Chapter 112 Restrictive Lung Disorders
Abstract
Resting lung volume (the volume at the end of a normal exhalation) is determined by the balance between the elasticity and inward recoil of the lung and the outward recoil properties of the chest wall. The former tends to reduce lung volume. Forces that tend to preserve or increase lung volume include the normal, inherent mechanical tendency of the chest wall (considered to consist of three components: the thoracic cage, diaphragm, and abdomen) to recoil outward, as well as tonic inspiratory muscle activity. Restriction of lung expansion may thus be the result of disorders of the lung parenchyma (intrapulmonary restriction) or disorders of structures adjacent to the lungs, such as the skeletal and soft tissue components of the chest wall (extrapulmonary restriction). The physiologic consequences of these two types of restriction are different and result in different effects on respiratory control mechanisms. In general, intrapulmonary restriction is characterized by stimulation of pulmonary vagal receptors, with subsequent tachypnea (e.g., rapid, shallow breathing) and hyperventilation. Patients with extrapulmonary restriction may exhibit blunted ventilatory chemosensitivity and hypoventilation. On the basis of differences during the awake state, one would expect these two groups to behave differently during sleep. Extrapulmonary restriction is most commonly associated with obesity, kyphoscoliosis, neuromuscular diseases, and pregnancy, and intrapulmonary restriction with interstitial lung disease and lung resection. Sleep disorders during pregnancy are reviewed in detail in Chapter 139, and neuromuscular diseases are reviewed in Chapter 88.
Obesity
Definition, Epidemiology, and Risk Factors
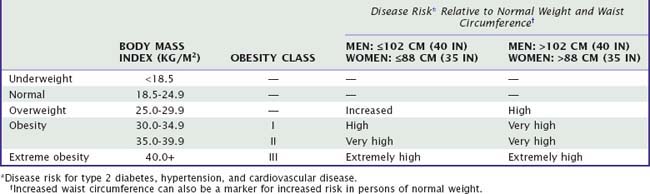
Obesity is a condition that occurs when body fat exists in excess to the extent that health is impaired.1 The most common clinical measure of obesity in adults and children is the body mass index (BMI).1,2 In adults, the BMI thresholds for overweight and obesity are 25 kg/m2 (Asians, 23) and 30 kg/m2 (Asians, 27), respectively. Children are overweight when their BMI is greater than or equal to the sex- and age-specific 95th percentile.2 In addition, waist circumference can be used to determine central obesity, which appears to be a better predictor of obesity-related diseases.3,4 Thresholds of 102 cm for men and 88 cm for women define central obesity.
In the United States, the prevalence of obesity in adults increased from 13% to 32.9% between the 1960s and 2003-2004. In 2003-2004, 66% of adults and 16% of children and adolescents were overweight.5 Similar data were reported for Europe6 and other developed countries. However, more recent U.S. data do not show a statistical difference in prevalence of obesity between the 2003-2004 survey (32.9%) and the 2005-2006 survey (34.2%).7
Numerous studies have shown that obesity increases morbidity and mortality.1,4 Obesity is the second leading preventable cause of disease and death in the United States, surpassed only by tobacco use. Overweight and obesity are associated with significant pathophysiologic alterations and clinically relevant consequences including insulin resistance, type 2 diabetes, dyslipidemia, hypertension, cholelithiasis, certain forms of cancer, steatosis hepatitis, gastroesophageal reflux, obstructive sleep apnea (OSA), degenerative joint disease, gout, lower back pain, and polycystic ovarian syndrome.8 Some of these conditions are components of metabolic syndrome. A significant subset of obese people are considered metabolically healthy: they appear to be protected from obesity-related metabolic abnormalities9 and have a less abnormal inflammation profile.10
Obstructive sleep apnea syndrome (OSAS) has been reported in about 51% of people with obesity11 and in a majority of people with morbid obesity.12 OSAS has been hypothesized to be a predisposing factor for the development of metabolic syndrome.13
Pathogenesis
Obese patients have compromised respiratory function while awake and seated. Their ventilatory function may become worse when they assume the supine position, and it may deteriorate further during sleep. However, the compromise of respiratory function is not the same in all types of obesity.14 Predominantly abdominal adiposity (central obesity) may restrict the descent of the diaphragm and limit lung expansion to a greater degree than adipose tissue mass in other locations.
Breathing in the Supine Position
In the supine position, the abdominal hydrostatic force is applied through the diaphragm to the lungs. In addition, the chest wall in obese patients (particularly in central obesity) is stiffer in the supine than in the upright position. Thus, FRC is further lowered in the supine compared with the seated or upright positions, which promotes greater and more widespread regional reduction of the 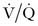 ratio and greater consequent hypoxemia. Moreover, varying degrees of increased upper airway resistance during sleep further increases the work of breathing. This would be the case even in the absence of obstructive apnea, given the normal increase in airway resistance that usually accompanies sleep (see Chapters 23 and 101).
ratio and greater consequent hypoxemia. Moreover, varying degrees of increased upper airway resistance during sleep further increases the work of breathing. This would be the case even in the absence of obstructive apnea, given the normal increase in airway resistance that usually accompanies sleep (see Chapters 23 and 101).
Breathing during Sleep
Obese patients may present with the following breathing abnormalities during sleep:
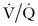 mismatch and hypoxemia. Moreover, the abnormal respiratory system mechanics that may accompany obesity, in conjunction with normal sleep physiology such as the tendency for reduced ventilation (relative to wakefulness), hypotonia of the intercostal muscles, and lower lung volume (see Chapter 22), can lead to oxyhemoglobin desaturation and hypercapnia in about 10% of obese subjects even in the absence of OSA.11
mismatch and hypoxemia. Moreover, the abnormal respiratory system mechanics that may accompany obesity, in conjunction with normal sleep physiology such as the tendency for reduced ventilation (relative to wakefulness), hypotonia of the intercostal muscles, and lower lung volume (see Chapter 22), can lead to oxyhemoglobin desaturation and hypercapnia in about 10% of obese subjects even in the absence of OSA.11Sleep Apnea and Hypoventilation
Obesity is the greatest risk factor for developing OSA. Indeed, apneas and hypopneas or respiratory effort–related arousals15 can be observed together or with predominance of one of these events. Although many obese patients develop repetitive sleep obstructive events, some do not. Increased weight alone is not sufficient to cause OSA, and differences in neuromuscular control of upper airway muscles and variations in ventilatory control mechanisms can play a role. In obese individuals, the accumulation of fat in the lateral parts of the pharynx increases the extraluminal pressure and may alter the geometry of the upper airway, predisposing to the collapse.16 The variability in upper airway fat deposition may be related to sex and genetic17 factors.
Nocturnal hypoventilation without daytime hypercapnia occurs in 29% of nonselected obese subjects and is frequently associated with OSA.8 Sometimes hypoventilation is also present during wakefulness, reflecting obesity–hypoventilation syndrome (OHS), which may also be a component of the pickwickian syndrome (see later discussion of obesity–hypoventilation syndrome). OSA is discussed in detail in other chapters of this section.
Diagnosis
As previously discussed, the BMI is used to diagnose obesity.1,2 Cutoff points of 25 and 30 kg/m2 in white adults identify overweight and obesity, respectively. Waist circumference, waist-to-hip ratio, and neck circumference are other metrics of adiposity and body fat distribution. Waist circumference is a better predictor of associated cardiometabolic disorders than BMI, and it could be an additional indicator for monitoring changes in obesity (Table 112-1).3
Diagnoses of breathing-related sleep disorders should be confirmed by performing comprehensive polysomnography with or without monitoring of end-tidal or transcutaneous CO2 tension (PCO2tc).18 Less complete polysomnography or a cardiopulmonary sleep study (in hospital or at home) may be considered in selected patients with suspected OSA (see Chapter 105).
Treatment
Achieving and maintaining normal weight is a challenge, and strategies for treating obesity are beyond the scope of this chapter. Several institutions, medical societies, and national health departments have guidelines for management and prevention of obesity.19,20
Dietary weight loss is recommended for overweight and obese patients with OSAS. However, lack of adherence to diet,21 regain of lost weight, and interindividual improvement variability are frequent.22 Large, randomized, placebo-controlled outcome studies are necessary for a full evaluation of the role of weight loss through dieting in OSAS patients. There are no randomized and controlled studies of the efficacy of bariatric surgery in sleep breathing disorders, but information from prospective long-term clinical series is now available. Accordingly, weight loss, secondary to bariatric surgery, leads to improvement in sleep quality and apnea-hypopnea index (AHI).23 Although a 1-year follow-up study showed complete resolution of OSA in only 1 of 24 patients,24 a meta-analysis reported a high resolution rate of sleep apnea (86%).25 Therefore, although more information is necessary, bariatric surgery may play a role in the treatment of morbidly obese patients with OSA, as an adjunct to less invasive and rapidly applicable first-line therapies (see Chapter 111).
Obesity–Hypoventilation Syndrome
Definition, Epidemiology, and Risk Factors
The term obesity–hypoventilation syndrome describes the concurrence of diurnal hypoventilation (PaCO2 > 45 mm Hg) and obesity (BMI > 30) when other causes of alveolar hypoventilation, such as severe obstructive or restrictive pulmonary disease, significant kyphoscoliosis, severe hypothyroidism, neuromuscular diseases, or other central hypoventilation syndromes, can be excluded. Although OHS can exist without sleep apnea, approximately 90% of patients with OHS have OSA.26,27
The prevalence of OHS is unknown, but it is believed to affect a minority of the obese population. The prevalence of OHS in subjects with suspected sleep apnea varies from 10% to 30%, depending on the study method, BMI score, geographic location (where the patients were selected), and the AHI cutoff point used to diagnose sleep apnea.28 Morbid obesity (BMI > 40) is associated with a higher prevalence of OHS.29 This type of obesity is considered more common in women than in men, and some studies have identified a higher prevalence of OHS in women.30–32 Nevertheless, when several series of patients were examined together,33 OHS was diagnosed more frequently in men (66%). There are no clear risk factors for development of OHS in obese subjects except morbid obesity.
Pathogenesis
Respiratory Mechanics
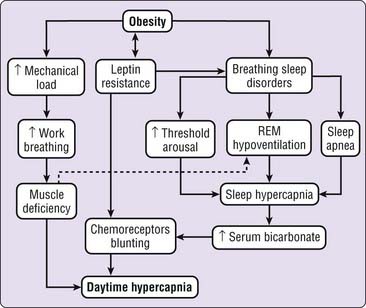
As discussed, the mechanical alterations on the respiratory system produced by obesity can cause respiratory muscle dysfunction34 and daytime hypercapnia.
Control of Breathing
Diminished responses in both hypercapnia and hypoxia have been observed in patients with OHS,35 but it is not entirely clear if the origin is primary (central) or is secondary to mechanical load. Some studies indicate that there is no genetic predisposition to diminished chemosensitivity,36 and that it can improve with noninvasive intermittent mechanical ventilation (NIMV),35,37 suggesting that a secondary origin is more probable. Because the mechanical alterations caused by obesity do not change with NIMV, the improvement in chemosensitivity would depend on other secondary factors (e.g., reduced buffering and muscle fatigue). On the other hand, the assessment of central neural drive presents some interpretative difficulties. First, ventilatory pump (e.g., the chest wall, including ventilatory muscles) dysfunction may prevent evaluation of the central neural control of breathing because the dysfunction may impair translation of the central drive to breathe into measured ventilation. Second, the increased buffering capacity to chronic elevation of PaCO2 could have a secondary effect on central “real” neural drive. These difficulties contribute to the absence of clear evidence to determine if reduced chemosensitivity has a primary or secondary origin.
The hormone leptin, produced by fat cells and thought to reduce appetite, may also act in the nervous system to increase ventilation. Thus hypoleptinemia, or central nervous system resistance to leptin’s ventilation-promoting effects, may promote daytime hypercapnia. Although leptinemia is elevated in obese subjects and OSA patients, the leptin level is almost twice as high in patients with OHS (obesity and daytime hypercapnia) as in patients with obesity without daytime hypercapnia, when measurements of AHI and obesity are similar.38 On the other hand, leptinemia decreases with NIMV treatment39 in the same way as daytime hypercapnia. Thus, some studies have showed correlation between leptinemia and daytime hypercapnia38,40 and between leptinemia and reduction of hypercapnic ventilatory response.41 Given that leptinemia is very high in patients with OHS and is associated with hypercapnia, the respiratory failure present in patients with OHS may result from leptin resistance in the central nervous system.
Sleep
When apneic events are present during sleep, they can cause transitory nocturnal hypoventilation (Videos 112-1 and 112-2![]() ). Daytime hypercapnia in patients with OSA has been associated with the ratio of event duration to interevent duration, which would indicate that diurnal and nocturnal hypercapnia depends on insufficient recovery from the hypoventilation produced by apneic events, especially during rapid eye movement (REM) sleep.42 Transitory hypercapnia would cause higher daytime serum bicarbonate (which results in increased blood buffering capacity) and consequent blunting of the central CO2 response.28 A similar mechanism can happen when nocturnal hypoventilation (especially during REM sleep) occurs without apneas and hypopneas.
). Daytime hypercapnia in patients with OSA has been associated with the ratio of event duration to interevent duration, which would indicate that diurnal and nocturnal hypercapnia depends on insufficient recovery from the hypoventilation produced by apneic events, especially during rapid eye movement (REM) sleep.42 Transitory hypercapnia would cause higher daytime serum bicarbonate (which results in increased blood buffering capacity) and consequent blunting of the central CO2 response.28 A similar mechanism can happen when nocturnal hypoventilation (especially during REM sleep) occurs without apneas and hypopneas.
As most of the previous mechanisms are interconnected, the combined action of all (or the majority) of them is a more realistic explanation for daytime hypercapnia in patients with OHS (Fig. 112-1).
Clinical Features
The common clinical features of patients with OHS are obesity, a plethoric complexion, dyspnea, cyanosis, and evidence of right heart failure including peripheral edema.29 Such patients may not complain of dyspnea even when hypoxemia and hypercapnia are present. If OSA is present, other symptoms, such as loud snoring, nocturnal choking episodes with witnessed apneas, excessive daytime sleepiness, morning headaches, and tiredness, can be evidenced.
Diagnosis
Clinicians should consider OHS in obese patients (BMI > 30) with daytime hypercapnia (PaCO2 > 45 mm Hg) without the existence of other diseases that might cause hypercapnia, such as severe obstructive or restrictive pulmonary disease, significant kyphoscoliosis, severe hypothyroidism, neuromuscular diseases, or other central hypoventilation syndromes. Pulmonary function tests can be normal, but more commonly they show a mild to moderate restrictive disorder associated with the mechanical alterations of obesity. Laboratory testing should include thyroid function tests to exclude severe hypothyroidism, and a complete blood count to rule out secondary erythrocytosis. An electrocardiogram and echocardiogram can demonstrate right ventricular hypertrophy, right atrial enlargement, or pulmonary hypertension.26,30
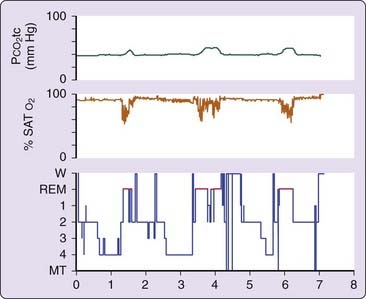
An overnight sleep study may demonstrate a spectrum of findings: episodes of upper airway obstruction, sleep fragmentation, transitory oxygen desaturation and significant hypoventilation (increase in PaCO2 of more than 10 mm Hg and oxygen desaturation) during REM sleep.31 In patients without relevant OSA, hypoventilation is the main finding (Fig. 112-2). There are no studies that recommend a limited cardiopulmonary sleep study (portable monitoring), instead of complete polysomnography, for the diagnosis of OSA in OHS.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


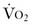 ) and carbon dioxide production (
) and carbon dioxide production (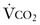 ). Thus, to sustain normal arterial blood gas tensions (Pa
). Thus, to sustain normal arterial blood gas tensions (Pa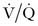 ), leading to a reduced
), leading to a reduced 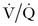 ratio and causing hypoxemia.
ratio and causing hypoxemia.