Chapter 140 Menopause
Abstract
Menopause is a normal event in a woman’s life. It is the physiologic centerpiece of a major developmental stage in the normal aging process. Most women now live long enough to become menopausal and can expect to live at least another 30 years beyond their final menstrual period. The number of postmenopausal women in the world is increasing due to longer life expectancy. In the United States, 27% of the population in 2000 consisted of women 45 years of age and older, and the estimate is now at 38%.1
Phases of the Menopausal Transition
Menopause is the anchor point of a woman’s transition to midlife and, because menstrual cycles can be irregular for many reasons, menopause can only be officially declared after 12 months of amenorrhea. Menopause occurs at a mean of 51.4 years of age for Western women, but the range can be as wide as 40 to 58 years of age. For many years, menopause was simply considered a consequence of depleted ovarian follicles (the primary source of estrogen and progesterone). However, it is now clear that it is a transitional process that begins several years before menopause itself, and continues for several years. This perimenopausal developmental transition in a woman’s life is a dynamic process that consists of a series of complex and interactive changes in the endocrine system and central nervous system that occur on a background of aging.2
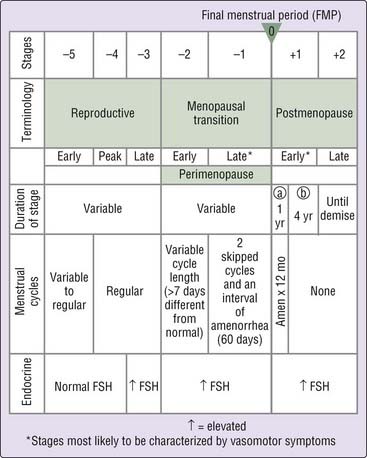
A menopause staging system has been developed to provide a consistent way to describe this transition in midlife women (Fig. 140-1).3 The early menopausal transition stage is characterized by elevated follicle-stimulating hormone (FSH) and a variable cycle length (more than 7 days’ deviation from normal). FSH levels increase in response to the absence of estrogen and progesterone that would normally provide negative feedback to the hypothalamus to inhibit FSH secretion. The late menopausal transition stage is characterized by two or more skipped menstrual cycles and an interval of amenorrhea (at least 60 days) as well as elevated FSH levels. The postmenopausal period is also divided into stages. The early postmenopause stage is defined as 5 years since the final menstrual period, and is subdivided into two segments: the first 12 months after the final menstrual period and the next 4 years. The late menopause stage starts 5 years after the final menstrual period and continues through the remaining life span.
Menopause is a universal phenomenon, but the timing of these transitional stages and associated signs and symptoms vary considerably from woman to woman. The age at which menopause occurs is strongly influenced by several factors: smoking, adiposity, race and ethnicity, and reproductive variables such as age at menarche, duration of breast-feeding, and use of oral contraceptives. The vasomotor signs and symptoms associated with menopause, such as hot flushes and night sweats, also vary to such a great extent that they are not specified as one of the staging criteria3 for ascertaining phases of menopause (see Fig. 140-1). The perimenopausal transition for an individual woman arises from a complex interaction of biological, cultural, psychological, and environmental factors.
The timing and duration of this midlife transition cannot be easily predicted. Midlife is considered to be between 40 and 60 years of age, but there is no generic midlife woman.4 In addition to the complexity of menopausal stages and hormonal fluctuations, midlife women can be having their first child, have grown children leaving home, and be caring for elderly parents or spouses. All of these scenarios can affect a woman’s sleep during menopause.
Normal Sleep Patterns During Menopause
Disturbed sleep and daytime fatigue are among the most common complaints of women during the menopausal transition.5,6 Population survey studies of menopause show that sleep problems are more common in perimenopausal and postmenopausal women compared with premenopausal women.
The prevalence of sleep disturbance varies widely by culture and from one study to another depending on how the sleep disturbance is defined and measured. As part of menopausal symptoms, trouble sleeping was reported by 54% to 57% of women between 40 and 60 years of age in the Ohio Midlife Women’s Study.6 The large Wisconsin Sleep Cohort Study found that perimenopausal and postmenopausal women were twice as likely to be dissatisfied with their sleep than premenopausal women.7 In the most comprehensive study of U.S. midlife women to date, the Study of Women’s Health Across the Nation (SWAN) has shown that difficulty sleeping is reported by 38% of women between 40 and 55 years of age, with higher levels among late perimenopausal (45.4%) and surgical postmenopausal (47.6%) women.8 Yet longitudinal data from the Penn Ovarian Aging Study would indicate that poor sleep is not necessarily associated with menopausal stage.9 The most common menopausal sleep complaint is difficulty falling asleep, but significant increases in self-reported nighttime awakenings and daytime drowsiness have also been described, even after controlling for confounding factors such as depression and age.4–10
Despite the incidence of insomnia complaints by menopausal women, very few studies have used objective techniques such as polysomnography (PSG) or actigraphy to assess sleep before, during, and after menopause. An actigraph study found that postmenopausal women underestimate their sleep time,11 and studies using PSG have not supported any prominent effect of menopause on sleep.7,12–15 One study may have shed more light on this discrepancy between women’s self-report and PSG findings. When Freedman and Roehrs recorded sleep for 4 nights in the laboratory, the 18 postmenopausal women with hot flushes had more arousals and disrupted sleep, but only in the first half of the night when there was less REM sleep.16
In addition to carefully controlled cooler ambient temperature in the sleep laboratory compared to the home, PSG may be less sensitive in detecting some important unknown element of sleep quality reflected by women’s ratings. Another possibility is that the impact of menopause on objective sleep measures is heterogeneous with a subgroup of women whose objectively measured sleep quality shows a menopause-related decline. This would not be surprising, given the heterogeneity of women’s experiences with hot-flush symptoms. Cultural attitudes and expectations about menopause can also influence women’s experiences and might affect perception of sleep quality.17
One prominent theory of reproductive aging is that menopause results from the aging of multiple pacemakers in the brain and ovaries that control and coordinate a variety of circadian and other rhythms.2 The suprachiasmatic nuclei of the hypothalamus are a primary source of these endogenous rhythms and their synchrony. The sleep–wake cycle is the most visible human circadian rhythm, and it is profoundly influenced by the suprachiasmatic nuclei and by other circadian rhythms, particularly rhythms of body temperature and melatonin secretion. Numerous studies have shown estrogen’s impact on circadian rhythms in female mammals.18 These data suggest that the circadian control of sleep might be changed or compromised with menopause. However, this hypothesis is purely speculative at present. No studies have attempted to distinguish menopause effects on circadian rhythm from the well-known effects of aging per se, and aging is repeatedly found to be associated with a decline in sleep quality regardless of sex.19,20
Although investigators studying midlife women (40 to 60 years of age) usually consider menopausal status when describing women’s health and experiences, the effects of depression and obesity are often ignored, and most sleep effects ascribed to menopause may in fact be due to aging. One approach to this issue is to control the confounding factors, such as age, with statistical techniques and treat age as a covariate.7 Another approach is to report experiences and symptoms for specific narrow age ranges in midlife, such that the specific contributions of age and menopause can be identified.4,12 These techniques might not be entirely successful because there is little overlap between ages of premenopausal and postmenopausal women. In short, sleep disturbance in some midlife women may be related to conditions associated with aging (e.g., thyroid dysfunction, depression, obesity) rather than menopause.
Common Clinical Conditions in Midlife Women
Hot Flushes and Night Sweats
It has long been suspected that at least some of the self-reported sleep disturbance associated with menopause is secondary to vasomotor symptoms of hot flushes and night sweats. A hot flush (often called a night sweat when it occurs during the night) is a sudden, transient, and recurrent subjective sensation of moderate to intense heat that usually begins in the upper body. It is primarily a thermoregulatory phenomenon21 with the characteristics of a heat dissipation response: peripheral vasodilation, which causes increased heat loss, and increased sweating, which causes evaporative cooling.
There is tremendous individual variability in hot flushes, likely a result of the variation in individual thermoneutral zones regulated by the hypothalamus. It is hypothesized that before menopause, when endogenous estrogen still interacts with the sympathetic nervous system, the thermoneutral zone is wider and thus it is easier to regulate body temperature within that zone. With inconsistent estrogen secretion during perimenopausal transition, or with absence of estrogen at menopause, the thermoneutral zone narrows considerably. This also occurs with chronic stress and increased sympathetic activation and has implications for therapeutic approaches to treat vasomotor menopausal symptoms with stress-reduction modalities rather than exogenous estrogen therapy.21
A hot flush typically lasts 3 to 5 minutes, but can be more than 20 minutes in some women. The perceived intensity of the flush can also vary widely, from severely disruptive to mild. Some women have hot flushes 20 or more times every day, whereas others report only 1 or 2 per week.2 The average number of hot flushes was 4.5 during the day and 2.0 per night from a study of women in the Pacific Northwest using self-report.22
Although hot flushes are generally associated with menopause, they are experienced during androgen-suppressive hormone treatment for men with prostate cancer, with use of selective estrogen receptor modulators such as tamoxifen for treatment of estrogen-sensitive breast cancer, and in diabetes or glucose intolerance. Surgical menopause, smoking, obesity, depression, and physical inactivity increase the likelihood of hot flushes.23,24
The prevalence of hot flushes also varies by racial and ethnic group. In the SWAN cohort of 15,000 women, approximately 39% of African Americans experienced hot flushes, compared with 26% of Latin Americans and 24% of whites. Asian Americans had the lowest prevalence (16% for Chinese Americans and 12% for Japanese Americans).23 It is not clear whether differences in the experience of hot flushes are the result of differences in lifestyle stressors, diet, cultural factors, or unidentified biologic factors.17
Hot flushes and night sweats are associated with reduced self-reported sleep quality, and this has been confirmed in a variety of study designs, ethnic groups, and nationalities.25,26 However, little is known about how hot flushes affect objectively measured sleep quality. Only a few studies with very small sample sizes have used PSG and a concurrent objective measure of hot flushes to compare sleep in women with and without hot flushes. They show that hot flushes disrupt sleep, as indicated by more nocturnal awakenings, lower sleep efficiency, and more sleep stage changes.27,28 Erlik and colleagues also assessed the effect of each nocturnal hot flush on sleep in nine menopausal women; of the 47 objectively measured hot flushes, 45 were associated with an awakening that occurred within 5 minutes before or after the flush.28
Some research suggests that hot flushes are unrelated to PSG sleep quality. Most of these studies relied on self-reports of hot flushes, often for nights other than the PSG nights. This is problematic because women are not always aware of hot flushes that occur during sleep. Freedman and Roehrs recently measured occurrence of hot flushes in relation to PSG awakening and documented that symptomatic women have 1 to 18 flushes during the night (average 5.2 ± 2.9). Some women who thought they were asymptomatic actually had hot flushes during sleep, and one women reported hot flushes and did not have a recorded hot flush while in the laboratory. A majority of awakenings (55%) occurred up to 2 minutes prior to the hot flush, but there was no difference in overall sleep efficiency between the symptomatic (83%) and asymptomatic (87%) groups.29 Upon further analysis, these awakenings occurred during the first part of the night and preceded the hot flush; of the 125 flushes that occurred during the second half of the night, none were during REM sleep, and the asymptomatic group had significantly more REM sleep (48 minutes) than the symptomatic group (35 minutes) during the second half of the night.16 Because thermoregulation is suppressed during REM sleep, these findings provide new information to help with reconciling discrepancies between women’s self-report of poor sleep associated with vasomotor symptoms and PSG findings of little difference in sleep architecture or wake time.
Data from large population studies suggest that some women continue to have hot flushes for many years, even decades, after menopause.30 Hot flushes are not always labeled “hot flushes” by older postmenopausal women because of their previous association with menopause. Rather, they may describe their vasomotor symptoms as waking up during the night “feeling too warm” and feeling the need to throw off bedcovers. Finally, not all women who have menopause-related sleep problems complain of hot flushes.31 This suggests that there are other reasons menopause may be associated with sleep disturbance.
Sleep disruption associated with hot flushes may be treated in several ways. HRT has historically been the standard treatment. Although HRT is still recommended for the short-term alleviation of vasomotor symptoms in women who do not have a history of breast cancer or stroke, patients and health care providers are increasingly reluctant to pursue this option because of health risks. Many other options are available.32 Antidepressants that inhibit serotonin reuptake (selective serotonin reuptake inhibitors [SSRIs]; e.g., paroxetine)33 and gabapentin34 have been used with some success. Complementary therapies help some women,32,35 although cohort data on effectiveness has been mixed. Relaxation therapies and stress reduction can improve thermoregulation, and appropriate changes in temperature of the sleeping environment and bedclothes may be helpful because a warm ambient temperature or elevated body core temperature increases the likelihood of hot flushes.16
Effects of Hormone Replacement Therapy on Sleep
Estrogen and progesterone levels fluctuate dramatically during the perimenopause and ultimately decline to very low postmenopausal levels.2 Consequently, estrogen therapy or HRT was commonly prescribed for midlife and older women on a long-term basis in an effort to counter hormone deficiency and protect against osteoporosis, heart disease, and Alzheimer’s dementia. However, the Women’s Health Initiative results abruptly reversed this practice. This national clinical trial showed that use of a common HRT regimen for 1 to 7 years significantly increased the risk of breast cancer, stroke, heart disease, and vascular dementia.36 Women are now being advised to use HRT for only a short time to provide relief from hot flushes and other symptoms during the menopausal transition.
Dozens of epidemiologic studies and small clinical trials have shown that estrogen therapy or HRT reduces hot flushes and concurrently improves self-reported sleep quality.37 The size of this effect varies from a significant but clinically small effect38 in some studies to other studies that document alleviation of hot flushes as highly predictive of improvements in sleep.31 PSG studies have generally replicated these findings. In women who were experiencing frequent hot flushes, estrogen therapy decreased the number28,39 and duration39 of nighttime awakenings, increased REM sleep,39–41 and shortened latency to sleep onset.40 However, in studies where many or all of the women had only mild or infrequent hot flushes, neither estrogen therapy42 nor HRT43 had an effect on PSG recorded sleep stages. The PSG studies were based on small groups of less than 20 women.
Progesterone
Virtually nothing is known about how changes in endogenous progesterone levels during menopausal transition affect sleep. Progesterone given by itself can have acute effects of sleepiness similar to sedative-hypnotic medications. A high clinical dose (300 mg) of micronized progesterone given orally 1.5 hours before bedtime significantly shorted latency to REM sleep and increased non-REM (NREM) sleep in a small group of young men.44 There are also numerous clinical reports and anecdotes about sleepiness as a side effect of progesterone, particularly daytime drowsiness that occurs in a significant percentage of women after taking progesterone in the morning as part of HRT.45 Clinically, women on HRT are often advised to take their progesterone in the evening because of this daytime drowsiness phenomenon.45
There may be significant differences among progestins in their impact on sleep. Montplaisir and colleagues46 found that conjugated equine estrogens (Premarin) plus micronized progesterone significantly improved the sleep of postmenopausal women, whereas the same estrogens plus the synthetic medroxyprogesterone acetate had no effect. This is consistent with emerging evidence of significant differences between progestins in their nonendometrial biological effects.47 For example, unlike medroxyprogesterone acetate, micronized progesterone is metabolized to potent neurosteroids such as allopregnanolone and pregnanolone. These neurosteroids interact with the same brain gamma-aminobutyric acid type A (GABAA) receptors as sedative-hypnotic medications, and they are soporific.48
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree