Figure 22.1. Effect of increasing intra-abdominal pressure (IAP) in closed and open-chest animals on intracranial pressure (ICP) and pleural pressure (PP). A. ICP: solid line closed chest, dotted line animals with thoracotomy. B. PP: solid line animals without sternotomy, dotted line animals with sternotomy. Modified from Bloomfield [9].
In a similar study in 5 pigs, Rosin et al. found no significant change from baseline in ICP when the IAP was 5 mmHg [22]. However, ICP and CPP were affected by increasing the IAP to 15 and 25 mmHg (p = 0.035 and 0.04 for ICP respectively). They concluded that low-pressure laparoscopy may reduce the adverse effects of pneumoperitoneum on ICP and CPP. It may therefore be advisable to use low pressures in laparoscopic surgery, especially when changes in ICP or renal perfusion may have significant clinical implications [22,23].
Hanel et al. measured cerebral hemodynamics in 10 pigs during laparoscopic insufflation to 12 mmHg [24]. Pneumoperitoneum significantly (p <0.05) increased CVP from 6.3 ± 2.1 to 11.1 ± 3.0 mmHg and sagittal sinus pressure from 8.0 ± 2.8 to 11.9 ± 3.0 mmHg. However, bilateral internal carotid artery blood flow (46.0 ± 7.4 vs. 47.7 ± 7.1 ml/100 g per minute), cortical cerebral blood flow, CBF (263 ± 115 vs. 259 ± 158 tissue perfusion units), and subcortical CBF (131 ± 145 vs. 133 ± 149 tissue perfusion units) did not change during CO2 pneumoperitoneum. The increases in sagittal sinus pressure are likely related to decreases in cerebral venous drainage caused by increases in IAP.
Other studies looked at the effect of combined abdominal and thoracic binding (ATB) on neurologic function during CPR: mean ICP was higher during ATB (46 ± 2 mmHg) than conventional CPR (20 ± 2 mmHg) [25]. However, the net brain perfusion pressure gradient (carotid artery pressure – ICP) was greater with ATB (14 ± 3 mmHg) than with conventional CPR (5 ± 0.4 mmHg). Cerebral blood flow was significantly greater during ATB CPR (32 ± 7% of pre-arrest cerebral flow) than during conventional CPR (3 ± 2%). They conclude that ATB CPR substantially improved brain perfusion by enhancing CPP in this experimental dog model. The same results were found by Ratjen et al. in lambs [26].
Palafox et al. looked at the effect of MAST suit on ICP in dogs [27]. During MAST inflation, they found a rise in ICP that mirrored the rise in CVP and never reached potentially harmful levels in the hemorrhaged animals and animals with a mass intracranial lesion. In the animals with cardiac tamponade and tension pneumothorax, inflation of the abdominal portion of the MAST suit produced a marked rise in CVP and ICP. This was altered by either relieving the lesion or reducing the pressure of the abdominal portion of the MAST suit.
Schob et al. compared the pathophysiological neurologic effects of carbon dioxide, helium and nitrous oxide pneumoperitoneum in 24 pigs [28]. The mean ICP increased significantly in all groups during peritoneal insufflation compared with the control group (p <0.005). The CO2-insufflated animals also showed a significant increase in PaCO2 (p <0.05) and ETCO2 (p <0.05), as well as a decrease in pH (p <0.05). After inflating the epidural balloon the ICP remained significantly higher in animals inflated with CO2 as compared with the He and N2O groups (p <0.05). They concluded that peritoneal insufflation with He and N2O resulted in a significantly less increase in ICP as compared with CO2. That difference was most likely due to a metabolically mediated increase in CPP. Further studies need to be conducted to determine the safety and efficacy of using He and N2O as inflation agents prior to attempting diagnostic or therapeutic laparoscopy in patients with potential closed head injuries.
In conclusion, these laboratory studies clearly showed that an IAP rise decreases the thoracic-abdominal compliance and increases intrathoracic pressures. This in turn reduces cerebral venous outflow, causing an ICP elevation.
22.2.2 Clinical Studies
Despite the convincing results of the previously mentioned animal studies, interest in this subject in human clinical research is very recent. In 1995, two early case reports were published that confirmed the results of the animal studies [29-31], but the first well conducted clinical study evaluating the relationship between IAP and ICP was published by Citerio et al. in 2001 [15]. They conducted a prospective non-randomised observational study to systematically measure the effect of increased IAP in 15 patients with traumatic brain injury. IAP was increased by positioning a soft 15-l bag of water on the patient’s abdomen. Naturally, patients could only be included in this study after the acute phase of their injury, when no intracranial hypertension was present. All patients were intubated and mechanically ventilated. Many parameters were measured: IAP, MAP, CVP, ICP, CPP, jugular bulb pressure (IJP), jugular bulb oxygen saturation and compliance of the respiratory system, divided into pulmonary and chest wall components. The authors found that placing a weight on the patient’s abdomen generated a significant increase in IAP and a concomitant and rapid increase in CVP, IJP and ICP. All these changes required only seconds to reach a plateau and remained stably increased until the IAP returned to baseline after the weight removal (Figure 22.2).
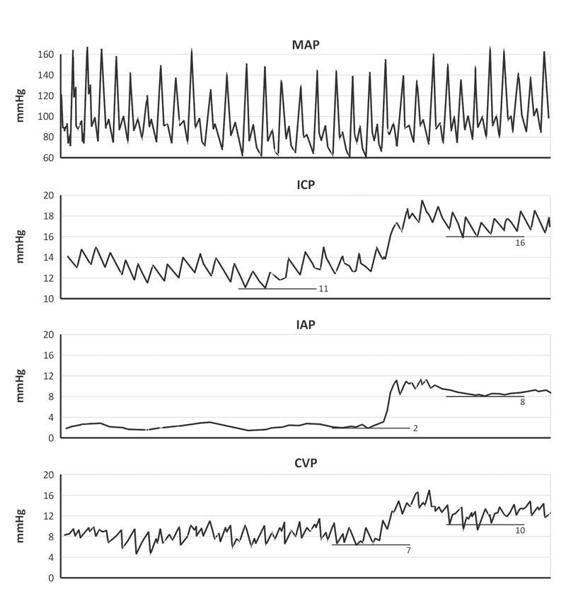
Figure 22.2. Effect of increased IAP, obtained with a weight application in a single patient. Computerized tracings are from top to bottom: MAP = mean arterial pressure; ICP = intracranial pressure; IAP = intra-abdominal pressure; CVP = central venous pressure. The IAP increased from 2 to 8 mmHg, this resulted in an increase in CVP from 7 to 10 mmHg and an increase in ICP from 11 to 16 mmHg. Hence the transmission from the abdomen to the thorax compartment can be calculated as ΔCVP (= 10 – 7) divided by ΔIAP (= 8 – 2) or thus 50%, and the transmission from the abdomen to the cranial compartment can be calculated as ΔICP (= 16 – 11) divided by ΔIAP (= 8 – 2) or thus 83%. Modified from [7,15].
Curiously, in this study a rise in MAP was observed, allowing for maintenance of a constant CPP in spite of increased ICP. This is not in concordance with the animal studies. This may be due to the lower levels of IAP generated in this human study compared to the animal studies and the use of different sedatives in animal and human studies or it may represent a true physiologic mechanism where increased intrathoracic pressure facilitates systolic ejection (although venous return is decreased). A second interesting finding from this study is that respiratory system compliance decreased significantly in all patients and this decrease was exclusively due to decreased compliance of the thoracic wall. Lung compliance did not change at all.
These findings confirm the hypothesis that IAH displaces the diaphragm upward, reducing the chest wall compliance and therefore respiratory system compliance. The pressure in the abdominal compartment (IAP) is directly transmitted to the thoracic compartment, where it increases intrathoracic pressures (CVP, esophageal pressure), and thereafter to the cerebral compartment (causing an increase in IJP and ICP). In other words, the ICP rise appears to be the result of an obstruction to the cerebral venous drainage, causing elevation of pressure in the intracranial compartment.
Deeren et al. found similar results in 11 patients with nontraumatic brain injury [20]. They found tight associations between IAP and ICP, ΔIAP and ΔICP, and ΔIAP and ΔCPP even at only slightly increased levels of IAP (Figure 22.3). In total eleven patients were studied with simultaneous IAP and ICP-monitoring because of ischemic (in four), hemorrhagic (in five) and metabolic (in two) encephalopathy. Two hundred fourteen consecutive paired data sets were compared. The mean IAP of each patient ranged from 3.8 to 11.8 mmHg, with a mean ICP from 6.7 to 15 mmHg and mean CPP from 70.8 to 123 mmHg. For ICP, the regression coefficient associated with IAP was 0.64 (standard error = 0.05; 95% confidence interval = 0.56 to 0.73; p <0.001, partial correlation = 0.70). For CPP, the regression coefficient associated with IAP was -1.36 (standard error = 0.3; 95% confidence interval = -1.94 to -0.78; p <0.001; partial correlation = -0.30). Cooke investigated key symptoms and signs of raised ICP in 39 patients after laparoscopic abdominal surgery and compared them with a control group of 33 patients after open operations [32]. They found that the incidence of headache and nausea was significantly higher in the laparoscopic group than in the control subjects and they concluded that these results could be explained by raised intracranial pressure exacerbated by the CO2 pneumoperitoneum, and that this effect is not mediated by raised expiratory CO2 levels intraoperatively.
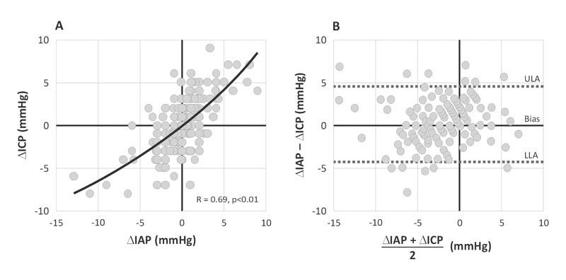
Figure 22.3. (A). Regression analysis between ΔIAP versus ΔICP (positive correlation). (B). Bland-Altman plot of ΔIAP and ΔICP.
IAP = intraabdominal pressure; ICP = intracranial pressure; LLA = lower limit of agreement; ULA = upper limit of agreement.
In an ophtamologic journal, Neville et al. have published a study in which 15 patients with normal opening pressures on lumbar puncture who were referred for CSF analysis, were asked to artificially increase their IAP through a Valsalva maneuver [33]. All patients were able to elevate their CSF pressure to values above 25 cmH2O and one patient achieved a value of 47 cmH2O! The authors were prompted to perform this study after the observation that patients were referred for fundoscopy due to increased CSF pressure while they had no symptoms of intracranial hypertension.
Recently several reports have been published alluding to neurologic deterioration related to increased IAP in patients with ventriculoperitoneal shunt (VPS) dysfunction [34-36]. These reports provide anecdotal evidence that transient and easily reversible increases in the IAP of adults with VPS can result in dysfunction. Although it may not be practical to delay shunt revision while attempting to correct constipation, meteorism, ileus, or small bowel obstruction, clinicians treating patients with these abdominal conditions should be aware that they could cause transient VPS failure [37]. Martinez-Lage et al. describe constipation as an under-appreciated cause of VPS malfunction in children and advise pediatric neurosurgeons to investigate the abdomen first before considering surgical valve revision of the VPS [38]. In a similar way colonoscopy can increase ICP [39].
Several reports also pointed towards obesity as a cause of increased IAP leading to idiopathic ICH and pseudotumor cerebri [40-43]. Interestingly, negative abdominal pressure, weight loss and bariatric surgery have been demonstrated to reduce the clinical signs of ICH (headache, blurred vision, etc.) and ICP [44-48]. Furthermore recent weight gain may be associated with relapses of idiopathic ICH [49]. Obesity associated sleep apnea may also be associated with idiopathic ICH [50, 51]. Sahuquillo et al. performed a study in 60 patients requiring a VPS for hydrocephalus [52]. They recorded BMI and measured IAP during the shunt surgery and found that there was a linear relationship between IAP and BMI. They conclude that the assumption that IAP is close to 0 mmHg does not apply in obese or overweight patients and neurosurgeons should take IAP into account when selecting both the most adequate differential pressure valve to be implanted and in which distal cavity to place the distal catheter to avoid shunt underdrainage induced by high IAP.
22.3 Clinical Importance of IAH in Patients at Risk for ICH
According to the previously mentioned modified Monro-Kellie doctrine, ICP remains constant over a relatively wide range of intracranial volume. In this plateau phase, an increase in intracranial volume due to e.g. cerebral edema, hematoma or mass lesion, will be compensated by the evacuation of cerebrospinal fluid through the foramen magnum at the base of the skull. However, when these compensation mechanisms are exhausted at higher intracranial volumes, any small increase in volume produces a marked increase in ICP rapidly leading to ICH (Figure 22.4).
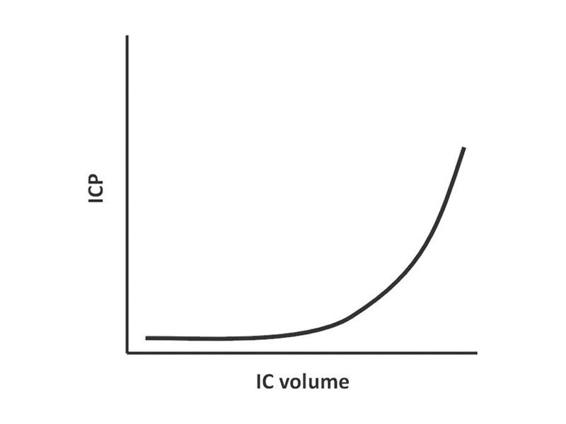
Figure 22.4. Relationship between intracranial volume (IC volume) and intracranial pressure (ICP) according to the Monro-Kellie doctrine.
When we combine this concept with the findings of the previously mentioned experimental and clinical studies we can speculate that in the presence of ICH the effect of increased IAP probably induces a more profound and harmful increase in ICP due to a different starting ICP position on the intracranial PV curve and different strain on the compensatory capacity. Therefore, it is crucial to measure IAP in patients with ICH at risk for IAH (or vice versa).
These findings are of great importance for patient care, for example in trauma patients, since abdominal trauma is commonly associated with head trauma and therefore patients with combined ICH and IAH may be more common in the ICU population than previously thought. Scalea et al. recently published a study describing 102 consecutive patients with severe TBI who underwent decompressive craniectomy (DC) for intractable intracranial hypertension [53]. Twenty-four patients in this series also had decompressive laparotomy (DL) due to what the authors describe as multiple compartment syndrome (MCS). The found that both DC and DL reduce ICP and can be used in sequence and conclude that MCS should be considered in multiply injured patients with increased ICP that does not respond to therapy. Joseph et al. performed a retrospective study and found that in their institution 17 patients in a period of 3 ½ years underwent DL for refractory intracranial hypertension [10]. All had failed maximal therapy including 14 who had had DC. Mean ICP was 30 ± 8.1 mmHg (range 20-40 mmHg) before decompression. No patients had evidence of abdominal compartment syndrome (ACS). Before decompression mean IAP was 27.5 ± 5.2 mmHg (range 21-35 mmHg). After abdominal decompression ICP dropped precipitously by at least 10 mmHg to a mean of 17.5 ± 3.2 mmHg (range 10-25 mmHg). The authors conclude that decompressive laparotomy can be a useful adjunct in the treatment of ICH failing maximal therapy following TBI.
Another obvious risk population are patients with existing VPS [52]. In the event of neurologic deterioration in these patients during critical illness, IAP should be monitored and IAH should be treated promptly. IAP should also be monitored in critically ill, sedated and mechanically ventilated patients with VPS since neurologic examination is unreliable in these circumstances. Table 22.1 summarizes the neurologic effects of increased IAP.
Stay updated, free articles. Join our Telegram channel
Full access? Get Clinical Tree
 Get Clinical Tree app for offline access
Get Clinical Tree app for offline access

|



