64 What was the first surgical procedure employed for Parkinson’s disease? Who performed it, and when? Partial caudate resection. This first ablative surgery for Parkinson’s disease was first reported by R. Meyers in 1942. When and by whom was stereotaxy introduced? In 1947, Spiegel and Wycis introduced human stereotaxy, demonstrating a reproducible technique for navigating to neural target structures. When was stereotaxy first shown to be effective in the treatment of tremor? In 1954, Hassler described lesioning of the ventral intermediate (Vim) nucleus of the thalamus for the treatment of Parkinsonian tremor. What event temporarily curtailed the continued adoption of stereotactic surgery for movement disorders? By whom and when did this occur? In 1968, Cotzias reported the clinical effectiveness of levodopa therapy. When and why did stereotactic neurosurgical procedures reemerge for the treatment of movement disorders? In the 1990s, stereotactic procedures were revisited for the treatment of complications from levodopa therapy. What is the origin of current deep brain stimulation (DBS) therapy? As reported in 1960 by Hassler, neurostimulation was found to reduce the magnitude of tremor in Parkinson’s disease. When and by whom was a permanent DBS system (implantable pulse generator and electrode array) implanted? In 1993, Alim Benabid was the first to report permanent implantation of a DBS system for the treatment of Parkinson’s disease. He initially implanted electrodes in the Vim nucleus of the thalamus, followed by studies involving implantation in the globus pallidus internus (GPi), followed by implantation in the subthalamic nucleus (STN). What are the advantages of DBS over ablative therapy? 1. The procedure is reversible; therefore, if the treatment or target is ineffective or it becomes desirable to reverse the treatment, this is possible by either simply turning the stimulation off or removing the IPG and/or electrode. 2. The procedure is nondestructive; therefore, no neural structures are sacrificed or compromised in the placement of the system. 3. The procedure is adjustable; therefore, the magnitude and other parameters governing the treatment may be varied to achieve successful or optimal treatment results and may be similarly adjusted to minimize side effects. What are the clinical features of Parkinson’s disease (PD)? Motor: four cardinal motor symptoms: 1. Tremor at rest (disappears with movement and sleep, typically of 4 to 6 Hz, i.e., 4 to 6 times per second) 2. Rigidity: increased muscle tone and joint stiffness produce “cogwheel rigidity” observed upon passive movement 3. Bradykinesia: slowness in the planning, initiation, and execution of movement (the most disabling early feature) 4. Postural instability: present later in the disease and causing frequent falls Other clinical features: 5. Autonomic failure: may be the presenting sign of PD or may be part of multiple system atrophy (MSA); includes orthostatic hypotension, sweating dysfunction, sphincter dysfunction, and erectile dysfunction 6. Cognitive decline: found in 84% of PD patients 7. Dementia: found in 48% of PD patients 15 years after diagnosis 8. REM sleep disorder: characterized by an increase in violent dream content, is considered a pre-parkinsonian state 9. Insomnia: typically sleep fragmentation (>50% prevalence) 10. Sensory abnormalities: hyposmia (olfactory dysfunction) confers a 10% increased risk for PD diagnosis in 2 years 11. Depression 12. Constipation1 What are the distinctions between PD and essential tremor (ET) with respect to clinical features?
Movement Disorders
64.1 History of Deep Brain Stimulation in Parkinson’s Disease
64.2 Diagnosis and Medical Management
Feature | PD | ET |
Age at onset | 55–75 | 10–80 |
Family Hx | +/− | ++ |
Tremor frequency | 4–6 Hz | 5–10 Hz |
Tremor movement | Supination/pronation | Flexion/extension |
Tremor modulation |
|
|
Rest | Increases | Decreases |
Movement | Decreases | Increases |
Ethanol | No effect | Decreases |
Limb tremor symmetry | Asymmetric | Symmetric |
Nonlimb functions involved | Face, jaw, lips, chin | Head, voice1 |
What are the classes and unique agents comprising medical treatments for PD?
• Anticholinergics
• Amantadine
• Dopaminergic drugs1
What are the classes and unique agents comprising medical treatments for ET?
• Alcohol
• Beta-blockers
• Primidone
• Topiramate
• Gabapentin
• Botulinum toxin1
64.3 Neuromodulation
What is the neuromodulation?
Neuromodulation is the altering of control of the activity of a portion of the nervous system, including central, peripheral, and autonomic
What are the advantages of neuromodulation over alternative therapeutic approaches?
Neuromodulation is reversible, adjustable, and nondestructive. It enables the provision of therapy, which may be customized, changed over time, and removed if desired, allowing an optimal balance of therapy with side effects.
Which technologies are being used to accomplish neuromodulation?
Electrical stimulation using implanted electrodes (deep brain stimulation [DBS], vagus nerve stimulation [VNS], spinal cord stimulation [SCS], peripheral nerve stimulation [PNS]), electrical stimulation using noninvasive coils (transcranial magnetic stimulation [TMS]), and pharmacotherapy (intrathecal drug pumps, e.g., baclofen, morphine, etc.)
For which indications has neuromodulation been approved by the FDA for clinical use?
Chronic pain, Parkinson’s disease, essential tremor, dystonia, epilepsy
64.4 Targets and Pathways
What are the four subcortical nuclei comprising the basal ganglia?
1. Striatum (caudate and putamen)
2. Globus pallidus (internal and external segments, GPi and GPe, respectively)
3. Subthalamic nucleus (STN)
4. Substantia nigra (pars compacta and pars reticulata, SNc and SNr, respectively)
What is the input structure within the basal ganglia circuitry?
Striatum (caudate and putamen)
What structures project to this basal ganglia input structure? With what neurotransmitters?
1. Cortex → excitatory glutamatergic (Glut) projections
2. Substantia nigra pars compacta (SNc) → dopaminergic (DA) projections
What are the output structures from the basal ganglia (BG)?
1. GPi → inhibitory GABAergic (GABA) projections
2. SNr → inhibitory GABAergic (GABA) projections
To which structure do motor output pathways from these basal ganglia project?
Ventral lateral thalamus
This structure then projects to what structures?
1. Cortex
2. Pedunculopontine nucleus (PPN) (→ on to the spinal cord)
The direct pathway involves projections originating where? Are they excitatory or inhibitory?
The direct pathway comprises direct inhibitory projections from the striatum to the GPi/STN.
What are stereotactic target coordinates for the STN?
Correct positioning of the DBS electrode tip in the STN (center of the motor territory of the STN) is as follows:
Target coordinates:
x-axis coordinate: 12 mm lateral to midline
y-axis coordinate: 3 mm posterior to midcommissural point
z-axis coordinate: 4 mm inferior to midcommissural point
Intraoperative test stimulation parameters for STN:
Frequency = 185 Hz
Pulse width: 60 ms
Voltage: gradually increased to effect (1- to 10-V range)
High voltage stimulation (6–10 V) → corticobulbar fibers; produces side effects:
• Dysarthria
• Facial contractions
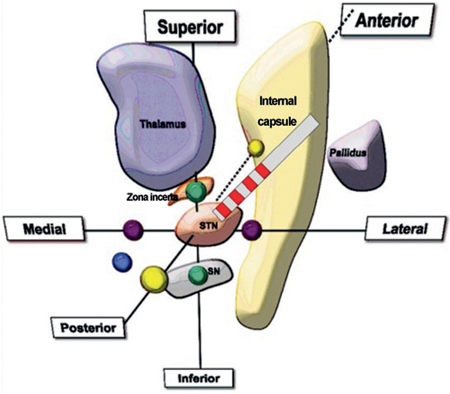
Fig. 64.1 Stereotactic target coordinates for the subthalamic nucleus (STN). SN, substantia nigra.







