[18F]FDG-PET and Temporal Lobe Epilepsy
The greatest clinical experience in evaluating patients with partial epilepsy has been gained with [
18F]FDG-PET. Several studies have identified interictal regional decreases in glucose consumption that are invariably ipsilateral to the seizure focus—typically, but not always, most pronounced in the temporal lobe (
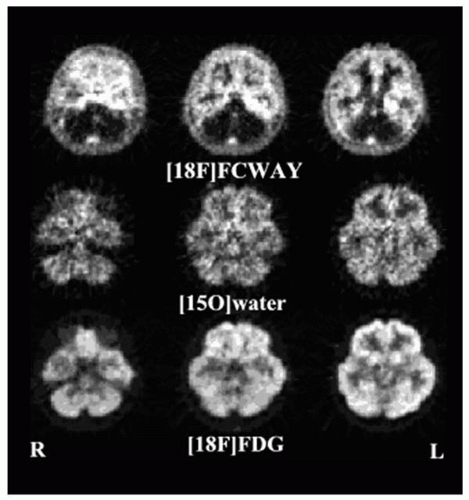
Fig. 76.1) (
4,
5,
6).
Regional hypometabolism is seen in 65% to 90% of patients with temporal lobe epilepsy; the figure is closer to 90% on recent-generation scanners and to 60% for patients who show normal findings on MRI (
7,
8,
9,
10). The area of decreased glucose utilization is often more extensive than the epileptogenic zone; it may extend into adjacent inferior frontal or parietal lobe neocortex (
6,
11,
12,
13) and occasionally into ipsilateral thalamus (
12,
14) and contralateral cerebellum (
6,
11,
12). The regional abnormalities are invariably unilateral to the ictal focus; however, lobar localization is somewhat less reliable, about 80% to 90%. The few reports of false lateralization have occurred after surgery (
5), when interpretation relied on nonquantitative analysis, or during subclinical seizures (
5,
15,
16). Focal interictal regional hypometabolism also predicts a good surgical outcome (
8,
17,
18,
19). Different investigators using different methods and regional analyses have found different regional hypometabolism predictive of good outcome: inferior lateral temporal, anterior lateral, and uncus (
8,
17,
19). Bilateral temporal hypometabolism portends a less optimistic surgical outcome and in half of patients reflects bilateral foci (
20). Patients with focal temporal abnormalities have a 93% chance of good surgical outcome; those without have only a 63% chance (
18,
19). The ability to confirm the focus and predict surgical outcome improves with quantitative means, typically when asymmetry indexes [AI; e.g., AI = 2(left − right)/(left + right)] are greater than two standard deviations from normative data, or about 10% to 15% (
18). Lesser degrees of asymmetry, though visually apparent, may result in misleading information and erroneous conclusions (
7,
18). Voxel-based statistical methods, performed in a standard anatomic atlas that allows comparison of individual patient images to normal control group data, have been advocated as an alternative means of reliable analysis (
21,
22). Given that [
18F]FDG-PET is often performed to confirm the focus, focal abnormalities may reduce the need for, or extent of, invasive monitoring when laterality of the focus is in doubt (
5,
18,
19). Issues of frontal versus temporal focus may not always be reliably resolved by interictal [
18F]FDG-PET studies, and invasive studies or other PET ligand studies may be needed. Conflicting localizing or lateralization data nearly always merit invasive monitoring.
Ictal [
18F]FDG-PET studies are uncommon because of technical constraints such as ligand availability and unpredictability of seizures. They may show profound focal increases in glucose consumption but may also demonstrate normal results or decreased consumption. The results depend on the delivery of ligand, time and duration of the seizure, and degree of offsetting postictal hypometabolism. Although interesting, they have limited clinical use (
23).
The reasons for regional hypometabolism are incompletely understood. Glucose consumption occurs primarily at the synapse. Regional hypometabolism appears to reflect a decrease in glucose influx from reduced glucose transport across the blood-brain barrier, which correlates with subsequent reduced phosphorylation (
24). Cell loss with ensuing synaptic loss and altered remote projections, as occurs with hippocampal atrophy in mesial temporal sclerosis, may account for a portion, but not all of regional hypometabolism in temporal lobe epilepsy. (
25,
26,
27,
28,
29,
30,
31). Hypometabolism does not correlate with lifetime generalized tonic-clonic seizures or frequency of complex partial seizures (
32). Dysplastic tissue with aberrant synaptic connectivity can have either decreased or normal glucose consumption (
33,
34). The abnormalities in some circumstances appear
to be functional, as some patients have profound decreases in glucose uptake and no discernible pathologic lesion; regional decreased glucose uptake may vary with relation to previous ictal events (
35) and clinical manifestations of the previous seizure (
13). In patients with mesial temporal sclerosis, the predominant regions that may manifest decreased glucose consumption are the lateral neocortex and, to a lesser extent, the frontal cortex. This may reflect the distant projection of functional loss in mesial structures. Frontal and contralateral hypometabolism appear to be reversible with successful temporal lobectomy (
26,
36,
37).
Studies differ in the extent to which patients with mesial temporal seizures show pronounced lateral hypometabolism: Mesial greater than lateral, lateral greater than mesial, and equal mesial and lateral temporal reductions in glucose uptake have been reported (
6,
7,
38,
39,
40). Patients with neocortical temporal epilepsy may have greater lateral than mesial metabolic abnormalities (
39). There is sufficient variability among patients that individual predictions of seizure focus within the temporal lobe cannot be based on [
18F]FDG-PET.
[
18F]FDG-PET will be abnormal when MRI shows significant abnormalities, for example, in mesial temporal sclerosis, tumor, vascular malformation, infarct, and most instances of cortical dysplasia. In this setting, [
18F]FDG-PET provides little additional information beyond that of MRI. [
18F]FDG-PET may be more sensitive than MRI in temporal lobe epilepsy in some circumstances. Current PET techniques are helpful in 85% to 90% of patients, volumetric MRI in 60% to 70%, and magnetic resonance spectroscopy (MRS) in 55%. Higher-resolution scanning techniques, including high-resolution fast spin echo, fluid-attenuated inversion recovery, T2 relaxometry, magnetization transfer, and high-resolution thin-cut spoiled gradient recall anatomic sequences, have reduced the utility of [
18F]FDG-PET (
9,
27). Comparison studies report varying efficacy results with different imaging modalities, which generally reflect the particular research strengths of the investigators rather than the intrinsic advantages of the techniques studied.
Although glucose consumption in temporal cortex is decreased, perfusion is often maintained, especially in lateral neocortex (
7,
41,
42). Interictal studies of cerebral blood flow using [
15O]water find a decrease in perfusion in only 50% of patients, but one fifth of these provide falsely localizing information (
7) (
Fig. 76.1). This experience is similar to that in interictal SPECT studies and quantitative perfusion ascertained by arterial spin-labeled fMRI (
8,
43,
44). These data suggest that vascular tone may be impaired in temporal lobe epilepsy and that the relationship between metabolism and perfusion is altered. For these reasons, interictal blood flow studies are unreliable markers of the epileptogenic zone and do not predict surgical outcome (
7,
45).
Metabolic abnormalities are less common in patients with recent-onset, nonrefractory, or well-controlled partial epilepsy. Within less than 3 years of seizure onset, 30% of adults with nonlesional epilepsy have focal findings on [
18F]FDG-PET (
46). From 40% to 50% of adults without refractory seizures of limited duration (more than 5 years) have focal abnormalities (
46,
47). In other studies, 20% of adults with well-controlled partial seizures had regional metabolic abnormalities (
48). In these adult populations, localization of seizures is less certain than in patients with refractory epilepsy—an important consideration because patients with extratemporal lobe epilepsy are less likely to have abnormal [
18F]FDG-PET studies (
49).
Chronic partial epilepsy typically begins during childhood. In a study of 40 children with recent-onset partial epilepsy (mean duration, 1 year) and normal MRI (except for mesial temporal sclerosis), 20% demonstrated regional hypometabolism, all ipsilateral to the presumed focus. All the abnormalities were found among the 32 children with a suspected temporal lobe focus. Although this population is at high risk for continued seizures, not all the children will ultimately develop refractory epilepsy; it remains to be seen whether [
18F]FDG-PET can predict epilepsy prognosis (
50). In contrast, 70% of children with chronic partial epilepsy (duration 10 years) have focal metabolic abnormalities. There is evidence that adult patients with a longer duration of epilepsy are more likely to have focal [
18F]FDG-PET abnormalities (
6,
41,
50). Partial seizures of longer duration are also associated with a greater dissociation between metabolism and blood flow (
7,
41). These [
18F]FDG and cerebral blood flow studies, along with cross-sectional studies using volumetric MRI, may be taken as evidence that temporal lobe epilepsy in some patients is associated with chronic and continued neuronal injury (
41,
51,
52,
53).
PET in Extratemporal Lobe Epilepsy
[
18F]FDG-PET is less efficacious in identifying the epileptogenic zone in extratemporal lobe epilepsy than in temporal
lobe epilepsy. Most extratemporal lobe epilepsy series include patients with structural lesions that, not surprisingly, show concordant hypometabolism. When patients with abnormal MRI findings are excluded, 20% to 50% of the relatively small populations remaining show regional decreases in glucose consumption (
10,
49,
78,
79,
80,
81). Some investigators have found a good correlation between regional hypometabolism and the epileptogenic zone; others have found a reasonable correlation with side, but not site, of ictal origin.
[
11C]FMZ-PET studies yield mixed and inconsistent results (
10,
82,
83). [
11C]FMZ binding may be reduced and is more restricted in cortical extent than are [
18F]FDG-PET abnormalities, when present; appears to correlate with the site of ictal activity; and, if resected, is associated with improved outcome (
83,
84,
85,
86). Patients with acquired lesions may have regional focal reductions in [
11C]FMZ binding concordant with the lesion but most marked at the margins (
82,
85). In other studies, two thirds of patients with neocortical epilepsy and normal MRI had [
11C]FMZ abnormalities, either increased or decreased, that were bilateral in half the subjects (
82,
87). Techniques that correct for gray-matter volume averaging may help in identifying abnormal [
11C]FMZ binding in cortical dysplasia or ectopic neurons in white matter, as well as in avoiding false-positive interpretations (
87,
88,
89,
90). In view of these mixed findings, the role of [
11C]FMZ in nonlesional epilepsy remains unclear. In patients with extratemporal lobe partial epilepsy, ictal SPECT may be a better identifier of epileptogenic cortex (see below).