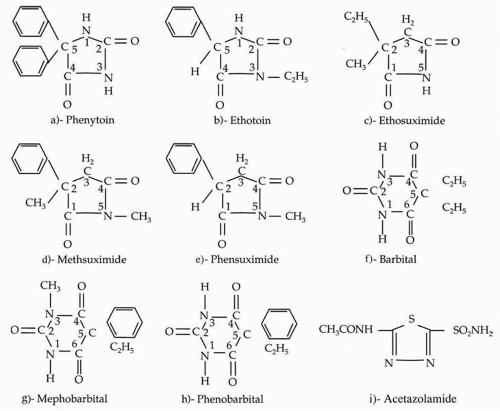
Chemistry and Mechanism of Action
The chemical structure of methsuximide (
N-2-dimethyl-2-phenyl-succinimide) is shown in
Figure 69.1D. Phenyl group substitution at the 2C position counteracts experimentally induced maximal electroshock seizures, whereas alkyl group substitution at the 2C position counteracts experimentally induced pentylenetetrazol seizures. Methyl group substitution at the 5N position adds to the antipentylenetetrazol effect and the sedative activity. Alkyl substitution at the 5N and 2C positions and phenyl substitution at the 2C position provide activity against pentylenetetrazol- and maximal electroshock-induced seizure activity (
11).
Methsuximide is a nonpolar chemical compound that is water soluble and slightly lipophilic. Its exact effects on excitable membranes are unknown. Because of its effectiveness against absence and partial seizures, the agent probably has more than one mechanism of action, including effects on transmitter release, calcium uptake into
presynaptic endings, and conductance of sodium, potassium, and chloride.
Absorption, Distribution, and Metabolism
Methsuximide is quickly absorbed through the GI tract, with peak plasma levels achieved in 2 to 4 hours. The agent is distributed evenly throughout the body and penetrates brain and fat tissue better than ethosuximide (
12). Because of its low protein binding and poor solubility, methsuximide equilibrates with CSF (B.J. Wilder, unpublished data, 1980). It is rapidly metabolized to
N-desmethyl-methsuximide or 2-methyl-2-phenyl succinimide (
12,
13,
14,
15) and has a mean half-life of 1.4 hours. Trough plasma concentrations of methsuximide are reportedly undetectable in fasting specimens (
16). A major active metabolite of methsuximide,
N-desmethyl-methsuximide, achieves high steady-state plasma levels and exerts a major anticonvulsant effect. The mean half-life of this metabolite is 38 hours (range, 37 to 48 hours) (
13), although some investigators (
16) have reported half-lives of 51.6 to 80.2 hours in patients who received maximal doses of methsuximide. Another methsuximide metabolite,
N-methyl-2-hydroxymethyl-2-phenylsuccinimide, was detected by means of gas chromatography and mass spectrometry of the serum of a patient with a fatal overdose of primidone and methsuximide.
Optimal clinical effect may be achieved with a nontrough
N-desmethyl-methsuximide plasma concentration of 20 to 24 μg/mL (
15), near the middle of the therapeutic range of 10 to 40 μg/mL reported by Strong and colleagues (
14). Browne and associates (
16) reported a therapeutic range of 10 to 30 μg/mL for fasting
N-desmethylmethsuximide plasma concentrations. Steady-state plasma concentration is reached between 8.1 and 16.8 days from onset of maintenance methsuximide dose. The usual dosage increase of 150 or 300 mg per day can be made at biweekly intervals to avoid toxicity. Methsuximide is no longer available in 150-mg tablets; biweekly dosage increments of one tablet (300 mg) every other day may be used (
16).
Efficacy and Clinical Use
Methsuximide has a wide spectrum of antiepileptic activity and is effective in patients with complex partial seizures (
15,
16,
17), generalized tonic-clonic seizures, and absence seizures (
18,
19,
20,
21). Wilder, Buchanan, and Uthman (
15,
22) found methsuximide to be an effective adjunctive agent in the management of refractory complex partial seizures. Twenty-one patients taking phenytoin, phenobarbital, primidone, or carbamazepine as monotherapy or in combination were studied. Of these patients, 71% achieved good to excellent control of complex partial seizures, and a dose reduction or discontinuation of 1 or more AED was possible in 42%. Optimal plasma levels and control of complex partial seizures were associated with daily methsuximide dosages of 9.5 to 11.0 mg/kg, with maximal seizure control observed at
N-desmethyl-methsuximide plasma levels of 20 to 24 μg/mL (conversion factor for methsuximide: μmol/L = 4.92 × μg/mL). A dose-response relationship was determined after the addition of methsuximide, and seizure frequency progressively decreased as
N-desmethyl-methsuximide serum levels increased.
Browne and colleagues (
16) described the use of adjunctive methsuximide in 26 patients with medically refractory complex partial seizures. The maximal tolerated dose of methsuximide was maintained for 8 weeks. Of the total population, eight patients (31%) had a 50% or more reduction in seizure frequency, and four (15%) became seizure free. Eight patients withdrew from the study because of adverse events and three because of increased seizure frequency (these patients had a history of severe seizure flurries before and after initiation of methsuximide treatment). Of the eight patients who responded, five continued to have a 50% or more reduction in frequency of complex partial seizures for 3 to 34 months.
Sigler and associates (
23) used methsuximide as add-on therapy in children with epilepsy refractory to first- and second-line AEDs. Forty patients (35.7%) had a 50% or more reduction in seizure frequency, and ten (8.9%) became seizure free during the short-term phase (mean, 9.1 weeks). Of the 112 patients studied, 22 (19.6%) continued to benefit from the drug (a 50% or more reduction in seizure frequency compared with baseline and absence of intolerable side effects) at long-term follow-up (mean, 3.7 years; range, 18 months to 7.1 years). In patients with good seizure control, fasting plasma levels of
N-desmethyl-methsuximide were 25.3 to 44.7 mg/L (mean, 36.0 mg/L); thus, effective plasma concentrations of
N-desmethyl-methsuximide in children were found to be higher than previously described. No serious or irreversible side effects were reported. Likewise, Tennison and colleagues (
24) used methsuximide as add-on therapy in children with complex partial and “minor motor” seizures refractory to first- and second-line AEDs; 15 patients (60%) had a 50% or more reduction in seizure frequency, and no serious adverse events were reported.
Other reports of methsuximide as adjunctive medical therapy for complex partial seizures showed complete seizure control in 0% to 38% of patients and a 50% or more reduction in seizure frequency in 6% to 100% of patients (
17,
19,
25,
26,
27,
28). In one study of previously untreated patients (
29), seizures were controlled in 18%, and 27% had a 50% or more reduction in seizure frequency.
Results of early studies of methsuximide showed some efficacy in patients with absence seizures (
19,
20,
21). In one study (
20), methsuximide was used in previously untreated patients; absences were not completely controlled in any patient, and only 20% had a greater than 50% reduction in
seizure frequency. Rabe (
30) reported that 10 of 16 patients became completely free of absences, and another five had seizure frequency reduced by 75%. He suggested two possible explanations for the greater effectiveness of methsuximide in his study compared with earlier work: Most of his patients had epilepsy of relatively recent onset, and he used considerably higher doses of methsuximide (1,200 to 2,100 mg per day) than did previous investigators.
Rabe also reported on the efficacy of methsuximide in four patients with juvenile myoclonic epilepsy (
30). Two patients became completely free of myoclonus, and two had a reduction in frequency of at least 75%. One case report described methsuximide used with primidone to be very effective in a 17-year-old boy with drawing-induced myoclonic seizures (
31). Hurst (
32) described five adolescent girls with juvenile myoclonic epilepsy who became seizure free taking methsuximide; four were maintained on monotherapy.
Tolerance to the anticonvulsant effect of methsuximide develops in approximately 50% of patients treated with maximal doses, and seizure frequency returns to baseline. The low overall efficacy of methsuximide relative to that of first-line AEDs may reflect the selectively more refractory seizures in the patients studied. Failures because of toxic reactions might have occurred when the dose of methsuximide was increased too rapidly. The dose should not be increased more often than every 2 weeks in adults receiving multidrug therapy (
16). Methsuximide should be considered in patients who are allergic to or whose disease is refractory to other AEDs.
Interactions with Other Agents and Adverse Effects
Methsuximide interacts with other AEDs, necessitating close monitoring of serum levels and adjustment of concurrent AED dose, especially in the face of clinical toxicity. Rambeck (
33) reported that concurrent administration of methsuximide increased the mean serum concentration of phenobarbital by 37% in patients receiving this agent and by 40% in patients receiving primidone. The mean serum concentration of phenytoin increased by 78%. Patients taking phenobarbital or phenytoin had increased serum levels of
N-desmethyl-methsuximide compared with patients taking methsuximide alone. These increases were attributed to competition by the drugs for a common hydroxylating enzyme system.
Conversely, the addition of methsuximide induces the metabolization of other AEDs (
34,
35). Methsuximide decreased the mean serum concentrations of carbamazepine (
16), valproic acid (
36,
37), lamotrigine (
35,
38), and topiramate (
39) when added to the treatment regimen. Methsuximide mitigated the effect of valproic acid on lamotrigine; the combination of valproic acid and lamotrigine increased the concentration of lamotrigine by 211% compared with lamotrigine monotherapy, however, if methsuximide was added, the increased concentration of lamotrigine dropped to 8% (
35).
GI disturbance, lethargy, somnolence, fatigue, and headache may be experienced, but these adverse effects are usually transient and dose-related. Other adverse experiences include hiccups, irritability, ataxia, blurred vision or diplopia, inattention, dysarthria, and psychic changes (
16). In some patients, headache, photophobia, and hiccups require withdrawal of methsuximide (
15). Transient leukopenia and a movement disorder have been reported (
40). Delayed, profound coma following methsuximide overdose has been described (
41). Charcoal hemoperfusion was successful in one case of methsuximide overdose (
42).