Gene knockout
Transgenic mice
Viral system
In vivo electroporation
Toxicity
No
No
Potential
No
Immunogenicity
No
No
Potential
No
Time specificity
No
No
Yes
Yes
Site specificity
No
No
No
Yes
Cost ($)
>20k
>5k
>2k
<0.2k
Procedure
Very complicated
Very complicated
Complicated
Simple
Integrity
Yes
Yes
Yes
No
Capacity
Unlimited
<10 kb
>10 kb
Multiple genes transfer
Very difficult
Difficult
Easy
Expression regulation
Remove
Upregulation
Upregulation/downregulation
Upregulation/downregulation
2 Materials
Materials and protocol may vary with different experimental purposes. Here we utilize an enhanced green fluorescent protein (EGFP) plasmid as an example to introduce the basic procedure of transferring a foreign gene into the embryonic brain , neonatal brain, and adult brain using electroporation .
2.1 Plasmid, Microinjection System, and Mice
1.
Microinjection glass needle for DNA injection in the in utero electroporation and neonatal electroporation: prepared using a P-97/IVF micropipette puller (Sutter Instruments, Novato, CA, USA) or equivalent. Conditions: pressure, 500; heat, 800; pull, 30; velocity, 40; time, 1 s.
2.
A modified metal injection needle is used for DNA injection into the adult rodent brain .
3.
ECM830 Electroporator with tweezer electrode (diameter: 7 and 10 mm) (Harvard Apparatus, Holliston, MA, USA) or equivalent.
4.
Purified pEGFP-N1 plasmid (>0.5 μg/μl) carrying GFP gene (CMV promoter) mixed with 100 μg/ml Fast Green (Amresco, Solon, OH, USA) (see Note 1 ).
5.
Pregnant ICR mice at embryonic stage 16 days (E16) used for the in utero electroporation .
6.
Newborn pups no older than 12 h used in the neonatal electroporation .
7.
8.
Nembutal.
9.
Hamilton syringe with a 30-gauge beveled needle (Hamilton Company, Reno, NV, USA).
10.
Microinjection pump.
11.
Stereotaxic apparatus.
12.
Isolated pulse stimulator (A-M System, Carlsborg, WA, USA).
13.
Surgical materials: Surgical scissors, gauze, sterile saline, 70 % ethanol, sutures.
3 Methods
3.1 In Utero Electroporation
1.
As shown in Fig. 1, anesthetize a timed-pregnant mouse with an intraperitoneal injection of 1 % Nembutal solution (60 mg/kg body weight).
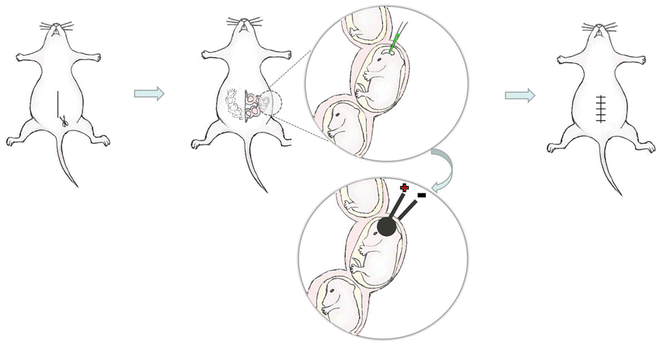

Fig. 1
The schematic diagram of in utero electroporation . (1) The abdominal cavity was opened and the uterus was exposed. (2) The DNA /RNA solution containing fast green was injected into the lateral ventricle. (3) Optimal electric pulses were delivered to the injected embryonic brain using tweezer electrode, and the anode was placed at the injected side. (4) The uterus was returned to the abdominal cavity, and the skin and muscle was sutured
2.
Place the operating board in a dissecting tray and place a stack of paper towels under the operating board to absorb spilled saline and prevent slipping of the board.
3.
Fix the mouse with its back on the operating board.
4.
Clean the abdomen with 70 % ethanol.
5.
Open the abdomen along the midline abdominal with scissors. 2–3 cm long incision is enough to operate.
6.
Cover the abdomen with a piece of folded gauze, which has an incision with the same size as abdominal incision.
8.
9.
Using the tweezer electrode, with the anode on the injected side, deliver five electric pulses at 50 V with 50 ms duration and 1 s interval to the injected embryonic head through the uterus wall.
10.
Place the uterus gently back into the abdominal cavity with hands after operating all the embryonic brain , and then fill the abdominal cavity with 37 °C pre-warmed normal saline.
11.
Suture the muscle and skin respectively, and then place the pregnant mouse on the 37 °C heat pad until awake.
12.
Sacrifice the mouse mother at a specific time point and dissect the electroporated brain . Keep the electroporated brain in normal saline until finishing the fluorescence observation.
3.2 Neonatal Electroporation
1.
Get Clinical Tree app for offline access

As shown in Fig. 2, keep the mouse mother out of the cage until finishing operating on all the pups (see Note 5 ). Newborn pups (12 h after birth) were anesthetized with diethyl ether and fixed by one hand.
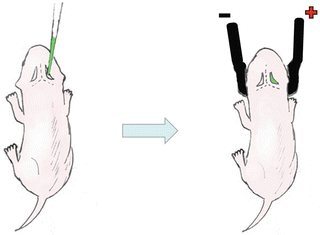

Fig. 2




The schematic diagram of neonatal electroporation . (1) The DNA /RNA solution mixed with fast green was injected into the lateral ventricle. (2) The Optimal electric pulses were applied to the injected head using tweezer electrode with the anode placed at the injected side
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree






