Chapter 108 Surgical Management for Obstructive Sleep-Disordered Breathing
Historical Prospective
In this chapter, obstructive sleep-disordered breathing (OSDB) will be used to collectively refer to snoring, upper airway resistance syndrome (UARS), obstructive sleep apnea (OSA), obstructive sleep hypopnea (OSH) and the combination of the latter two, obstructive sleep apnea–hypopnea (OSAH). These are described in detail in other chapters in this section. The spectrum of OSDB reflects various degrees of the upper airway obstruction occurring episodically during sleep, combined with some yet-unidentified central nervous system abnormality of ventilatory control.1–3 This disordered breathing results in cyclic exposure to hypoxia, sleep fragmentation, altered sleep architecture, and subsequent excessive daytime sleepiness (EDS) as well as other clinically important consequences that are discussed in detail elsewhere in this volume.
The pathophysiologic mechanism(s) culminating in upper airway obstruction during sleep are multifactorial, but they generally reflect a disadvantageous influence on the delicate balance of forces that are necessary to maintain upper airway patency in the presence of forces that promote collapse during sleep. Ideally, surgical approaches strive to intervene at the specific anatomic regions that become obstructed during sleep. Medical approaches, including risk modification (manipulation of body position during sleep; avoidance of alcohol and sedating medications), weight loss, and positive airway pressure modalities are usually the first avenues of treatment (see Chapters 106 and 110). Patients who are unable or unwilling to comply with medical management may be candidates for surgery.
Rationale
The rationale for surgical intervention is to improve the quality of life, reduce the risk for morbidity, and potentially enhance longevity. Thus, this rationale incorporates the behavioral and pathophysiologic consequences caused by sleep-related upper airway obstructions that are described in detail in Chapter 79. In addition, by virtue of its inherent invasiveness, the potential benefit to risks ratio is a preeminent consideration.
Indications and Contraindications
The indications and relative contraindications to surgery are listed in Box 108-1 and Box 108-2, respectively. Surgery may be appropriate in patients with an apnea–hypopnea index (AHI; the average number of apneas plus hypopneas per hour of sleep) that is at least 20; an AHI less than 20 in conjunction with severe daytime sleepiness is also an indication for surgery. In addition, a trial of continuous positive airway pressure (CPAP) that alleviates symptoms can provide a further rationale for surgery by linking symptoms to sleep-related upper airway dysfunction. For example, if a patient’s symptoms (such as EDS) do not improve during a CPAP trial despite alleviation of OSDB, it is unlikely that surgery will improve symptoms, and an alternative etiology for them should be sought.
Pretreatment Recommendations and Concerns
Because EDS has numerous other causes, such as narcolepsy, insomnia, and sleep deprivation, the workup should include a complete medical and sleep history, along with a head and neck evaluation, to assess health and detect comorbid medical conditions. Pretreatment evaluation should include full nocturnal polysomnography (PSG) and not cardiopulmonary testing, which provides more limited information. A radiographic cephalometric analysis, along with fiberoptic nasopharyngoscopy, is helpful in evaluating soft tissue and skeletal anatomy of the airway. Sleep endoscopy is now being tested in some centers in an attempt to further evaluate the airway in OSDB.4 However, at this time there are no reported clinical outcome studies that support the routine use of this diagnostic method.
Rama and colleagues5 evaluated the literature from 1980 to 2002 that reported on a variety of imaging techniques to identify the site(s) of obstruction during sleep, including three-dimensional computed tomography (CT) or magnetic resonance imaging (MRI). They concluded, in part, that identifying “the precise location of the sites of obstruction in patients with OSAH may not be possible using current imaging techniques.” Computational modeling6 and advanced three-dimensional software are now also being investigated. Unfortunately, most of these studies tend to suffer the same confounders because there are at present no known clinical surgical-outcome trials to support correction of the designated site of obstruction as predicted preoperatively. No single test or procedure should be relied on to make a definitive surgical treatment plan. A systematic medical and surgical review should determine the type of sleep disorder, establish parameters of severity, identify comorbidity factors, identify probable sites of upper airway obstruction, decide if treatment is emergent or elective, and assess the risk-to-benefit ratio of surgery.
Although the exact etiology of OSDB remains incompletely understood, the areas producing clinical anatomic obstruction are usually well defined.7–10 A major confounder in determining the area of obstruction is that other, less-perceptible regions may be participating in the obstructive process. In addition, our examinations are performed while the patient is awake. In this regard, when the patient is asleep the airway can change enough to adversely affect the patency of areas in which the possibility of obstruction is less obvious during wakefulness. OSDB can be viewed as a surgically responsive problem caused by the significant contribution from areas of disproportionate anatomy and diffuse upper airway collapse. A systematic approach and application of a surgical protocol will yield improved clinical outcomes for children and adults.
Clinical Evaluation in Assessing Surgical Candidacy
Clinical Examinations
Deflection of the nasal septum, enlargement of the turbinates, and webs, polyps, or masses are potentially important in the patient with OSDB. The oral cavity should be examined by assessing dental health, class of occlusion (I, II, or III), oral mucous membranes, and tongue. Issues that mitigate surgical candidacy can include pathology, infection, soft tissues that could not be removed safely, and bony jaw abnormalities. Further examination includes evaluating the length of the soft palate, redundancy of the lateral pharynx, size of the tonsils, and the tongue, vallecula, epiglottis, larynx, and pyriform sinuses. Cephalometric radiographs assist in the overall evaluation of the soft tissue and bony configuration. Other potential upper airway imaging methods include MRI and volumetric CT.11,12 Fujita13 proposed a classification of obstructive regions in the upper airway that is useful in planning therapy and for standardization of scientific reporting (Table 108-1).
Table 108-1 Fujita Classification of Obstructive Regions
| TYPE | LOCATION |
|---|---|
| I | Palate (normal base of tongue) |
| II | Palate and base of tongue |
| III | Base of tongue (normal palate) |
From Fujita S. Pharyngeal surgery for obstructive sleep apnea and snoring. In Fairbanks D, Fujita S, Ikematsu T, et al, editors. Snoring and obstructive sleep apnea. New York: Raven Press; 1987. p. 101-128.
Cephalometric Imaging
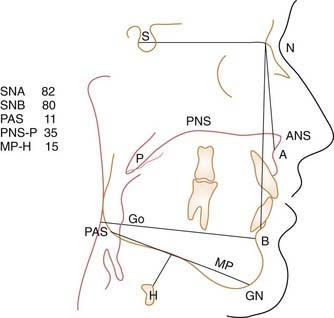
Cephalometric images provide reliable presurgical and postsurgical data on soft tissue and skeletal relationships, length of soft palate, posterior airway space (PAS), and hyoid position (Fig. 108-1). The use of cephalometric radiographs and the anthropomorphic measurements (except the subspinale [SNA] angle and the sella-nasion-supramentale [SNB] angle) seen in Figure 108-1 were first described in 1983 by Riley and coworkers.14 Although easily obtained and relatively inexpensive, cephalometric evaluation is limited in that it is a two-dimensional view and the patient is evaluated while awake and seated, not asleep and recumbent. This technique should not be relied on alone to determine the most appropriate surgical intervention.
Approach to Surgical Treatment
A systematic approach to the surgical management of patients with OSDB is essential for quality care. A philosophy of treatment is outlined in Box 108-3.
Medical management primarily consists of reducing risk factors and using one of several available positive airway pressure therapy modalities to stabilize the upper airway during sleep. Weight loss was recognized more than 30 years ago as an important treatment, and the merit of weight loss in obese patients with OSDB is well documented. Peppard and colleagues15 reported that weight gain in OSDB, even as minor as 10% (relative to stable weight), can result in an approximate increase in a patient’s AHI of 32% (see Chapter 106). Although the ability of patients with OSDB to achieve and maintain adequate weight reduction through dietary means has been limited, a resurgence of interest in obesity with increasing success in therapeutically significant weight reduction through surgery may yield new methods of control (see Chapter 106).
Issues in Evidence-Based Medicine
The American Medical Association16 reported that surgical placebo controls should be used only when no other trial design will yield the requisite data. Moreover, unlike trials evaluating medical therapy, such trials for sleep-disordered breathing (SDB) surgery are clearly unethical and impractical. Sham surgery (and attendant anesthesia and postoperative analgesia) has inherent and usually unacceptable risks and discomfort to patients with an underlying abnormally collapsible upper airway (some degree of edema is likely even with a sham procedure). Furthermore, even a sham procedure can alter the feasibility of subsequent surgical intervention. Blinding of the patient and investigator to visible evidence of the intervention is impossible, thereby limiting the value of RCT design. The likelihood of recruiting patients who are willing to accept these risks, especially in the context of randomization to a placebo arm, places another fundamental barrier to the conduct of the RCT design in the context of surgical intervention of SDB.
Evidence-based medicine has been defined as “integrating individual clinical expertise and the best external evidence.”16 Most surgical results for SDB, such as widening of the upper airway and improved function, may be objectively assessed by, for example, preoperative and postoperative polysomnography, esophageal manometry, and airway imaging including cephalometrics, CT, MRI, three-dimensional volumetric imaging, and direct comparison to preoperative and postoperative management with positive airway pressure. In most cases, this should be sufficient to support efficacy and effectiveness.
We believe that although surgical trials are still limited by inability to blind study participants, high-quality evidence can be ethically and feasibility acquired through a design that randomizes patients to an experimental surgical intervention, a current clinical standard surgical intervention for the specific patient group, or the current gold-standard medical treatment. This has been our practice.17,18 Thus, it is important that patients be made aware of the limitations of data available for some procedures and be presented with estimates that are the most accurate available of risks, benefits, and outcomes so that they can make an informed decision.
Two-Phased Surgical Protocol
The Powell-Riley surgical protocol is a two-phase approach that directs surgical treatment toward the specific regions of obstruction during sleep. Its benefits are listed in Box 108-4. Phase I surgical intervention addresses all three levels of airway obstruction and includes nasal reconstruction, uvulopalatopharyngoplasty (UPPP), and a limited mandibular osteotomy with genioglossus advancement–hyoid myotomy and suspension (GAHMS). Radiofrequency (RF) technology using temperature-controlled RF (TCRF) or coblation in combination with GAHMS may be applied in phase I. The specific procedures are described later.
The protocol sequence for addressing nasal, oropharyngeal, retropalatal, or lingual obstruction during sleep is presented in Box 108-5. A patient needing intervention at any of these three anatomic levels should complete phase I surgery, in a sequence that would least jeopardize the upper airway during recovery, before moving on to phase II. This sequence should not interfere with the patient’s ability to use CPAP after surgery. Phase II represents skeletal reconstruction and consists of bimaxillary advancement (BMA), commonly referred to as maxillary and mandibular osteotomy (MMO). Combining phase I and phase II in one surgical procedure is discouraged because of an increased possibility of postoperative edema and upper airway compromise. In addition, phase II might not be necessary.
Box 108-5 Powell-Riley Protocol for Upper Airway Reconstruction
Phase I
Surgical Expectations
Because of concern about postoperative upper airway compromise secondary to surgery, we established a CPAP and surgical strategy19 All patients are encouraged to use CPAP, and those who have an AHI greater than 40 and an oxyhemoglobin desaturation (SaO2) less than 80% are required to use CPAP for at least 2 weeks before surgery, unless they cannot tolerate the device. Patients are also maintained on CPAP immediately after extubation and are encouraged to continue using this therapy during sleep until 2 weeks before the 4- to 6-month postoperative PSG reevaluation. Patients with very severe sleep apnea (AHI > 60, SaO2 < 70%), and significant comorbidities such as severe cardiac sequelae or morbid obesity with body mass index (BMI) of 40 who are intolerant of nasal CPAP should be considered for temporary or permanent tracheotomy.
Powell-Riley Phase I
Before discharge from the hospital, patients undergo monitoring, which includes assessment of at least oxyhemoglobin saturation over 2 nights as well as bedside fiberoptic nasopharyngoscopy while the patient is awake and sitting to evaluate the airway. If significant edema is noted, the patient remains in the hospital until discharge is considered safe. Four to 6 months after phase I (sufficient time for healing, weight stabilization, and neurologic equilibration), the patient undergoes a PSG, as well as a new clinical sleep assessment and clinical examination to assess outcomes.20 Patients who are incompletely treated may be candidates for phase II.
Defining Clinical Response
Early reporting methods defined patients who responded positively to surgery as those who demonstrated a 50% or greater reduction in the number of abnormal breathing events per hour of sleep. However, clinicians and researchers no longer consider such a definition valid. A more stringent surgical goal combines specific PSG parameters with quality-of-life issues, and the outcome should compare favorably with the results of medical management (specifically, positive airway pressure [PAP]). Our criteria for designating a positive surgical responder are presented in Box 108-6.