Figure 84.1. Example of electrocardiogram.
Hyperglycemic crisis is common acutely after SAH, and are due primarily to catecholamine induced stress. The glucose values typically are in the 200-300 mg/dl range. The severity of hyperglycemia correlates well with the clinical severity of the SAH, with Hunt Hess grades 4-5 having the highest blood glucose levels. The degree of hyperglycemia however is an independent predictor of poor outcome, even when severity of SAH is factored in [8]. Hence, immediate evaluation and regularly scheduled glucose checks are needed. In the acute phase of SAH, with the potential for impending brain ischemia, glucose control is advisable, although the exact thresholds are presently unknown. Moderate glucose control with goals of 100-140 mg/dl seem to be a reasonable goal, in the absence of data suggesting otherwise.
Intracranial pressure (ICP) crisis related to acute hydrocephalus is a serious concern after SAH. Patients may develop hydrocephalus resulting in elevation of ICP to values ranging from 30-60 mmHg. Diagnosis is frequently made by computerized tomographic imaging. An example of this is shown in Figure 84.2. Placement of an external ventriculostomy catheter is frequently needed, and should be done before exposing the patient to the supine position for angiography and embolization.
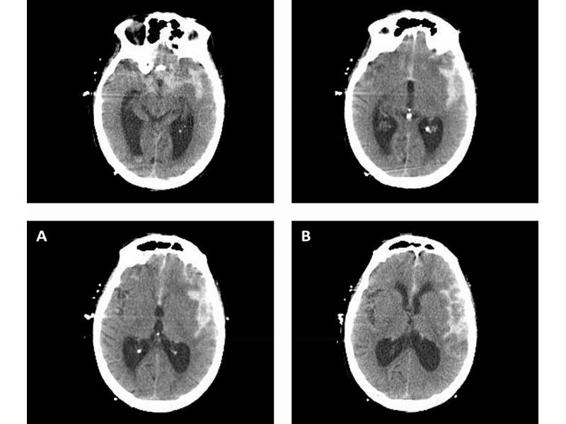
Figure 84.2. Example of computerized tomographic imaging. Progressive wavelet level: 3-4 (A). Progressive wavelet level: full (B).
Close attention to fluid balance, with a goal of early euvolemia, is necessary. This can be facilitated by placement of a radial arterial line and a central venous catheter into the subclavian or the internal jugular vein. Measurement of blood pressure and right atrial (RA) pressure allow for prompt control of hypertension and for management of fluid status. These patients often require control of systemic hypertension and volume resuscitation. Before the aneurysm is secured, the mean arterial blood pressure is ideally kept at the lower range of blood pressure autoregulation (e.g., 80 to 90 mmHg), and the RA pressure at 5 to 10 mmHg. Agents that are commonly used include intermittent bolus doses of labetalol (10 to 20 mg every 1 hour) or other agents such as nicardipine or hydralazine. Antiadrenergic compounds are usually the most successful in treating the catecholamine-induced increase in blood pressure. If this does not work, then continuous infusions of esmolol or nitroprusside may be needed to control blood pressure. One must consider that reduction in blood pressure may also reduce cerebral perfusion pressure and thereby induce brain ischemia. Thus, if uncertain about ICP, clinicians should maintain the MAP above 90 mmHg to avoid ischemia. Preventing acute kidney injury is also an important consideration, given that patients are exposed to radiologic contrast agents. While there is no consensus about how best to prevent acute kidney injury, several options have been demonstrated to be of benefit [14]. Pre-angiography hydration and the minimum dose of intravenous contrast are the two most critical elements to prevent acute kidney injury during angiography. The use of N-acetylcysteine, sodium bicarbonate infusions, and acetozolamide have been advocated by many to be useful. In a randomized controlled trial, Merten et al. demonstrated that the use of sodium bicarbonate in the maintenance hydration fluid resulted in less contrast-related acute kidney injury.
84.3 Critical Care During the Endovascular Procedure
Until recently, the critical care of patients during the endovascular therapy has not received much attention. Important considerations during the procedure include adequate airway protection and oxygenation, sedation, blood pressure management, and management of ICP. Airway protection may be attained through the use of elective endotracheal intubation. If the patient is somewhat somnolent or has a preoperative Hunt and Hess grade of 3 or worse, then endotracheal intubation is indicated. Oxygenation and airway protection are further facilitated by adequate sedation. Sedation that can be easily adjusted and is rapidly metabolized is most useful and permits the interventional team to examine the patient intermittently if needed. Agents that fullfill these requirements include short-acting benzodiazepines, such as midazolam and propofol. Additionally short-acting inhalational agents may be used. A caveat that should be kept carefully in mind is that all of these agents may reduce blood pressure and consequently reduce cerebral perfusion pressure if autoregulation is impaired, which is often the case. Thus, close monitoring of blood pressure and supplemental use of vasoactive agents may be necessary to avoid adverse reductions of blood pressure.
Similar concerns of hemodynamic support and adverse cardiac responses to sedatives and anesthetics should be kept in mind during the intervention. Cardiac arrhythmias are well known to occur early after SAH, and they require prompt attention. Below, we consider several possible cardiopulmonary complications that may first arise during interventional treatment. A minimum of cardiac and central venous pressure monitoring has been our standard for making prompt diagnoses of such difficulties. Airway and respiratory support, with maintenance of the airway and normal carbon dioxide through endotracheal intubation and mechanical ventilation is critical.
Measurement of ICP and cerebral perfusion pressure is an important aspect of critical care during the endovascular treatment. A ventriculostomy permits drainage of CSF and subarachnoid blood, and it is the preferred method for acute management of changes in ICP, without the use of hyperventilation or diuretics (i.e., mannitol) [15]. These latter two methods of reducing ICP have adverse side effects, such as brain ischemia from hyperventilation or hypovolemia from mannitol-induced diuresis. These are best avoided in the acute phases of SAH. ICP should be maintained in a normal range of 15 to 20 mmHg. This range should permit adequate perfusion pressures while the risk of aneurysm rerupture is avoided. The latter may occur owing to unintentional overdrain-age of CSF or sudden changes in ICP. Thus, with use of the ventriculostomy, the drainage point should be set to a water level between 15 and 20 cm so that sudden declines in ICP are avoided. In addition to monitoring the acute angiogram and embolization treatment, the clinicians should monitor ICP during papaverine infusion and angioplasty for vasospasm, because they both have been associated with increased cerebral blood volume and increases in ICP. Finally, monitoring ICP can provide guidance as to the adequacy of sedation and blood pressure in the setting of a comatose patient who is unable to be adequately examined.
84.4 Post-procedural Neurocritical Care
84.4.1 Treating Medical Complications
Recently, Solenski and colleagues [16] have shown that medical complications after SAH are responsible for additional morbidity and mortality (approaching 40%), nearly equal to the primary neurologic morbidity. The attributable death rate caused by medical complications is approximately 23%. Thus, early assessment of arterial blood gases, chest radiograms and airway stability, and continuous pulse oximetry are required for the appropriate treatment of these patients. Indeed anticipation of full ventilator support during the period of aggressive intravascular volume therapy should be anticipated and the family and medical consultants informed and educated about this issue.
Acute pulmonary edema and hypoxemia are nearly universal in SAH [17]. This is because of neurogenic pulmonary edema and is not to be related to hypertensive, hypervolemic, and hemodilutional (HHH) therapy or fluid overload. Initial widened alveolar-arterial PO2 gradients, pulmonary edema, and respiratory distress occur acutely in more than 80% of patients and lead to prolonged hospital stays and increased costs. In addition to the acute cardiac insufficiency, there seems to be a neurogenic origin for this hypoxemia that is out of proportion to cardiac failure or fluid overload. The need for aggressive maintenance of intravascular volume in euvolemic or hypervolemic states elicits the subtle pulmonary dysfunction and triggers fulminant pulmonary edema. Treatment of acute pulmonary edema may include the use of dobutamine [18], gentle diuresis, and augmented positive end-expiratory pressure (PEEP).
Early cardiac dysfunction, including diffuse contraction band necrosis and segmental wall motion abnormalities, occurs in a sizable minority of SAH patients. [19] The dysfunction is caused in large part by neurogenic sympathetic overactivity as well as by the surge in systemic catecholamines. Several reports of cardiac arrhythmias in the acute stages of SAH exist. The occurrence of arrhythmias and wall motion abnormalities predisposes the patient to decreased cardiac output and thus reduced cerebral perfusion. This cardiac dysfunction is often reversible but can give rise to early pulmonary and cardiovascular insufficiency. If doubt arises as to the function of the cardiovascular system or the pulmonary function, prompt evaluation with a right heart catheterization or echocardiogram should be entertained.
In addition, evaluation for concurrent myocardial ischemia or pneumonia should be performed. Management of these complications encompasses many common means (i.e., antibiotics) with the caveat that volume depletion and diuresis should be minimized in patients who have primary ischemic injury or in whom vasospasm or reduced cerebral perfusion pressure exist. In the latter situation, a relative compromise in tolerating excessive pulmonary edema may be necessary to ensure adequate perfusion of the brain. Contrary to typical intensive or cardiac care protocols, hydration rather than diuresis is the goal.
Hyponatremia is a frequent complication after SAH. This is likely due to cerebral salt wasting. Replenishment with hypertonic saline and supplemental mineralocorticoid administration, typically with fludrocortisone, is necessary, in order to maintain sodium in the normal range, 138-145 mEq/dl.
Avoid of fever is a very important feature as well. Fever can exacerbate neurologic damage, and has been linked as an independent factor affecting poor neurologic outcome [20,21]. The use of surface cooling devices or intravascular cooling devices has become commonplace. The goal temperature is typically 36-37°C, with strict avoidance of any fever. The use of antishivering protocols is critical to implementing this control and avoiding unnecessary metabolic challenges to the patient [22]. Medications such as buspirone, and meperidine are important adjunctive agents that limit the extent of shivering and thereby facilitate the positive effects of cooling.
84.4.2 Treating Neurologic Complications
Often, obstructive hydrocephalus occurs within the first few hours after SAH. It can be life threatening because of brain-stem compression and occlusion of large as well as smaller penetrating blood vessels. Deterioration of the level of consciousness early after the primary event is the most common presentation. The use of a ventriculostomy for ICP measurement and drainage of CSF should be performed on an individual basis for acute hydrocephalus, based on the severity of clinical neurologic dysfunction and the CT scan appearance of trapped temporal horns or obliterated basilar cisterns. Several reports suggest a beneficial effect of ventriculostomy to relieve hydrocephalus and to improve patient outcome [15,23,24]. The use of ventricular drainage is a usefull early test of possible neurologic viability and chose operative candidates in whom neurologic improvement occurred after CSF drainage. Thus, the ventriculostomy may serve as both a therapeutic device as well as an indicator of which severe-grade patients should be aggressively treated.
Many have found that early drainage or lavage of the CSF space decreases the incidence of vasospasm after SAH [25] and that the infusion of thrombolytic agents may aid in this process with the timing of such intervention usually after the aneurysm has been secured. However, sudden drainage of CSF may precipitate rerupture of the aneurysm, mainly because of the transmural pressure across the aneurysm wall exceeding the intraventricular pressure. Thus, caution must be exercised during placement of a ventricular drain and during draining of fluid. One must limit the amount of CSF drained and the equilibrium CSF pressure at which the fluid drains. A practical approach is to drain CSF if the ICP exceeds 20 mmHg until the aneurysm is occluded. The drain is set at a 15 to 20 cm height such that the intraventricular pressure does not drop below this value during the draining process. Thus, one may avoid excessive ICP or dangerous hydrocephalus while avoiding unintentional aneurysm rerupture.
Rehemorrhage after the primary event is the most worrisome initial neurologic complication. Kassell and co-workers [9,11] have shown that early operative management decreases the risk of rehemorrhage and improves outcome. In most centers, early clipping of the aneurysm or endovascular treatment occurs within 24 to 48 hours after rupture. Subsequent intensive care unit care then makes use of neurologic monitoring, especially the neurologic examination, ICP monitoring, transcranial Doppler ultrasound, and continuous EEG [6,26]. These techniques are used in a complementary fashion to detect and treat vasospasm and other neurologic complications such as seizures. Refinements in the application of these techniques and the use of hypervolemic-hypertensive-hemodilutional therapy offer the hope of decreasing the morbidity from vasospasm and improving the outcome [6].
In summary, the prompt recognition of SAH and the acute management of neurologic and medical problems is necessary to prevent additional morbidity and to maximize the neurologic recovery of patients suffering from SAH.
84.4.3 The Use of Neurologic Monitoring
One of the hallmarks for neurocritical care is the use of specific neurologic biomarker monitoring instruments to detect delayed cerebral ischemia, seizures, and other forms of neurologic deterioration [6,26]. Continuous EEG monitoring has the potential to detect vasospasm-related cerebral ischemia [1,6]. Several quantitative parameters, namely the percent alpha variability (PAV) and the alpha/delta ratio are sensitive indicators of cerebral ischemia. A typical example of the serial changes in cEEG PAV are seen in Figure 84.3. PAV detects vasospasm before more conventional measures, such as the transcranial Doppler ultrasound, CT angiography, or the clinical exam. In our hands, we use PAV as an early indicator of cerebral ischemia, and will induce hypertension while awaiting confirmatory testing, even if the neurologic exam is unchanged.
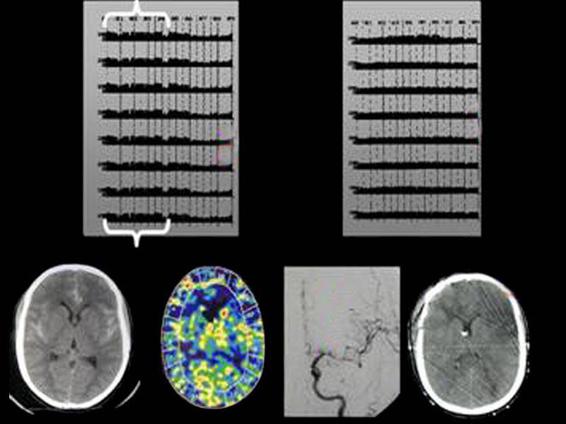
Figure 84.3. A typical example of the serial changes in continuos EEG PAV.
Cerebral microdialysis is another important neurologic monitor for the detection of vasospasm-related cerebral ischemia. The principal findings on microdialysis with ischemia are: 1) a reduction in brain glucose and 2) an increase in the lactate/pyruvate ratio. Several groups have demonstrated utility in using cerebral microdialysis for this type of monitoring.
Similarly brain tissue oxygen monitoring (PbtO2) is a regional tissue monitor that indicates the balance of oxygen delivery and oxygen consumption in the brain. This monitor is capable of detecting reductions in the combination of cerebral blood flow and oxygen content [27].
84.5 Conclusions
The acute management of aneurysmal SAH requires a comprehensive approach entailing acute critical care and stabilization, occlusion of the aneurysm, and intensive care management of acute neurologic complications. The development of endovascular treatment of aneurysms has added an important, less-invasive treatment to the regimen available in treating aneurysms. An integrated approach of providing critical care before, during, and after the occlusion of the aneurysm is an important concept that should be the goal. Debate and controversy remain regarding which aneurysms are best suited to endovascular treatment or to surgical treatment; an evolving experience and clinical trials will provide further guidance. Nonetheless, some aneurysms may require both surgery and endovascular treatment [19]. Finally, close neurologic observation and directed specialized monitoring techniques are required for providing directed critical care in the acute period after SAH.
References
1. Claassen J, Hirsch LJ, Frontera JA, et al. Prognostic significance of continuous EEG monitoring in patients with poor-grade subarachnoid hemorrhage. Neurocrit Care 2006; 4: 103-12
2. LeBlanc R. The minor leak preceding subarachnoid hemorrhage. J Neurosurg 1987; 66: 35-9
3. Locksley HB. Natural history of subarachnoid hemorrhage, intracranial aneurysms and arteriovenous malformation: based on 6368 cases in the cooperative study. J Neurosurg 1966; 25: 219-39
4. Waga S, Ohtsubo K, Handa H. Warning signs in intracranial aneurysms. Surg Neurol 1975; 3: 15-20
5. Pubic Health Service: Detailed Diagnoses and Procedures, National Hospital Discharge Survery, 1990. Hyattsville, MD, US Department of Health and Human Services. DHHS publication PHS 92-1774. Series 13, 1992
6. Vespa PM. The three tenors of subarachnoid hemorrhage: fever, anemia, and hyperglycemia. Crit Care Med 2006; 34: 897-8
7. Mayberg MR, Batjer HH, Dacey R, et al. Guidelines for the management of aneurysmal subarachnoid hemorrhages. Circulation 1994; 90: 2592-605
8. Wartenberg KE, Schmidt JM, Claassen J, et al. Impact of medical complications on outcome after subarachnoid hemorrhage. Crit Care Med 2006; 34: 617-23
9. Kassell NF, Torner JC. Aneurysmal rebleeding: A preliminary report from the Cooperative Aneurysm Study. Neurosurgery 1983; 13: 479-81
10. Kassell NF, Torner JC, Haley EC. The International Cooperative Study on the Timing of Aneurysm Surgery I: Overall management results. J Neurosurg 1990; 73: 37-47
11. Kassel NF, Torner JC, Jane JA, et al. The International Cooperative Study on the Timing of Aneurysm Surgery. Part 2: Surgical results. J Neurosurg 1990; 73: 37-47
12. Diringer MN. Neuroendocrine regulation of sodium and volume following subarachnoid hemorrhage. Clin Neuropharmacol 1995; 18: 114-26
13. Vinuela F, Duckwiler G, Mawad M. Guglielmi detachable coil embolization of acute intracranial aneurysms: Perioperative anatomical and clinical outcome in 403 patients. J Neurosurg 1997; 86: 475-82
14. Ozcan EE, Guneri S, Akdeniz B, et al. Sodium bicarbonate, N-acetylcysteine, and saline for prevention of radiocontrast-induced nephropathy. A comparison of 3 regimens for protecting contrast-induced nephropathy in patients undergoing coronary procedures. A single-center prospective controlled trial. Am Heart J 2007; 154: 539-44
15. Kawai K, Nagashima H, Narita K, et al. Efficacy and risk of ventricular drainage in cases of grade V subarachnoid hemorrhage. Neurol Res 1997; 19: 649-53
16. Solenski NJ, Haley EC, Kassell NF, et al. Medical complications of aneurysmal subarachnoid hemorrhage: A report of the multicenter, cooperative aneurysm study. Participants of the Multicenter Cooperative Aneurysm Study. Crit Care Med 1995; 23: 1007-17
17. Vespa PM, Bleck TP, Brock DG, et al. Impaired oxygenation after acute subarachnoid hemorrhage. Neurology 1994; 43: A344
18. Knudsen F, Jensen HP, Petersen PL. Neurogenic pulmonary edema: treatment with dobutamine. Neurosurgery 1991; 29: 269-70
19. Fernandez A, Schmidt JM, Claassen J, et al. Fever after subarachnoid hemorrhage: risk factors and impact on outcome. Neurology 2007; 68: 1013-9
20. Jacob WA, Van Bogaert A, DeGroot-Lasseel MHA. Myocardial ultrastructural and hemodynamic reactions during experimental subarachnoid hemorrhage. J Mol Cell Cardiol 1972; 4: 287-98
21. Naidech AM, Bendok BR, Bernstein RA, et al. Fever burden and functional recovery after subarachnoid hemorrhage. Neurosurgery 2008; 63: 212-7
22. Badjatia N, Strongilis E, Gordon E, et al. Metabolic impact of shivering during therapeutic temperature modulation: the Bedside Shivering Assessment Scale. Stroke 2008; 39: 3242-7
23. Rosenthal G, Hemphill JC 3rd, Sorani M, et al. Brain tissue oxygen tension is more indicative of oxygen diffusion than oxygen delivery and metabolism in patients with traumatic brain injury. Crit Care Med 2008; 36: 1917-24
24. Suarez-Rivera O. Acute hydrocephalus after subarachnoid hemorrhage. Surg Neurol 1998; 49: 563-5
25. Zambraski JM, Spetzler RF, Lee KS, et al. Phase I trial of tissue plasminogen activator for the prevention of vasospasm in patients with aneurysmal subarachnoid hemorrhage. J Neurosurg 1991; 75: 189-96
26. Jordan KG. Continuous EEG and evoked potential monitoring in the neuroscience intensive care unit. J Clin Neurophysiol 1993; 10: 445
27. Shanlin RJ, Sole MJ, Rahimifar M, et al. Increased intracranial pressure elicits hypertension, increased sympathetic activity, electrocardiographic abnormalities and myocardial damage in rats. J Am Coll Cardiol 1988; 12: 727-36
General References
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree






