Chapter 81 Pharmacologic Treatment of Insomnia
Benzodiazepine Receptor Agonists
Abstract
A 2005 National Institutes of Health State of the Science Conference concluded that the benzodiazepine receptor agonists (BzRAs) were the only drugs for which adequate scientific evidence exists to support their use for chronic insomnia.1 This chapter reviews what is known about efficacy, effectiveness, and safety of the nine BzRAs approved for the treatment of insomnia by the United States Food and Drug Administration (FDA). A number of other BzRAs are approved for the treatment of insomnia in other countries, and research with those drugs is highly consistent with the data reviewed herein. In addition, implementation and assessment strategies for the pharmacologic treatment of insomnia are discussed.
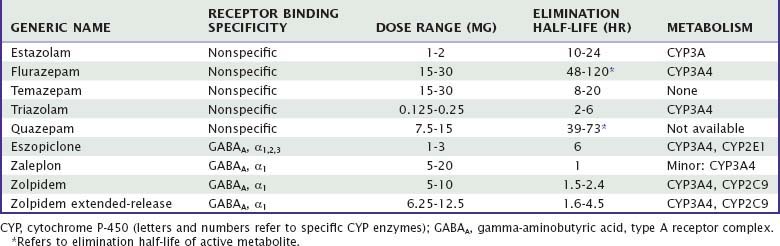
The group of drugs referred to as “benzodiazepine receptor agonists” are categorized according to the well-described common mechanism of action of these medications. All BzRAs act as allosteric modulators of gamma-aminobutyric acid (GABA) activity by binding to inotropic benzodiazepine receptors at the GABAA receptor complex. BzRAs serve to further open chloride ion channels and facilitate GABA inhibitory activity. Some of these drugs (Table 81-1) have a benzodiazepine chemical structure (i.e., estazolam, flurazepam, quazepam, temazepam, triazolam) and others do not (i.e., eszopiclone, zaleplon, zolpidem). BzRAs demonstrate affinity for four benzodiazepine receptor subtypes (referred to as α1, α2, α3, and α5) located at the GABAA pentameric complex. Benzodiazepines tend toward comparable affinity for the four receptor subtypes, whereas the affinities of zolpidem and zaleplon for the α1 subtype are higher than for other subtypes. Eszopiclone shows less preference for α1, having some activity at α2 or α3 (or both). The functional significance of these binding differences for effect on sleep in humans remains unclear. In contrast, preclinical investigations of knock-in rodent strains suggest different functional characteristics of the benzodiazepine receptor subtypes.2
The BzRAs are generally recommended as first-line hypnotics for several reasons. All BzRAs have been shown to be efficacious, although there are some differences among drugs, associated with pharmacokinetic properties, and marketed dose. Compared with other central nervous system (CNS) drug classes, the margin of safety or therapeutic index (i.e., the effective dose relative to lethal dose) is wide.3 For example, barbiturates have margins of safety on the order of two to four times the effective dose, whereas for BzRAs, the margin of safety can be as great as 100. Abuse of or dependence on BzRAs is uncommon in the therapeutic context, most likely accounted for by the relatively mild reinforcing effects and self-administration patterns. Summaries of the efficacy, effectiveness, and safety characteristics of BzRAs follow. Unless otherwise stated, the summary material refers to the FDA-approved dosages of the drugs.
Efficacy and Effectiveness
Primary Insomnia
Most insomnia clinical trials have documented the efficacy of hypnotics using patients’ reports, PSG, or both, in patients with primary insomnia. A meta-analysis of clinical trials with benzodiazepines and zolpidem found that these drugs in aggregate produce reliable improvements in the sleep of persons with chronic insomnia. Importantly, the median duration of the studies included in the analysis was only 1 week.4 Other meta-analyses5–8 largely concur regarding short-term efficacy. However, the utility of meta-analyses to assist the clinician is fairly low, because they combine data from several different drugs with widely different pharmacokinetics, which affects the outcome variables examined in the meta-analysis. Additionally, two or more doses of a drug may be included in the analysis. It is much more instructive to examine the strengths and weaknesses of individual drugs as opposed to generalizing across all drugs in a category. Simply put, meta-analyses tell us about the pharmacology of a group of drugs, but they are not informative about the properties of individual drugs or what the best drug is for a given patient. Later, similarities among BzRAs are discussed, but important differences among drugs will be emphasized.
Tolerance is defined as a reduction of a drug’s effect with repeated administration of a constant dose, or the need to increase the dose to sustain a specific level of effect. Despite frequent speculations in the medical literature, tolerance to the hypnotic effects of BzRAs does not develop in subjects in the vast majority of studies, at least for therapeutic doses and for the periods of time that have been studied. Investigations that are often cited as evidence for developing tolerance, such as the one by Mitler and colleagues,9 show gradual improvement in sleep over time in the placebo group, in the face of a constant effect in the drug group, resulting in loss of statistical significance between groups. Yet the within-group comparison of an early drug effect versus a late drug effect fails to show any change in drug activity with repeated use. Thus, it cannot be concluded that tolerance has developed, but rather that unspecified changes occur over time with placebo, such as spontaneous remission, regression to the mean (if there are sleep-disruption criteria for entering the study), sleep hygiene influences inherent in protocol adherence, Hawthorne effects, and true placebo effects.10 This is further evidenced by the fact that patients rarely escalate the nightly use of hypnotics even with long-term use.11
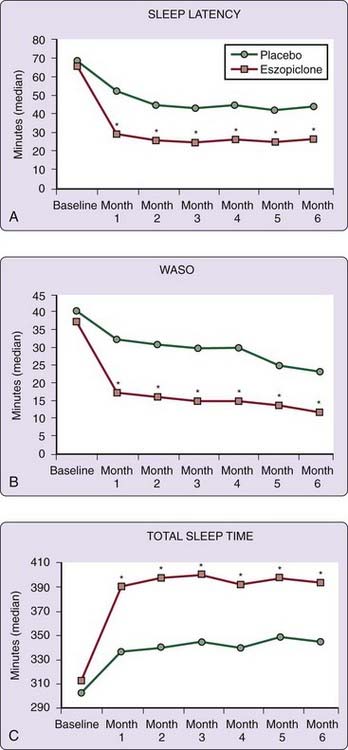
Nearly 30 years ago, Oswald and colleagues12 reported that lormetazepam and nitrazepam, two benzodiazepines available in some European countries, retained their effect by some patient estimates of hypnotic efficacy during 24 weeks of nightly use. More recently, in rigorous PSG studies, zolpidem 10 mg and zaleplon 10 mg were shown to retain efficacy for 5 weeks of nightly use.13,14 A 2003 landmark study of several hundred patients with primary insomnia showed continued hypnotic efficacy of eszopiclone for 6 months of nightly use.15 Patient reports of sleep latency and WASO were significantly reduced with eszopiclone 3 mg as compared with placebo at each monthly time point. Total sleep time, number of awakenings, and sleep quality were also better than with placebo at each monthly time point. These 6-month findings have been replicated by Walsh and colleagues16 and are shown in Figure 81-1. Examination of open-label extension data of the 2003 6-month, eszopiclone study suggested sustained efficacy for a total of 12 months.17 Other evidence for sustained efficacy of nightly BzRA use includes a 3-month trial with indiplon, a nonmarketed hypnotic.18
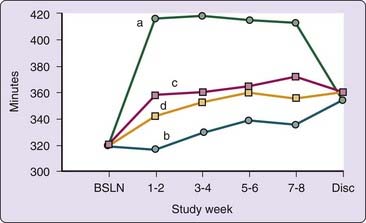
Efficacy of nonnightly use of zolpidem 10 mg has been investigated for up to 12 weeks.19,20 In these studies, ratings of sleep latency, total sleep time, number of awakenings, and sleep quality were all improved on nights when zolpidem was taken (an average of 3 or 4 nights per week), as compared with placebo (also an average of 3 or 4 nights per week). Additionally, investigator global ratings, which considered both medication nights and no-medication nights, indicated reduced insomnia severity with zolpidem. Total sleep time data on nights when a pill (either zolpidem or placebo) was taken or not taken during an 8-week study are shown in Figure 81-2. Importantly, an examination of sleep on nights when no drug was taken immediately after a zolpidem night failed to reveal evidence of rebound insomnia.14 A 6-month study with an extended-release formulation of zolpidem used 3 to 7 nights per week has generated similar results over the duration of the study.21
Comorbid Insomnia
Comorbid insomnia refers to insomnia coexistent with other medical or psychiatric conditions. Although BzRAs have not been extensively studied in patients with comorbid insomnia, the published data indicate improvements in sleep similar to those seen with primary insomnia. One of the earliest studies relevant to BzRA efficacy in comorbid insomnia examined rheumatoid arthritis patients with insomnia using PSG techniques. Triazolam 0.25 mg was found to reduce sleep latency and number of awakenings and to increase total sleep time across six nights of treatment.22
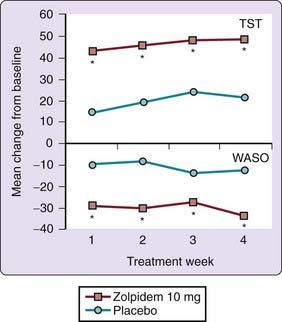
Zolpidem 10 mg increased total sleep time and improved sleep quality in a 4-week placebo-controlled study in depressed patients with refractory insomnia despite adequate control of depressive symptoms with selective serotonin reuptake inhibitor (SSRI) medication.23 Figure 81-3 contains total sleep time data and wake after sleep time values during that 4-week study. Preliminary reports indicate that zolpidem extended-release coadministered with escitalopram improves subjective measures of sleep in patients with comorbid depression24 and in those with comorbid anxiety.25
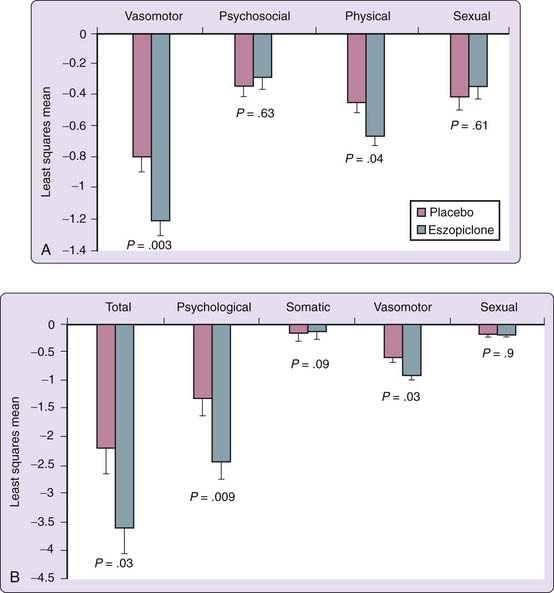
A number of studies have been conducted to assess efficacy of eszopiclone in comorbid insomnia. Eszopiclone combination therapy with fluoxetine was superior to fluoxetine and placebo in improving sleep in patients with newly diagnosed major depressive disorder and comorbid insomnia.26 In addition, the combination-therapy group was found to have a significantly greater reduction in depressive symptoms, a higher rate of remission, and a faster time to the onset of the antidepressant effect. Similarly, eszopiclone co-therapy with escitalopram was superior to escitalopram plus placebo in improving the sleep of patients with insomnia and generalized anxiety disorder,27 and anxiety and depressive symptoms were reduced further in patients receiving co-therapy. Finally, insomnia patients treated with eszopiclone 3 mg for 4 weeks during perimenopause or early menopause reported significantly improved sleep throughout the trial, but in addition they had significant improvements in mood, quality of life, and menopause-related symptoms (Fig. 81-4).28
Effectiveness Studies
Using an epidemiologic methodology, Ohayon and colleagues29 reported that of 532 patients describing chronic insomnia and long-term use of hypnotic medications to help them sleep, 67% rated their sleep quality as improved “a lot,” and only 14.4% reported little or no improvement with medication. Balter and Uhlenhuth30 interviewed subjects who in the past year either reported significant trouble with insomnia or had taken a medication to help them sleep. They found very high satisfaction rates among users of hypnotics when they asked, “Taking into account both the positive effects on your sleep and daytime functioning and any negative effects you may have experienced, would you take this medication again for the same purpose?” Affirmative responses were received from 84% of those taking triazolam, 82% for flurazepam, 74% for temazepam, and 61% for over-the-counter aids. In summarizing the findings of this intensive interview study, the authors concluded that “the benefits of treatment with prescription hypnotics are substantial and far outweigh the risks as defined in this study.” It is important to recognize that in both of these studies, the cited data were obtained from either current users of sleep-promoting medications or those who had used them at some time in the past year. In these studies, the severity or nature of the insomnia is not characterized, nor are the duration, dose, or pattern of hypnotic use or other aspects of the patients. Clearly, effectiveness data in the management of insomnia needs to be an important future research priority.
Long-term open-label studies conducted in both adult and elderly subjects in outpatient settings also provide some information about effectiveness.17,31–34 Zolpidem, zaleplon, and eszopiclone have been evaluated over periods of 6 to 12 months in such studies. In general, patients and physicians report sustained benefit of BzRAs for the duration of the studies, without adverse reactions unique to long-term use. The generalizability of results and the confidence level regarding any conclusions drawn must be tempered by the self-selection of those remaining in these open-label studies.
Impact on Daytime Symptoms of Insomnia and Comorbid Conditions
Diagnostic criteria of insomnia include some form of subjective daytime impairment perceived to be consequent to the sleep disruption, such as fatigue, sleepiness, concentration and memory problems, and mood changes. If the daytime impairment is mediated by the insufficient or disturbed sleep, it should be possible to demonstrate a reduction of impairment with improved sleep. Elimination or reduction of the daytime problem could be viewed as another measure of treatment efficacy. Until recently, few studies have shown daytime consequences of insomnia, especially on laboratory-based tasks of psychomotor and cognitive performance. There is growing evidence, however, that in addition to the perceived daytime dysfunction, insomnia patients have a substantially increased risk of future mood and other psychiatric disorders,35–38 higher rates of health care use and health care costs,39–42 elevated risk of falling,43–45 reduced quality of life,46,47 and increased absenteeism from work.39,48,49 Cognitive impairments are reported less often, but recent investigations have differentiated insomniac subjects from normal sleepers.50,51
Until recently, the focus of research on the daytime effects of BzRAs used to treat insomnia was the assessment of residual sedation rather than relief of daytime symptoms or other consequences. However, the few studies that have systematically assessed patient-reported daytime measures generally show improvement. For example, in the two 6-month eszopiclone studies of patients with primary insomnia previously discussed,15,16 patients’ reports of daytime alertness, ability to function during the daytime, and physical sense of well-being were all significantly better in the eszopiclone group than in the placebo group.
The waking impact of chronic comorbid insomnia may differ from that of primary insomnia. For example, patients with insomnia and periodic limb movement disorder52 and those with insomnia and rheumatoid arthritis53 have been shown to have lower than optimal daytime alertness as determined by multiple sleep latency test (MSLT) scores, which improve significantly after 6 nights of treatment with triazolam. Three studies of older adult patients showed increased daytime alertness on the MSLT resulting from increased sleep consolidation and duration with BzRA treatment.54–56 In part, then, the difficulty in finding systematic daytime impairment in chronic insomnia may be the result of differing daytime consequences in subgroups of insomniac patients.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree