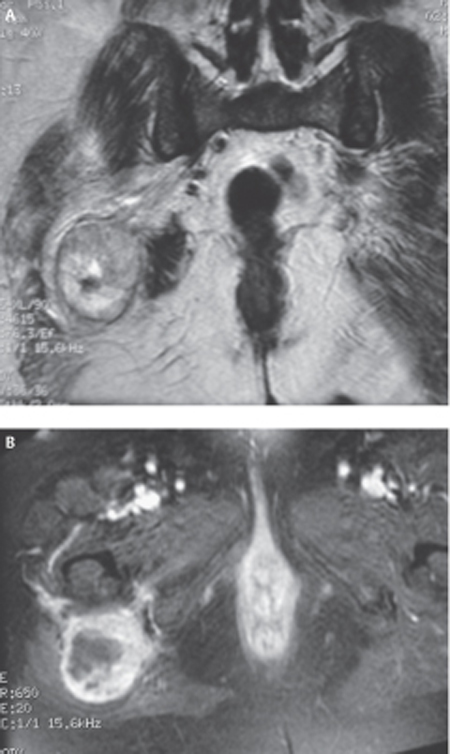
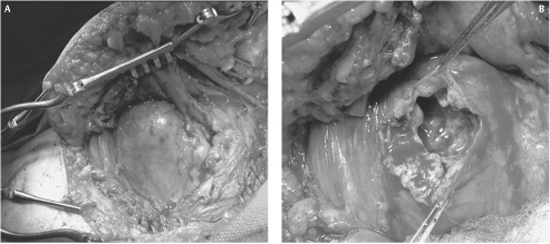
53 Neurogenic Sarcoma of the Sciatic Nerve A 62-year-old female presented with an 11-month history of progressively worsening right foot and leg symptoms. She had a preexisting diagnosis of neurofibromatosis type 1 (NF1) with multiple peripheral neurofibromas; however, to her knowledge, she did not have a lesion affecting one of the larger peripheral nerves, spinal cord, or brain. Her mother and one of her children were also diagnosed with NF1. Initially, the pain was described as a sharp, electric-shock sensation involving the right metatarsal head, plantar fascia, and pulp of the second and third toes. The pain was aggravated by walking but still present at rest. Amitriptyline, Neurontin (Pfizer, Inc., New York, NY), and Dilaudid (Abbott Laboratories, Abbott Park, IL) were prescribed with little benefit. Subsequently, two nerve blocks were attempted over the right lateral ankle and right medial ankle in the area of the origin of the plantar arch with only transient relief of pain. Over the next few months, her symptoms progressed and she required a cane for ambulation. She complained that the pain was now constant, the bottom of her foot was numb, and her right leg weakening; in particular, she was dragging her right foot. Figure 53–1 (A) Coronal T2- and (B) T1-weighted, gadolinium-enhanced magnetic resonance images of the patient’s right buttock lesion show regions of signal inhomogeneity and variable enhancement. On physical examination, right foot dorsiflexion was graded as 2/5, plantar flexion was 4/5, knee flexion was 4/5, with the remainder of her motor power examination normal. Tone in her right foot was diminished with an absent right ankle jerk. On sensory examination, she had hyperesthesia to pinprick over the right heel and medial sole, particularly in the pulp space of the second and third toes. No obvious mass was palpated deep to her plantar arch. Straight leg raising was negative with full range of motion documented in her back and hips. Importantly, however, a Tinel sign could be elicited by palpation and percussion over her right buttock, even though no mass lesion was appreciated. A computed tomographic (CT) scan of her lumbar spine showed no evidence of disk herniation or compromise of the neural foramina or spinal canal. Electromyography (EMG) demonstrated evidence of active denervation in multiple muscle groups of the right leg, including the tibialis anterior, gastrocnemius, and biceps femoris longus. Magnetic resonance imaging (MRI) of the pelvis confirmed a soft tissue mass in the region of the right sciatic nerve measuring 7 x 6 x 5.5 cm below the level of the right acetabulum posterior to the right proximal femur at the level of the femoral neck and greater trochanter (Fig. 53–1). Preoperative chest x-ray was negative. At the time of surgery, the patient was placed in the prone position and a curvilinear incision fashioned around the lateral aspect of the buttock into the buttock crease and extending down the posterior midline of the proximal thigh. The gluteus maximus and medius muscles were separated from the lateral pelvic brim and mobilized medially to expose the nerve at the sciatic notch. A generous cuff of muscle was left attached to the greater trochanter to facilitate closure. With this exposure, an abnormality was encountered that resembled a large, grapefruit-sized lesion with induration and edema of the surrounding fat, but no obvious tumor infiltration into adjacent soft tissue structures. A nerve stimulator was used to identify an electrically silent area of the tumor capsule so that decompression of the necrotic tumor and intratumoral biopsy at multiple sites could safely be performed without injury to overlying fascicles (Fig. 53–2). The presumptive diagnosis of neurogenic sarcoma of the right sciatic nerve suggested by the rapid clinical progression and imaging features was confirmed by quick-section by an experienced neuropathologist. The tumor capsule was reapproximated and the incision closed without further attempts at tumor resection. Final pathology demonstrated an anaplastic, spindle cell sarcoma. Because of the uniformity of cellular atypia and the absence of a transition between a lower-grade tumor (e.g., neurofibroma) and sarcoma, the tumor was felt to have arisen de novo and not from sarcomatous degeneration of a preexisting neurofibroma. The patient was referred to our specialized musculoskeletal oncology unit for more definitive management. Neurogenic sarcoma of the sciatic nerve The sciatic nerve receives contributions from the anterior and posterior divisions of the L4, L5, S1, and S2 nerve roots, as well as the anterior division of the S3 nerve root. The anterior divisions combine to form the tibial (or medial) division of the sciatic nerve, whereas the posterior divisions constitute the peroneal (or lateral) division of the sciatic nerve. These divisions unite and leave the pelvis as the sciatic nerve via the sciatic notch deep to the gluteus maximus muscle. In the midportion of the buttocks, the sciatic nerve crosses the dorsal surface of the ischium, obturator internus, gemelli, and adductor magnus muscles to enter the thigh between the medial and lateral masses of the hamstring muscles. At a variable level above the popliteal fossa in the thigh, the two divisions of the sciatic nerve branch to form the tibial nerve and common peroneal nerve. These nerves are responsible for all the movements of the foot and toes. Two points deserve special mention. First, branches of the sciatic nerve to the hamstring muscles (semitendinosus, semimembranosus, and part of the adductor magnus muscles) arise from the medial aspect of the sciatic nerve as it courses through the thigh. Only the branch to the short head of the biceps femoris arises laterally (from the peroneal division). Thus dissection of the sciatic nerve in the intramuscular plane is best approached from a posterolateral direction. Second, because the divisions of the sciatic nerve may branch far above the popliteal fossa, injury to the sciatic nerve, even in the proximal thigh, may involve only one division while completely sparing the other. Figure 53–2 (A) Operative photomicrographs showing open dorsal exposure of the sugluteal mass. (B) The lesion had displaced the involved sciatic nerve ventrally and allowed safe biopsy and evacuation of the internal cystic contents via this approach. Sciatic nerve dysfunction in NF1 patients deserves special consideration. Patients with NF1 are predisposed to the development of multiple dermal neurofibromas that are benign and pose no risk of malignant transformation. They typically present as an asymptomatic, palpable mass or a painful subcutaneous lump. However, NF1 patients may also suffer from larger, deeper plexiform neurofibromas, with the medium and large nerves more likely to be affected than small nerves. The buttock, thigh, brachial plexus, and paraspinal regions are the most common sites, and overall the sciatic nerve is the most commonly affected nerve. Plexiform neurofibromas present with sensorimotor deficits and pain in the distribution of a known nerve. These tumors may also occur as isolated lesions in non-NF1 patients but occur in NF1 patients in over 50% of cases. Unlike their benign dermal counterparts, plexiform neurofibromas can undergo sarcomatous degeneration in 3 to 5% of NF1 patients and 1% of non-NF1 patients. Neurogenic sarcomas may also arise de novo. The diagnosis of neurogenic sarcoma requires a high index of suspicion. Therefore, rapid clinical progression of a sciatic neuropathy syndrome with an enlarging buttock or thigh mass in an NF1 patient should alert the clinician to the possibility of neurogenic sarcoma. A systematic approach is required for the evaluation of foot drop or unilateral foot pain. Foot drop occurs from weakness of the anterior tibialis muscle innervated by the deep peroneal nerve (which receives components from the L5, and to a lesser extent, L4 nerve roots). The patient’s history and meticulous physical examination will allow localization of the lesion to the central nervous system (cortical lesion involving the motor strip or spinal cord injury, or spinal cord pyramidal pathway), resulting in so-called spastic foot drop, or peripheral nervous system (L4 or L5 root, lumbosacral plexus, sciatic nerve, common peroneal nerve, or deep peroneal nerve lesion). Rarely, peripheral neuropathy (e.g., Charcot-Marie-Tooth) or nonneurogenic causes (e.g., muscular dystrophy), must also be considered. In the context of NF1, this determination is particularly important because lesions that present with foot drop may arise anywhere along the neuraxis (e.g., cerebral astrocytoma involving the contralateral motor strip, spinal canal neural tumor, or neurofibromas arising from the individual components of the peripheral nervous system). A similar approach is adopted for the patient presenting with plantar foot pain. Pain may arise from injury to local soft tissue structures (muscles, ligaments, and tendons), joint disease, or peripheral vascular disease. Radiculopathy (involving the L4, L5, or S1 roots), peripheral neuropathy (e.g., diabetic neuropathy), and injury to the plantar nerves may also be causative (e.g., trauma, plantar nerve schwannoma or neurofibroma, entrapment of the plantar nerves in the foot, or entrapment of the tibial nerve at the ankle). Injury to the sciatic nerve in the thigh may initially present with isolated foot pain and normal neurological examination and has previously been described in patients with sciatic nerve schwannomas. In the context of the current popularity of the diagnosis “tarsal tunnel syndrome, ” it is important to remember that pain starting at the medial arch, radiating into the plantar foot, may be caused by injury along the entire extent of the tibial division of the sciatic nerve and is not restricted to dysfunction of the nerve at the medial malleolus. In patients that present with foot drop from injury to the peripheral nervous system, localization of the lesion can often be made by a systematic physical examination. The following steps in motor examination are initially taken to help localize the lesion to the nerve roots (L4 and L5) and lumbosacral plexus, sciatic nerve, common peroneal nerve, or deep peroneal nerve. Step 1 Check for function of the obturator nerve (arising from the L2 and L3 nerve roots) that innervates the thigh adductors. In a patient with foot drop, weakness of thigh adduction localizes the lesion to the lumbosacral plexus. Step 2 Check for function of the femoral nerve (arising from the L2, L3, and L4 nerve roots) that innervates the quadriceps femoris and iliopsoas muscles. In a patient with foot drop, weakness of knee extension or hip flexion also localizes the lesion to the lumbosacral plexus. Step 3 Check for function of the superior and inferior gluteal nerves that arise from the coalescence of the posterior divisions of the L4, L5, S1, and S2 nerve roots in the pelvis prior to the formation of the sciatic nerve. Weakness of gluteal muscle groups in a patient with foot drop indicates a very proximal lesion involving the nerve roots, lumbosacral plexus, or sciatic nerve in the pelvis or a large lesion at the sciatic notch. Step 4 Check for function of the branches of the sciatic nerve in the thigh distal to the sciatic notch but proximal to its branching into the tibial nerve and common peroneal nerve at a variable distance above the popliteal fossa. In a patient with foot drop, if the examiner documents weakness of knee flexion (hamstring branch), then the lesion can be localized to the sciatic nerve in the proximal thigh or buttock. Step 5 Check for function of the tibial nerve that mediates foot inversion and plantar flexion. In a patient with foot drop, sparing of these functions may indicate injury to the peroneal nerve distal to the take-off of the tibial nerve in or above the popliteal fossa. Interestingly, the peroneal division of the sciatic nerve appears to be more vulnerable to injury than the tibial division, and it is not unusual to see isolated injury to the peroneal division of the sciatic nerve above the knee. Furthermore, L4 and L5 radiculopathies may also present with apparently normal tibial nerve function; however, weakness of foot inversion is often a subtle sign that can be used to distinguish injury of the common or deep peroneal nerves from L4 and L5 radiculopathies, most commonly secondary to lumbar disk herniation. Other elements of the history and physical examination, as well as ancillary investigations, will often provide clues to the culprit lesion (as discussed later). Step 6 Check for function of the superficial peroneal nerve that innervates peronei muscles and mediates foot eversion. Sparing of foot eversion in a patient with isolated foot drop localizes the lesion to the deep peroneal nerve distal to its take-off from the common peroneal nerve applied to the posterior aspect of the head of the fibula. A common clinical dilemma arises when trying to distinguish foot drop caused by L5 (and less commonly, L4) radiculopathy from a lesion of the sciatic or peroneal nerves. Although clues on the motor examination may be useful, as described earlier, the classic clinical syndrome and remainder of the physical examination are often of paramount importance. First, sciatica is often a common feature of radiculopathy but not of lesions affecting the sciatic nerve itself or its branches. A painless foot drop, or one associated with a nonsciatic pain syndrome, is more likely due to injury of the sciatic nerve or common (and deep) peroneal nerves than radiculopathy. Second, root lesions will cause dermatomal (L4 or L5) sensory changes, whereas lesions affecting the sciatic nerve or its divisions will result in hypo- or hyperesthesia in the classical distributions of the common peroneal nerve (small area over the dorsum of the foot extending laterally over the ankle joint and lower leg) and tibial nerve (plantar surface of the foot and the posteromedial surface of the leg). Importantly, unless the posterior cutaneous nerve of the thigh is simultaneously damaged with the proximal sciatic nerve, there will be no sensory symptoms or signs over the back of the thigh. Furthermore, injury to the sciatic nerve alone cannot result in complete stocking sensory loss below the knee because a portion of the medial calf and foot is always supplied by the femoral nerve (saphenous branch). Third, loss of the ankle jerk results from injury to the tibial division of the sciatic nerve or S1 nerve root. Finally, ancillary maneuvers like the straight-knee leg-raising test (Lasègue sign), bent-knee leg-raising test (Kernig sign), and Tinel sign (elicited by percussion or palpation over the course of the sciatic nerve) are very useful in distinguishing a lesion of the nerve roots and sciatic nerve. It is also important to appreciate any mass lesions along the course of the sciatic nerve or its divisions because this may be a simple clue to the presence of a peripheral nerve tumor. These are usually nonpulsatile, firm, and mobile perpendicular to, but not along the longitudinal axis, of a known peripheral nerve. Lastly, a careful search for the peripheral stigmata of NF1 aids in the diagnosis of a peripheral nerve tumor. EMG is an important adjunct in the evaluation of a patient with foot drop, in particular, when attempting to differentiate between proximal lesions involving the nerve roots or lumbosacral plexus. Loss of motor units and denervation injury in the paraspinal muscles indicate involvement of the dorsal rami that exit nerve roots proximal to the lumbosacral plexus. Therefore, paraspinal muscle involvement on EMG localizes the lesion to the nerve roots. Remember, loss of motor units and denervation injury are only reliable 3 weeks after the onset of symptoms. Despite the utility of EMG in localizing a lesion to the nerve roots, lumbosacral plexus, sciatic nerve, common peroneal nerve, or deep peroneal nerve, it is not recommended as part of the routine preoperative evaluation of a patient with a known peripheral nerve tumor because it does not help in differentiating between different tumor types or grades. CT and MRI are important adjuncts in the evaluation of peripheral nerve tumors; however, like EMG, they cannot reliably distinguish between the common subtypes of peripheral nerve tumors or determine whether a lesion is benign or malignant. Schwannomas are typically hypointense on T1-weighted MRI sequences and hyper-intense on T2-weighted images, enhance homogeneously with administration of contrast, are well circumscribed, and sometimes demonstrate a discrete nerve of origin or exit. In contrast, neurofibromas tend to be fusiform or multinodular in shape, and a distinct nerve of origin or exit is typically not visualized. The diagnosis of NF1 obviously increases the likelihood that a peripheral nerve tumor is a neurofibroma. The MRI characteristics of very rare peripheral nerve tumors, like lipomas and ganglion cysts, are pathognomonic, but unfortunately, this is not the case for the more common schwannomas and neurofi bromas. Although CT and MRI cannot differentiate between benign and malignant tumors, nonhomogeneous enhancement, suggestive of intratumoral hemorrhage or necrosis, and tumor spread into adjacent compartments are suggestive of aggressive growth. In one large series of neurogenic sarcomas, 56% of these tumors demonstrated nonhomogeneous enhancement, a feature also sometimes seen with atypical schwannomas and neurofi bromas. In an NF1 patient with a known or suspected plexiform neurofibroma who develops a rapidly progressive neurological syndrome referable to a known nerve, rapid tumor growth on serial imaging, or nonhomogeneous contrast enhancement, neurogenic sarcoma must be suspected and an appropriate management scheme initiated. Recently, patients with NF1 are having their tumors screened using 18FDG-PET (positron emission tomography). A hypermetabolic focus within tumors suggests malignant transformation. The management of neurogenic sarcomas is challenging and all cases should be referred to a multidisciplinary team at a tertiary-care center with special interest in the investigation and treatment of these rare and lethal tumors. The first, and most important, consideration remains the diagnosis of neurogenic sarcoma and requires a high index of initial suspicion. Multiple open biopsies from electrically silent areas of the tumor should be sent for intraoperative quick-section, and if consistent with malignancy, the incision should be closed without further attempts at resection. Even after appropriate biopsy of suspicious lesions, the diagnosis of neurogenic sarcoma is often far from certain. There are no gross pathological, histological, immunohistochemical, or ultrastructural markers that are characteristic of neurogenic sarcomas, and they are sometimes very difficult to differentiate from benign peripheral nerve tumors (e.g., ancient and cellular schwannomas) and other soft tissue sarcomas. The involvement of a senior neuropathologist with experience in the diagnosis of these tumors is essential. Once pathological confirmation is established, the clinician embarks on a metastatic survey that includes a thorough general physical examination to exclude overt metastatic disease, in addition to imaging of the chest (x-ray or CT scan), the most common site of distant tumor spread. Although the presence of distant metastases at the time of initial diagnosis is rare (in one large series, only two of 18 patients with neurogenic sarcoma had clinically obvious distant metastases), the initial metastatic survey determines, in large part, if the patient will receive aggressive or palliative therapy. If distant metastases are detected at the time of initial diagnosis, the approach to therapy is palliative. Palliative radiation may be administered locally, to the systemic metastases, or both. More aggressive measures may include the resection of a solitary pulmonary metastasis. The role of chemotherapy in the treatment of soft tissue sarcomas remains controversial, but various (mostly adriamycin-based) chemotherapeutic protocols are often used, with limited benefit. More commonly, the initial metastatic survey is negative, and patients are referred for aggressive management of their tumors. Simple excision of neurogenic sarcomas is not sufficient. Local recurrence, and the resulting increased likelihood of systemic metastasis, does nothing to improve patient survival. Therefore, the aim of surgery must be wide oncological resection with negative tumor margins (i.e., tumor-free margins equal to or greater than 2 cm in all directions). Unfortunately, in the majority of cases, anatomical constraints (e.g., intrapelvic origin of tumor) and aggressive tumor spread along the perineural plane of the involved nerve often make wide oncological resection impossible. In these cases, tumor-free, but not wide, margins are the surgical imperative, and radiation therapy, either before or after surgery, is required. It is also for this reason that many centers now advocate limb-salvage rather than limb disarticulation surgery. Radiation is administered preoperatively if dissection along critical structures is anticipated, e.g., popliteal artery. As discussed earlier, the role of chemotherapy in the management of soft tissue sarcomas is controversial. It is essential that all decisions be made by a multidisciplinary team comprising surgeons (neurosurgery, orthopedics, and plastics), oncologists (medical and radiation), and allied health professionals (nurses, physiotherapists, and occupational therapists) on an individual basis. These patients require aggressive rehabilitation and close follow-up to give them the best functional recovery and to prolong life. Despite aggressive multimodal therapy, the overall prognosis for neurogenic sarcomas is poor. The survival rate in one large series was 64%, with only 30% of patients likely to be disease-free at 5 years. These numbers are significantly worse than those for all soft tissue sarcomas, suggesting that the subgroup of neurogenic sarcomas carries a worse overall prognosis. In the majority of patients, the ultimate cause of death is systemic spread with a predilection for the lung parenchyma. Three prognostic factors are important in neurogenic sarcomas. First, tumors larger than 5 cm in diameter are more likely to present with neurological deficits, correlate with poorer pathological grade, and present a more formidable surgical challenge for complete en bloc resection with tumor-free margins. Second, higher tumor grade is correlated with worse prognosis. Importantly, in one large series, 15 of 18 patients had an escalation in tumor grade on formal pathological review when compared with grading of the tumor on quick-section, emphasizing the need for expert review of all tissue samples by an experienced sarcoma pathologist. Third, the surgeon’s ability to obtain tumor-free margins is critical. If this cannot be accomplished, patients almost certainly die of their disease. It cannot be overemphasized that patients should be managed by multidisciplinary teams at tertiary care centers with major clinical and research efforts in the treatment and investigation of neurogenic sarcomas.
 Case Presentation
Case Presentation
 Diagnosis
Diagnosis
 Anatomy
Anatomy
 Characteristic Clinical Presentation
Characteristic Clinical Presentation
 Differential Diagnosis
Differential Diagnosis
 Diagnostic Tests
Diagnostic Tests
 Management Options
Management Options
 Outcome and Prognosis
Outcome and Prognosis
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


