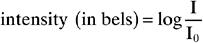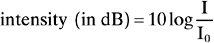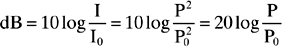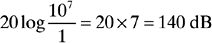Chapter 14 Hearing and Balance: The Eighth Cranial Nerve
Auditory and Vestibular Receptor Cells Are Located in the Walls of the Membranous Labyrinth
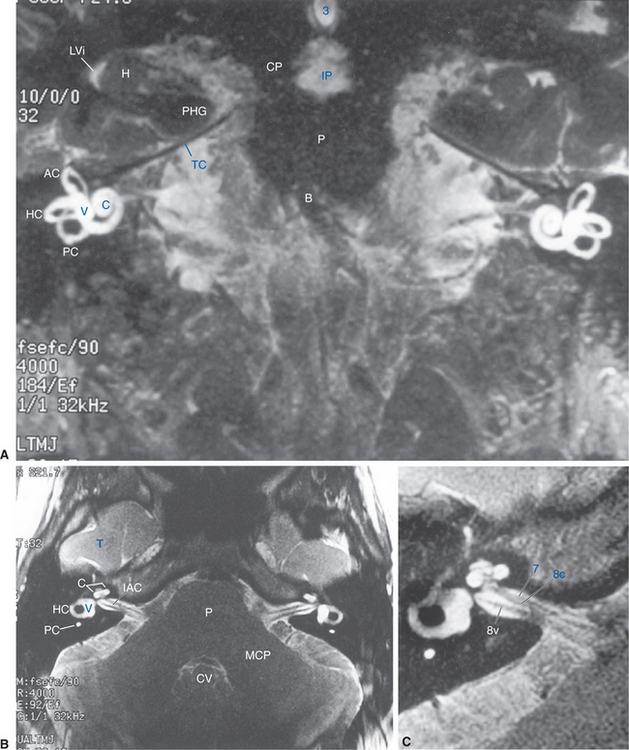
The structures innervated by the eighth nerve are embedded in the temporal bone (Figs. 14-1 and 14-2), where the receptor cells form parts of the walls of a convoluted, membranous tube that is suspended within a bony tube (Fig. 14-3).
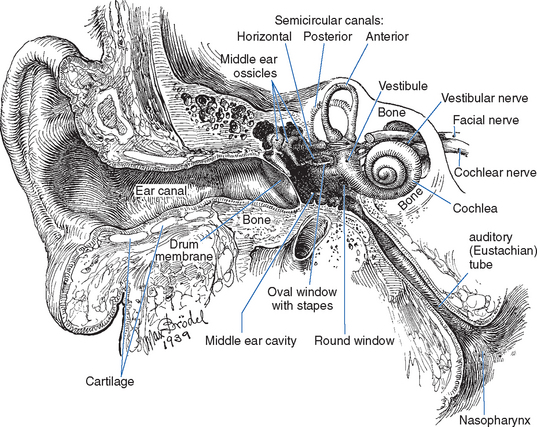
Figure 14-1 The outer, middle, and inner ears, showing the bony labyrinth embedded in the temporal bone.
(From Brödel M: Three unpublished drawings of the anatomy of the human ear, Philadelphia, 1946, WB Saunders.)
The Membranous Labyrinth Is Suspended within the Bony Labyrinth, a Cavity in the Temporal Bone
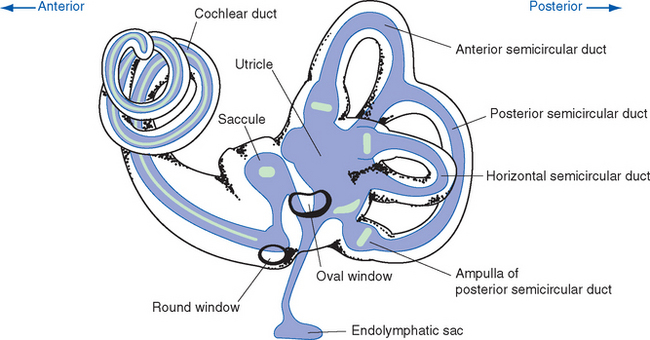
The walls of the bony tube are formed by the hardest bone in the body—the petrous (“like a rock”) portion of the temporal bone. Because the tube consists of so many twists and turns, it is called the bony labyrinth. The coiled cochlea (Latin for “snail shell”) extends anteriorly from an enlargement called the vestibule, to which three semicircular canals are attached. The membranous labyrinth, the membranous tube suspended within the bony labyrinth, generally follows the same contours (Fig. 14-3). Hence there is a cochlear duct within the bony cochlea and a semicircular duct within each semicircular canal. However, the vestibule contains two enlargements of the membranous labyrinth, the utricle (to which the semicircular ducts attach) and the saccule (which is connected to the cochlear duct and to the utricle).
Endolymph Is Actively Secreted, Circulates through the Membranous Labyrinth, and Is Reabsorbed
Endolymph is produced continuously by specialized cells in one wall of the cochlear duct and in several other locations in the membranous labyrinth, using an active pumping mechanism that results in a positive electrical potential inside the membranous labyrinth. Endolymph seems to have a path of circulation and reabsorption reminiscent of that of CSF. The semicircular ducts communicate with the utricle, and the cochlear duct communicates with the saccule. The ducts interconnecting the utricle and saccule meet in a Y-shaped junction through which endolymph can enter the endolymphatic duct, leave the labyrinth, and reach the endolymphatic sac in the dura covering the temporal bone, where some of it is reabsorbed. Just as obstruction of CSF flow causes expansion of the ventricles and neurological symptoms, obstruction of endolymph flow was naturally thought to cause ballooning of the membranous labyrinth and otological symptoms. Méniàre’s disease is characterized by transient attacks of vertigo, nausea, and hearing loss accompanied by ringing in the ears (tinnitus); its defining anatomical feature is swelling of the membranous labyrinth (referred to as endolymphatic hydrops), usually attributed to endolymphatic blockage. Some cases of hydrops are asymptomatic, however, and there are occasional cases of patients with Méniàre’s symptoms but no hydrops. Although there are probably multiple causes of Méniàre’s disease, in many cases the hydrops is likely to be a secondary result of some still-unknown process that disrupts fluid secretion in the membranous labyrinth and damages hair cells.
Auditory and Vestibular Receptors Are Hair Cells
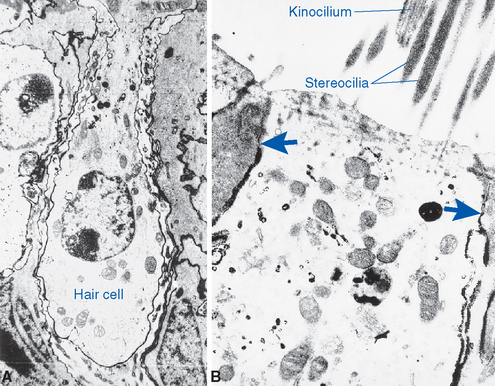
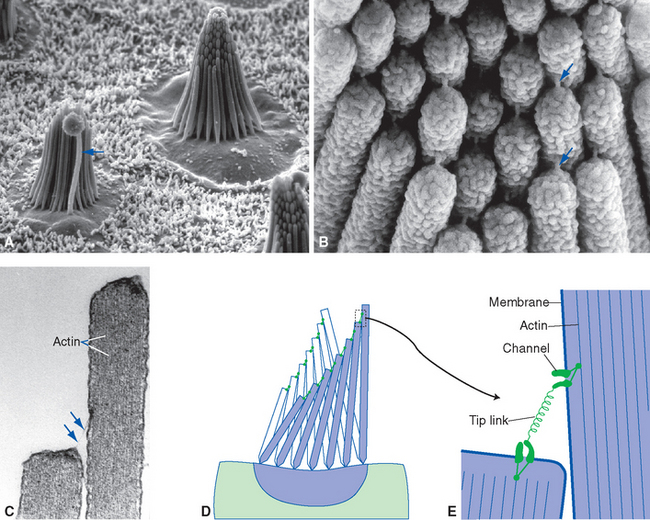
Hair cells of the labyrinth were named for the array of a hundred or so specialized microvilli that project as a bundle from one end of the cell into the endolymphatic interior of the membranous labyrinth (Fig. 14-4); the other end of each hair cell synapses on peripheral processes of eighth nerve fibers, which in turn convey auditory and vestibular information to the central nervous system (CNS). Hair cell microvilli, somewhat illogically referred to as stereocilia, are arranged asymmetrically: they are lined up in graduated rows, so that the tallest are toward one side of the hair cell. Adjacent to the tallest stereocilia of each hair cell in the semicircular ducts, utricle, and saccule is a single true cilium, the kinocilium. Cochlear hair cells have kinocilia that degenerate during fetal development, indicating that these processes play no essential role in the transduction process; kinocilia may instead be important for establishing the anatomical asymmetry of hair cells or for some mechanical connections of the hair cells in which they persist.
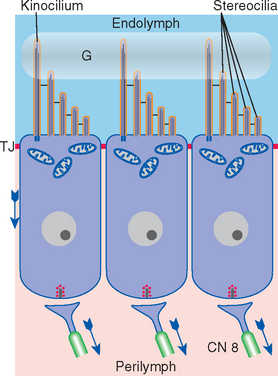
Figure 14-4 Schematic view of typical hair cells. Tight junctions (TJ) near the microvillar end of the cells join them to one another or to neighboring supporting cells, forming part of the diffusion barrier between endolymph and perilymph (see Figs. 14-5 and 14-12). Hence each hair cell is bathed partly in endolymph and partly in perilymph. The kinocilium (when present) and the tallest stereocilia are inserted into a gelatinous mass (G) that couples each hair bundle to mechanical forces. Transduction channels are located near the tips of the stereocilia, and arrows indicate the direction of information flow. CN, cranial nerve.
Hair cells are grouped into six discrete clusters in different parts of the labyrinth (Table 14-1; see also Fig. 14-3), and fundamental aspects of the arrangement of each cluster are the same. The stereocilia of the hair cells (and the kinocilium, when present) protrude into the endolymphatic space inside the membranous labyrinth, where the tips of the kinocilium and the tallest stereocilia are usually embedded in a specialized mass of gelatinous material, one for each cluster of hair cells. Movement of the gelatinous mass relative to the hair cells causes deflection of stereocilia, which in turn causes a receptor potential through a transduction mechanism described a little later.
In sections through the labyrinth (see Fig. 14-9) it often looks as though the hair cells and associated structures are bathed in the endolymph that fills the membranous labyrinth. If this were the case, however, eighth nerve fibers reaching the bases of hair cells would also be passing through endolymph. Endolymph has such a high K+ concentration that standard nerve fibers could not work in its presence. In fact, the real barrier between endolymph and perilymph is a series of tight junctions near the tops of the hair cells, between them and neighboring supporting cells. Because perilymph is continuous with the CSF of subarachnoid space, marker substances introduced into cisterna magna infiltrate the sensory epithelia of the labyrinth and surround the hair cells, stopping only at the array of tight junctions (Fig. 14-5). Hence the stereocilia and the apical surfaces of hair cells are exposed to endolymph, whereas other surfaces of the hair cells, and the nerve fibers they contact, are bathed in perilymph (see Fig. 14-9E).
Hair Cells Have Mechanosensitive Transduction Channels
Stereocilia are packed full of cross-linked actin filaments (Fig. 14-6C), which makes them rigid. In response to mechanical deformation they do not bend but rather pivot at their bases, where they are attached to the hair cell. Various linking molecules interconnect neighboring stereocilia, so the whole hair bundle moves as a unit in response to mechanical stimuli. Some of these links are symmetrical, connecting a given stereocilium to all of its neighbors. In addition, fine filamentous connections called tip links extend from the tip of each stereocilium to its next tallest neighbor (Fig. 14-6). Tip links have a special role in the transduction process, which (at least in a conceptual sense) is remarkably straightforward (Fig. 14-7). A mechanically gated cation channel is located at one or both ends of each tip link and is normally open part of the time. Deflecting the hair bundle toward the tallest stereocilia stretches the tip links, increasing the probability of channel opening. The channels are permeable to most small cations, and because K+ ions are the most abundant cations in endolymph, they flow down the electrical gradient from the positive endolymph into the negative interior of the hair cells. The resulting inward K+ current depolarizes the hair cells,* causing the opening of voltage-gated Ca2+ channels and increased release of transmitter onto eighth nerve endings. The excitatory transmitter (probably glutamate) then causes an increased firing frequency in the eighth nerve fibers. Deflecting the hair bundle in the opposite direction decreases the tension on the tip links; the transduction channels close, baseline K+ current stops, the hair cells hyperpolarize, and transmitter release and firing rate diminish. Deflecting the hair bundle in a perpendicular direction (i.e., parallel to the rows of stereocilia) has no effect on the tip links and so does not cause a receptor potential.
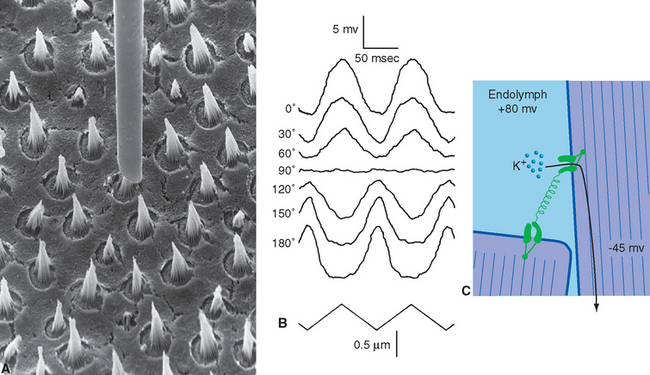
Figure 14-7 Transduction in hair cells. The otolithic membrane (see Fig. 14-27) was removed from the macula of a bullfrog’s saccule. A, A glass micropipette was slipped over a bundle of stereocilia and used to wiggle it in various directions. B, Receptor potentials were simultaneously recorded using a second micropipette (not shown). The bottom trace indicates the time course of 0.5-μm movements, and the traces above show the dependence of the resulting receptor potentials on the direction of deflection. Movement toward the kinocilium (0 degrees) produces a depolarizing receptor potential; movement away (180 degrees) produces a hyperpolarizing receptor potential. Movement perpendicular to the plane of the tip links (90 degrees) has no effect on the hair cell. C, Stretching a tip link opens cation channels in the stereocilia. Even though the K+ concentration in endolymph is about the same as that inside the stereocilia, the positive endolymphatic potential and the negative membrane potential of the hair cell combine to form a large electrical driving force that moves K+ ions through these open cation channels.
(A and B, from Shotwell SL, Jacobs R, Hudspeth AJ: Ann N Y Acad Sci 374:1, 1981. C, modified from a drawing provided by Drs. David Corey and G. M. G. Shepherd, Howard Hughes Medical Institute, Harvard Medical School.)
Subtle Differences in the Physical Arrangements of Hair Cells Determine the Stimuli to Which They Are Most Sensitive
Hair cells in all parts of the labyrinth use the same basic transduction mechanism, initiated by movement of a gelatinous mass and deflection of stereocilia. The critical variable in different parts of the labyrinth is the physical coupling between the gelatinous masses and the stereocilia. This is the key: variations in the way these gelatinous masses are made up and arranged, and the way different parts of the membranous labyrinth are suspended mechanically, set up some hair cells to respond to sound, others to head movement, and still others to head position.
The Cochlear Division of the Eighth Nerve Conveys Information About Sound
The auditory system faces a basic mechanical problem: the sound vibrations that it must detect are propagated in air, whereas the auditory receptor cells (like other elements of the nervous system) live in a fluid-filled environment. Water is harder to move than air, and nearly all the sound energy incident on a simple air-water interface is reflected. Fluids in small channels like the labyrinth are even harder to move, with the result that if the auditory receptor organ (the organ of Corti) and its fluid surroundings were mechanically coupled to the outside world by a simple membrane, it would receive no more than 0.1% of the sound energy that fell on the membrane. One major task of the air-filled outer and middle ears (Fig. 14-1) is therefore to transfer sound as efficiently as possible to the fluid-filled inner ear.
The Outer and Middle Ears Convey Airborne Vibrations to the Fluid-Filled Inner Ear
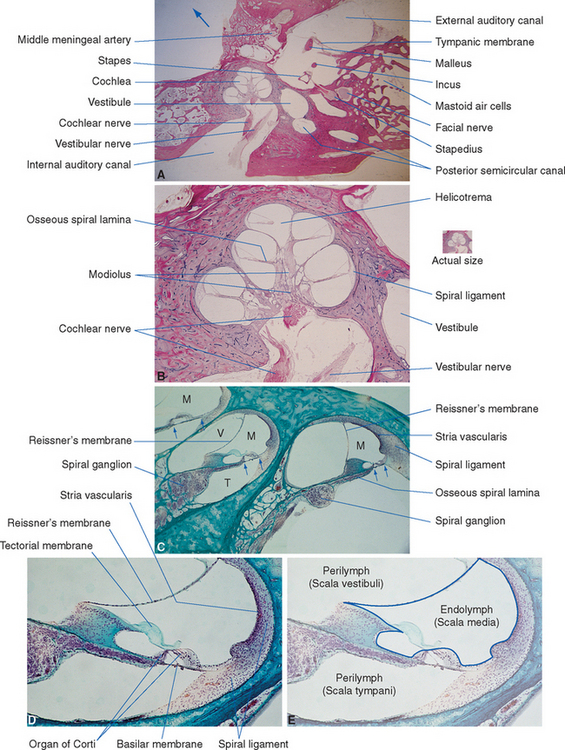
The outer ear is basically a complicated funnel consisting of the auricle (or pinna) and the external auditory meatus or canal; it conducts sound to the tympanic membrane. Sound-induced vibrations are then transferred along a chain of three small bones, or ossicles, that traverse the middle ear cavity (an air-filled cavity in the temporal bone). The handle of the malleus is attached to the medial surface of the tympanic membrane, so movements of this membrane are transferred directly to the malleus. The malleus in turn is attached to the incus, which is attached to the stapes, so sound-induced vibrations eventually reach the oval-shaped footplate of the stapes. The footplate of the stapes occupies a hole in the temporal bone called the oval window; on the other side of the oval window is the perilymph-filled vestibule of the bony labyrinth. The vestibule leads directly to the cochlea, which contains the organ of Corti. Thus, vibration of the tympanic membrane ultimately results in movement of the fluids of the inner ear.
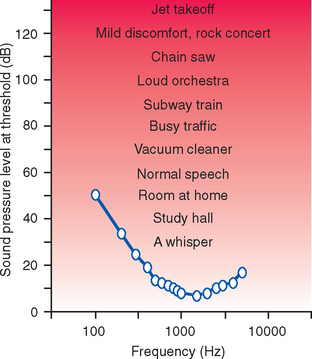
The chain of middle ear ossicles acts as a lever system with a small mechanical advantage, so a given force at the tympanic membrane results in a slightly greater force at the footplate of the stapes. More importantly, the active, or moving, area of the tympanic membrane is about 15 times that of the footplate of the stapes. The net result of the mechanical advantage and the size difference is that stapedial vibrations have a much greater force per unit area of the footplate; this force is sufficient to move the perilymph, and more than 60% of the sound energy incident on the tympanic membrane is successfully transferred to the inner ear. Hence the middle ear apparatus acts as a transformer, much as an electrical transformer alters the voltage and current of a source to better match the requirements of a particular circuit. The effectiveness of this system is quite extraordinary. At a threshold of 3000 Hz (the frequency to which we are most sensitive) the tympanic membrane moves a distance somewhat less than the diameter of a single hydrogen atom.* By the time such a threshold vibration of the tympanic membrane reaches the cochlear hair cells, it deflects their stereocilia through an angle of only about 0.003 degree. Bending the Empire State Building through an angle of 0.003 degree would deflect its top by less than an inch!† From this threshold we can hear over a 10 million–fold range of sound pressure levels before sounds become painfully loud. (Because it is cumbersome to keep track of all the zeros in such large numbers, a logarithmic decibel scale, as described in Box 14-1, is used to express sound pressure levels.) In addition, although we are most sensitive at about 3000 Hz, the frequency range of human hearing in healthy young adults extends from about 20 to 20,000 Hz (Fig. 14-8). All this is accomplished with a surprisingly small number of nerve fibers: in contrast to the million or so axons in an optic nerve, there are only about 30,000 fibers in a human cochlear nerve. Somehow, the CNS analyzes the information carried by these 30,000 fibers, enabling us to detect and interpret the myriad sounds of nature, music, and human language.
BOX 14-1 The Frequency and Intensity Range of Human Hearing
So a sound pressure level 10 million times greater than threshold is 140 dB above threshold (Fig. 14-8):
Multiple mechanical properties of the outer, middle, and inner ears collaborate to determine the sensitivity of ears to sounds of various frequencies. The combined result of all these factors is that we are most sensitive in the 1000- to 3000-Hz range important for spoken language, somewhat less sensitive at higher frequencies, and much less sensitive at lower frequencies (Fig. 14-8).
The Cochlea Is the Auditory Part of the Labyrinth
The bony part is the cochlea, which coils through 2¾ turns from its relatively broad base to its apex. (The cochlea lies on its side in the temporal bone, with its base facing medially and posteriorly [see Fig. 14-26], but for the sake of simplicity, it is usually discussed as though it sits upright on its base.) The cochlea has a core of spongy bone called the modiolus, from which the osseous spiral lamina projects like the threads of a screw (Fig. 14-9). A winding cavity within the modiolus parallels the spiral lamina and houses the spiral ganglion, which contains the cell bodies of the auditory primary afferent fibers. The central processes of these cells collect at the base of the cochlea to form the cochlear division of the eighth nerve; the peripheral processes pass in bundles through a series of canals in the osseous spiral lamina to innervate the auditory receptors.
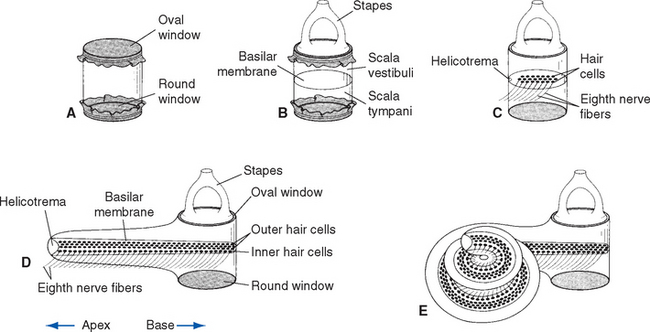
The cochlear duct (the auditory portion of the membranous labyrinth) is firmly anchored to the bony labyrinth in such a way that the duct is triangular in cross section (Fig. 14-9C). One corner of the triangle is attached to the osseous spiral lamina, and the other two corners are attached to the outer wall of the bony cochlea. The result is that the cochlear duct and osseous spiral lamina act as a partition between two perilymphatic spaces (except at the apex of the cochlea, where perilymph can pass from one space to the other through a small opening called the helicotrema [Greek for “the hole in the spiral”]). The perilymphatic space above the cochlear duct is called scala vestibuli because it is directly continuous with the perilymph of the vestibule. The space below the cochlear duct is called scala tympani because it ends blindly at the round window membrane (also called the secondary tympanic membrane). The space enclosed by the cochlear duct is filled with endolymph and is called scala media. Each of the three walls of the cochlear duct has a different structure (Fig. 14-9D). The thin vestibular (or Reissner’s) membrane borders scala vestibuli and probably serves mainly as a diffusion barrier between the endolymph and perilymph, playing no great role in the mechanical properties of the cochlea. The spiral ligament, thickened periosteal tissue adhering to the inner surface of the bony cochlea, forms the second wall. It includes on its endolymph-facing surface the stria vascularis, a specialized secretory epithelium that produces most of the endolymph in the membranous labyrinth. Finally, the basilar membrane spans the gap between the edge of the osseous spiral lamina and the spiral ligament, completing the “floor” of the cochlear duct and separating scala media from scala tympani.
Vibrations reaching the stapes footplate are transferred to the perilymph of the vestibule, adjacent to scala vestibuli. Although perilymph is incompressible, the round window membrane is elastic, allowing these vibrations to enter the labyrinth. When the stapes footplate moves inward, the round window membrane bulges out; when the footplate moves outward, the membrane is drawn inward. In the process, small quantities of perilymph are displaced within the cochlea. Most of this energy passes directly from scala vestibuli to scala tympani, deforming the cochlear duct (Fig. 14-10). The cochlear duct contains the auditory receptors, and this deformation stimulates some of them. Static pressure changes and vibrations of very low frequency simply move a little perilymph through the helicotrema and do not deform the cochlear duct.
Traveling Waves in the Basilar Membrane Stimulate Hair Cells in the Organ of Corti, in Locations that Depend on Sound Frequency
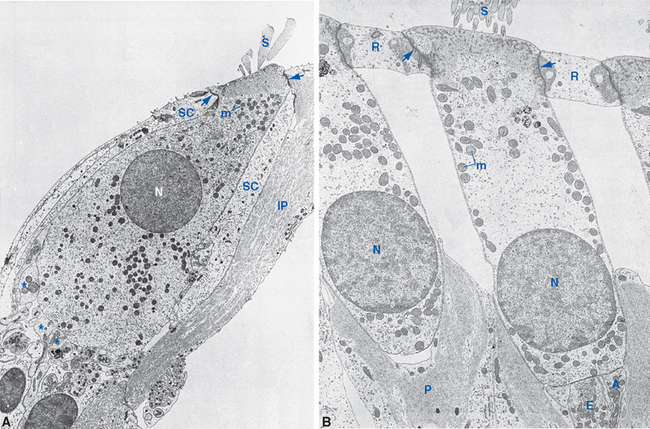
The organ of Corti (Figs. 14-11 and 14-12) is a strip of hair cells and supporting cells about 35 mm long that rests on the basilar membrane. The hair cells are arranged in two groups: a single row of about 3500 inner hair cells near the edge of the osseous spiral lamina, and a band of about 15,000 outer hair cells three to five cells wide, directly above the flexible basilar membrane. The two groups are separated by a perilymphatic space called the tunnel of Corti, through which the peripheral processes of eighth nerve fibers must pass on their way to the outer hair cells. The stereocilia of the outer hair cells are inserted into the gelatinous tectorial membrane, so that vibration of the basilar membrane causes oscillations of the hairs and therefore oscillation of the membrane potential of the hair cells. The stereocilia of the inner hair cells, in contrast, are not attached to the tectorial membrane; these hair cells apparently are stimulated directly by endolymph squirting back and forth through the narrow space between their tops and the tectorial membrane.
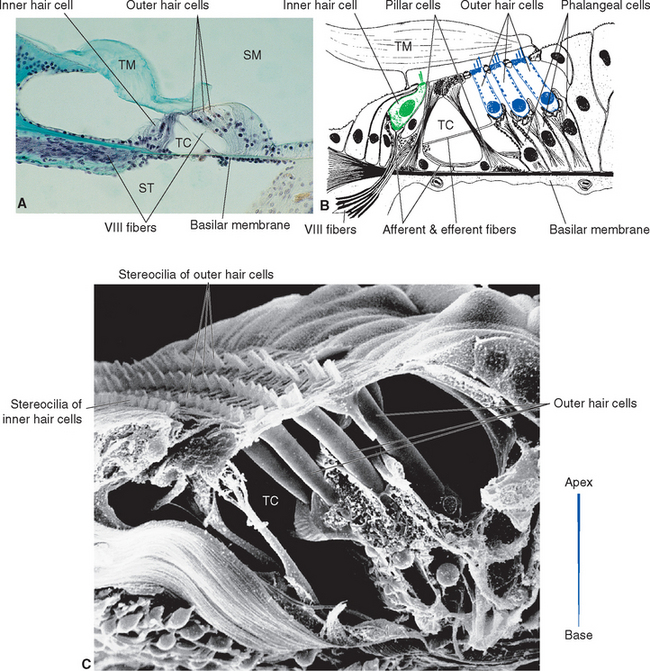
Figure 14-11 Structure of the organ of Corti. A, Light micrograph of a human organ of Corti (enlarged from Fig. 14-9D). B, Drawing of a cross section of the organ of Corti. Of the several types of supporting cells in the organ of Corti, two in particular make major contributions to its mechanical stability. The pillar cells produce microtubule-filled processes (see Fig. 14-12A) that frame the tunnel of Corti (TC). The phalangeal cells (Deiters’ cells) form cup-shaped, microtubule-filled structures that support the outer hair cells (see Fig. 14-12B); thin processes of these cells also extend to the tops of the outer hair cells, where their platelike expansions fill the spaces between outer hair cells, forming the reticular lamina. C, Scanning electron micrograph of the organ of Corti of a guinea pig. The tectorial membrane has been removed, and the stereocilia of the three rows of outer hair cells can be seen protruding into scala media; normally these stereocilia would be embedded in the tectorial membrane. No inner hair cells are present in this view, but their stereocilia can also be seen protruding into scala media. To the right of C, the actual size of an unrolled basilar membrane is shown. SM, scala media; ST, scala tympani; TM, tectorial membrane.
(A, courtesy Dr. Allen L. Bell, University of New England College of Osteopathic Medicine. B, from Northern JL: Hearing disorders, ed 2, Boston, 1984, Little, Brown. C, from Bredberg G: In Evans EF, Wilson JP, editors: Psychophysics and physiology of hearing, New York, 1977, Academic Press.)
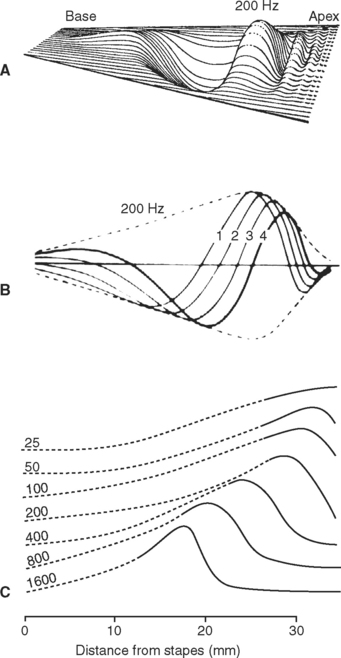
A pressure pulse delivered to scala vestibuli by movement of the stapes causes a traveling wave of deformation to move along the basilar membrane (Fig. 14-13A and B), much as waves spread from the site of a pebble dropped into a body of water. However, because the mechanical properties of the basilar membrane vary progressively along its length, the traveling wave reaches its peak amplitude at a location that depends on the frequency of the stimulus. The basilar membrane is about 100 μm wide and relatively stiff at the base of the cochlea, and about 500 μm wide and relatively floppy at the apex (Fig. 14-11, inset). The entire basilar membrane responds to intense low-frequency sounds, but closer to threshold it is driven most effectively by sounds of progressively higher frequencies as one moves from the apex to the base of the cochlea (Fig. 14-13C). Because the organ of Corti (which contains the auditory receptor cells) rests on the basilar membrane, different receptor cells respond best to sounds of different frequencies.* Individual eighth nerve fibers respond to a broad range of frequencies when the sound is intense but are sharply tuned to a narrow range of frequencies at threshold (see Fig. 14-15). This mechanical tuning of the basilar membrane is the beginning of a tonotopic organization within the auditory system, analogous to the somatotopic organization of the somatosensory system and the retinotopic organization of the visual system; in this case, particular frequencies are mapped in an orderly fashion onto particular areas of relay nuclei and auditory cortex (see Fig. 14-19).
< div class='tao-gold-member'>
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree