Chapter 78 Models of Insomnia
Up until the late 1990s there were only two models regarding the etiology and pathophysiology of insomnia. The relative lack of theoretical perspectives was due to at least three factors. First, the widespread conceptualization of insomnia as owing directly to hyperarousal may have made it appear that further explanation was not necessary. Second, the long-time characterization of insomnia as a symptom carried with it the clear implication that insomnia was not itself worth modeling as a disorder or disease state. Third, for those inclined toward theory, the acceptance of the behavioral models (i.e., the 3P behavioral model and the stimulus control model1,2), and the treatments that were derived from them, might have had the untoward effect of discouraging the development of alternative or elaborative models.
Since the 1990s there has been a proliferation of theoretical perspectives on the etiology and pathophysiology of insomnia that includes ten human models* and three animal models. In this chapter, six models (Box 78-1) are described and critiqued: the classic 3P behavioral model,1 the stimulus control model,2 and four models that are arguably the most influential of the modern perspectives†: the neurocognitive model,3 the psychobiological inhibition model,4 the Drosophila model,5,6 and the cage exchange model.7
Box 78-1 Potential Implications for Treatment of Insomnia
Spielman Model
One implication for treatment is that sleep compression need not occur in a radical fashion, but could be accomplished over days or weeks.19
The Stimulus Control Model
Basic Description
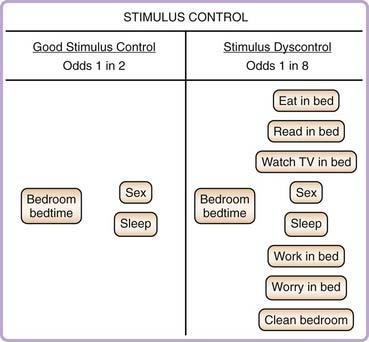
Stimulus control, as originally described by Bootzin,2 is based on the behavioral principle that one stimulus may elicit a variety of responses, depending on the conditioning history. A simple conditioning history, wherein a stimulus is always paired with a single behavior, yields a high probability that the stimulus will yield only one response. A complex conditioning history, wherein a stimulus is paired with a variety of behaviors, yields a low probability that the stimulus will yield only one response. In persons with insomnia, the normal cues associated with sleep (e.g., bed, bedroom, bedtime, etc.) are often paired with activities other than sleep. For instance, in an effort to cope with insomnia, the patient might spend a large amount of time in the bed and bedroom awake and engaging in activities other than sleep. The coping behavior appears to the patient to be both reasonable (e.g., staying in bed at least permits the patients to rest) and reasonably successful (engaging in alternative activities in the bedroom sometimes appears to result in cessation of the insomnia). These practices, however, set the stage for stimulus dyscontrol, the lowered probability that sleep-related stimuli will elicit the desired response of sleepiness and sleep. Figure 78-1 provides as schematic representation of stimulus control and stimulus dyscontrol.
Strengths and Weaknesses
The treatment that is derived from stimulus control theory is one of the most widely used behavioral treatments, and its efficacy has been well established.8–12 The success of the therapy, however, is not sufficient evidence to say that stimulus dyscontrol is the factor, or one of the factors, responsible for predisposition to, the precipitation of, or the perpetuation of insomnia.* This is the case because the therapy includes active components that are not based solely on learning or behavioral theory. For instance, the treatment specifies that the patient should spend awake time somewhere other than the bed and that the sleep schedule should be fixed. These two interventions also influence the homeostatic and circadian regulation of sleep. Thus, the efficacy of stimulus control therapy does not necessarily provide evidence for the stimulus control model. In fact, one investigation found that the reverse of stimulus control instructions also improved sleep continuity.13
Implications for Current and Future Research and Therapeutics
Given the efficacy of stimulus control therapy, as it is classically rendered, it would be useful to determine how much treatment outcome from cognitive behavior therapy (CBT) owes to the manipulation of this factor. One way to assess the relative importance of stimulus control would be as part of a dismantling study. To date no such study has been conducted as a single, large-scale, randomized trial.* Alternatively, experimental studies could be used to determine which, if any, specific stimuli are most associated with sleep continuity disturbance and whether alteration of these stimuli produces enhanced clinical gains.
The 3P Model
The 3P behavioral model,1 also known as the Spielman model, the three-factor model, or the behavioral model is the first fully articulated model of insomnia to gain widespread acceptance. The model delineates how insomnia occurs acutely and how acute insomnia becomes chronic and self-perpetuating. The model is based on the interaction of three factors. The first two factors (the predisposing and precipitating factors) represent a stress-diathesis conceptualization of how insomnia comes to be expressed. The third factor (the perpetuating factor) represents how behavioral considerations modulate chronicity. A schematic representation of this model is presented in Figure 78-2.
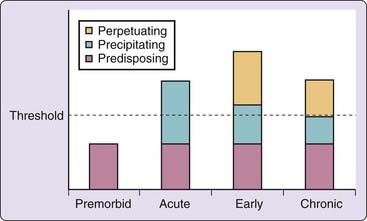
Figure 78-2 The classic 1987 rendition of the 3P model. There is a more recent representation of the model in Chapter 144. The reader is encouraged to compare the two versions of the model. The differences (e.g., allowing the predisposing factors to be represent as variable with time), while subtle, are theoretically important.
Basic Description
Predisposing factors extend across the entire biopsychosocial spectrum. Biological factors are likely to include increased basal metabolic rate, hyperreactivity, and/or fundamental alterations to the neurotransmitter systems associated with sleep and wakefulness.* Psychological factors include worry or the tendency to be excessively ruminative. Social factors, although rarely a focus at the theoretical level, include such things as the bed partner keeping an incompatible sleep schedule or social pressures to sleep according to a nonpreferred sleep schedule (e.g., child rearing).
The classic version of the 3P model focuses primarily on the last of these considerations. Excessive time in bed (or sleep extension) refers to the tendency of patients with insomnia to go to bed earlier or to get out of bed later or to engage in napping. The patient enacts such changes (compensatory activities) to increase the opportunity to get more sleep; these changes are likely highly self-reinforcing (in the short term) because they allow lost sleep to be “recovered” and the daytime effects of lost sleep to be ameliorated. The tendency toward sleep extension is, in the long term, problematic. Sleep extension leads to a mismatch between sleep opportunity and sleep ability.1,14 The greater the mismatch, the more likely the person will spend prolonged periods wake during the given sleep period, and that this will occur regardless of what predisposed the individual to the insomnia and precipitated it.
Strengths and Weaknesses
The greatest strengths of the 3P model is that the therapy based on the theory (sleep restriction) is conceptually appealing to sleep medicine clinicians and scientists, the model is highly face valid for patients (especially when it is delivered as part of therapy), and the therapy itself (which is also compatible with, and a logical clinical application of, the two-process model of normal sleep15) appears to be very efficacious. The equivocation regarding efficacy represents one of the models weaknesses.
There have been very few studies evaluating sleep restriction therapy as a monotherapy,8,9 and no studies evaluating the relative efficacy of sleep restriction therapy (using dismantling designs) as component of CBT. It is therefore difficult to assess the extent to which treatment efficacy supports the 3P model itself. Further, even if there were large-scale studies showing that sleep-restriction therapy produces large effects, the validation of the model would still require empirical studies (see later).
In the original model it is implied that the predisposition to insomnia varies across patients but is a trait factor (static over time) within the individual patient. Presumably the postulated between-subject variability means that some patients are not prone to insomnia, some are marginally at risk, and still others are at high risk. Although it stands to reason that the vulnerability for insomnia exists on a continuum (i.e., is normally distributed), it is also plausible that everyone is at risk for acute insomnia and that this is so to the extent that insomnia represents an adaptive response to stress (i.e., real or perceived threat prevents the inhibition of wakefulness; this idea is addressed by the cage exchange model and the psychobiological inhibition model). The postulate of within-subject variability (risk being static over time) also may be open to question. Some predispositions may be indeed be hardwired (addressed by the Drosophila model) but it also stands to reason that some predispositions vary over the lifespan (e.g., new sleep environments or partners, pregnancy or childrearing, altered hormonal status, effects of aging. The newer rendition of the 3P model (reviewed in Chapter 144) reconciles this issue by explicitly allowing predisposing factors to vary with time).16
Implications for Current and Future Research and Therapeutics
Most of the tenets of the 3P model are untested and await empirical demonstrations. Several avenues for research are possible. Family studies or medical anthropology studies could be used to evaluate the predisposition toward insomnia. Stress-induction studies in good sleepers, like those, for example, conducted by Hall and colleagues,17,18 could be use to produce acute insomnia and to evaluate how a variety of biopsychosocial factors mediate the magnitude of the stress response. Longitudinal studies could be used to confirm whether the putative perpetuating factor of sleep extension does indeed mediate the transition from acute to chronic insomnia.
As for therapeutics, the 3P model has served as the conceptual basis for one treatment modality in particular: sleep restriction. This therapy, while believed by many to be the single most potent component of CBT, was developed to target one particular factor (of the three) and only as it is expressed in one particular form (i.e., sleep extension). This may explain the overall value of multicomponent CBT in that the other treatment components, it can be argued, address other perpetuating factors (e.g., stimulus control addresses engaging in nonsleep activities in the bedroom, cognitive therapy addresses the problem of catastrophic or dysfunctional thinking about insomnia, sleep hygiene addresses the misuse of counterfatigue measures). Thus, the question at hand is: In what ways might the 3P model lend itself to identifying alternative treatment targets with standard or alternative methods?19 One possibility is to develop therapies or adapt existing therapies to target predisposing factors. Such treatments could be used to increase treatment response, decrease the risk for reoccurrence (as an adjuvant to traditional CBT), or prevent first episodes of insomnia.
The Neurocognitive Model
Basic Description
The neurocognitive model3 is based on, and is an extension of, the 3P behavioral model as described by Spielman and colleagues.1 The central tenets of the model include:
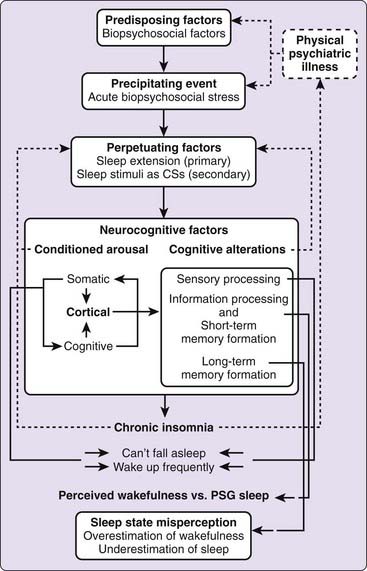
As with the “3P” behavioral model of insomnia, it is posited that acute insomnia occurs in association with predisposing and precipitating factors and that chronic insomnia occurs in association with perpetuating factors.1 The primary perpetuating factor is a form of instrumental conditioning that occurs with sleep extension. The neurocognitive model posits that classical conditioning can also serve as perpetuating factor for chronic insomnia and stipulates that hyperarousal needs to be construed and assessed in terms of its component domains: cognitive, somatic, and cortical arousal. With these considerations in mind, it is suggested that repeated pairing of sleep-related stimuli with insomnia-related wakefulness (arousal) ultimately causes sleep-related stimuli to elicit (or maintain) higher than usual levels of cortical arousal at around sleep onset or during the sleep period. This form of arousal is not thought to be paralleled by somatic arousal (which is posited to be more characteristic of acute insomnia) and is thought to precede, and act as the biological substrate for and precipitant of, cognitive arousal in the context of chronic insomnia.
Conditioned cortical arousal is hypothesized to be self-reinforcing, and for essentially two reasons. First, because sleep-related stimuli (X) act as conditioned stimuli for cortical arousal (Y), the pairing is self-reinforcing. That is, if X elicits Y, and the occurrence of Y reinforces the association of X and Y, then pairing is self-reinforcing. Second, because cortical arousal permits processes associated with wakefulness, it is likely that the elicited arousal will, on each occasion, be amplified because of ongoing sensory processing, enhanced information processing, and long-term memory formation. Taken together, these considerations virtually guarantee that the insomnia will, in the absence of its original precipitants, continue unabated and will not be subject to extinction, as usually occurs with classical conditioning. See Figure 78-3 for a schematic representation of the model.
Strengths and Weaknesses
Strengths
The indirect evidence derives from observations about the effects of sleep on long-term memory in good sleepers and perceived wakefulness during sleep recorded on a polysomnograph (PSG) in patients with insomnia. With respect to the former, there is good evidence that normal sleepers cannot recall information from periods immediately prior to sleep,20–23 during sleep,24–28 or during brief arousals from sleep.29,30 Thus, normal sleep is indeed characterized by a dense amnesia for events occurring at around sleep onset and during sleep.
With respect to the latter, there is substantial evidence that when awakened from PSG-defined sleep, patients with insomnia (as opposed to good sleepers) tend to perceive themselves to be awake rather than asleep.31–38 This tendency, better known as sleep state misperception, is consistent with the neurocognitive model’s perspective regarding sensory and information processing during sleep. That is, if one cue for “knowing” that one is asleep is the lack of awareness for events occurring during sleep, and if it is the case that patients with insomnia exhibit increased levels of sensory and information processing during sleep, then it would be expected that the greater level of awareness for events occurring during PSG-defined sleep serves to blur the phenomenologic distinction between sleep and wakefulness so that patients with insomnia would have difficulty indentifying PSG sleep as sleep. In this instance, what remains open to question is whether sleep state misperception can be correlated with objective measures of cortical arousal—such as by quantitative electroencephalography (qEEG), analyses of cyclic alternating pattern (CAP), or brain metabolic functional imaging—or with objective measures of increased sensory and information processing during sleep (i.e., via evoked-response potentials [ERPs]).
The direct evidence pertains to whether patients with insomnia exhibit increased cortical or central nervous system (CNS) arousal as measured by qEEG and positron emission tomography (PET), increased sensory or information processing as measured by ERPs, an attenuation in the normal mesograde amnesia of sleep, or association between sleep state misperception and objective measures of cortical arousal or ERP abnormalities. Patients with primary insomnia have been found to exhibit higher levels of cortical arousal (in terms of increased NREM high-frequency EEG) as compared to good sleepers39–43 or patients with insomnia comorbid with major depression.43a Cortical arousal (as well as increased activity involving subcortical areas and circuits) has also been observed in patients with insomnia using PET techniques.44,45 Altered sensory and information processing have been observed with ERPs.46–47 Correlational analyses provide evidence that that beta and gamma activity is negatively associated with the perception of sleep quality17,48 and is positively associated with the degree of subjective-objective discrepancy.43 There is some evidence that patients with sleep state misperception disorder (paradoxical insomnia) have been found to exhibit more beta and gamma EEG activity than good sleepers or patients with primary insomnia.49 One study shows that patients with insomnia are better able to recognize word stimuli played during sleep-onset intervals and during early NREM sleep. This latter finding provides support for the hypothesis that there is an attenuation in the normal mesograde amnesia that accompanies sleep in patients with chronic insomnia.
Implications for Current and Future Research and Therapeutics
The neurocognitive model may provide some insight into the potential mechanisms of action of existing therapeutics and also some guidance regarding potential targets for new treatments. In the case of existing therapeutics, pharmacotherapy might effective to the extent that the various compounds block sensory and information processing or promote amnesia within the sleep period. This idea, first espoused by Mendelson,50–55 seems probable given the effects of benzodiazepines and benzodiazepine receptor agonists on arousal thresholds and memory formation. Sleep restriction therapy might also work via these mechanisms to the extent that this treatment modality serves to deepen sleep (augment the endogenous form of sleep-related mesograde amnesia).
Potential avenues for new medical treatments include the assessment of compounds that have greater-than-normal amnestic potential for their efficacy as hypnotics, provided that such effects can be limited to the desired sleep period and the use of diurnal stimulant therapy to promote wake extension and thereby their potential to diminish nocturnal cortical arousal via increased sleep pressure. Potential avenues for behavioral treatment include inpatient protocols that use more-intensive forms of sleep restriction therapy to promote counterconditioning, such as what is now being done with intensive sleep retraining therapy.56
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree