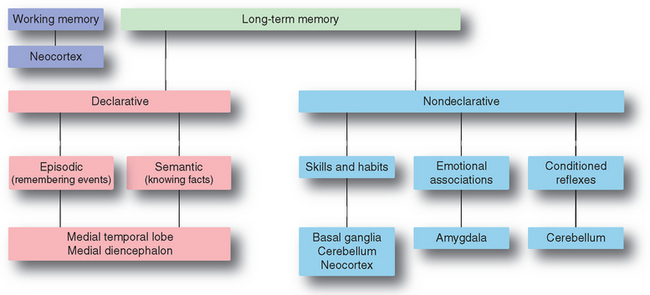
Figure 24-11 Major categories of learning and memory, and the principal parts of the CNS associated with each.
Most forms of memory, especially declarative memory, have multiple steps. Some form of information is first encoded and enters working memory, basically the small amount of information that we can keep “in mind” at one time. Some fraction of the things that enter working memory then goes through a consolidation phase, becoming long-term memories stored in some part of the CNS.
Cortical Maps Are Adjusted throughout Life
The cerebral cortex is the site of long-term storage of several forms of memory, both declarative and nondeclarative. These take the form of networks of neurons, interconnected by strengthened (or sometimes new) synapses. One way in which expanded synaptic connections among cortical neurons are manifest is in the reorganization of cortical maps. These maps were traditionally considered stable in adults, but it is now clear that parts of them can expand or shrink substantially (Fig. 24-12). Extended, skilled use of a body part causes its representation in motor and somatosensory cortex to expand at the expense of neighboring areas; the changes start within hours, so they probably depend on preexisting, previously inactive connections. Conversely, in cases of immobilization or amputation, surrounding areas take over the territory of the affected part. If damage occurs early in life, before plasticity has declined to its adult level, the changes can be extensive (Fig. 24-12D and E). An extreme example is the conversion of visual cortex to other functions in the congenitally blind (Box 24-1; Figs. 24-13 and 24-14).
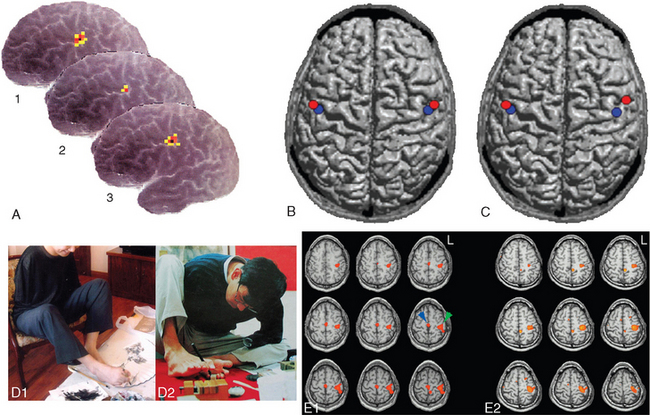
Figure 24-12 Plasticity in cortical maps. A, Short-term plasticity in motor cortex. The subject was a 54-year-old woman who had been blind all her life and worked as a Braille proofreader. The area of left motor cortex devoted to her right first dorsal interosseous muscle was mapped using transcranial magnetic stimulation on a Friday after she had been reading Braille all week (1), at the end of a 10-day vacation during which she read little Braille (2), and at the end of the next week of work (3). Activity-dependent changes in the amount of motor cortex devoted to this muscle used in Braille reading can be seen easily. Consistent with these changes, she reported difficulty reading Braille at normal speeds for a period of time after her vacation. B and C, Use-dependent changes in somatosensory cortex in professional musicians. B, Average source of evoked potentials after electrically stimulating the contralateral median (red) and ulnar (blue) nerves in 35 nonmusicians, providing a measure of the size of the hand representation in somatosensory cortex. C, Average source of evoked potentials after electrically stimulating the contralateral median (red) and ulnar (blue) nerves in 15 right-handed violin players. By this measure, the representation of the left hand in somatosensory cortex is enlarged substantially. D and E, Much more extensive changes in motor cortex in two individuals who lost both upper extremities early in life and subsequently learned to perform very skilled movements with their feet. D1, A 30-year-old man who had bilateral amputations at the shoulder at age 4 following a high-voltage accident went on to become a professional painter and sculptor. D2, A 33-year-old man who had amputations at age 8 above the left elbow and below the right shoulder, also following a high-voltage accident. He too went on to become a sculptor and is shown carving a stone seal. E1 and E2, Functional magnetic resonance imaging of both men as they performed a flexion-extension task with their right toes reveals activation in the paracentral lobule (blue arrow), where the foot is represented (see Fig. 3-30), and also laterally in the precentral gyrus (green arrow) at a level where the hand would ordinarily be represented.
(A, from Pascual-Leone A et al: Ann Neurol 38:910, 1995. B and C, from Schwenkreis P et al: Eur J Neurosci 26:3291, 2007. D and E, from Yu X et al: Magn Reson Imaging 24:45, 2006.)
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree