Figure 70.1. Pro-con red blood cell transfusion.
Transfusion of red blood cells, may, however be associated with negative effects. The delivery of oxygen to the tissue can be lower than expected, and the transfused red blood cells units can enhance an inflammatory response with possible impairment of microcirculation.
Clinical studies have demonstrated an association between red blood cell transfusion and increased mortality and morbidity in general critical care patients. These results cannot be directly transferred to the neurocritical care patient. For the neurocritical care patient, we still lack convincing studies of effects on outcome of red blood cell transfusion.
This chapter will discuss:
- The normal physiology concerning red blood cells and hemoglobin.
- The beneficial and harmful effects of red blood cell transfusion.
- The target levels of hemoglobin in patients with neurological/neurosurgical acute injury such as traumatic brain injury, subarachnoid hemorrhage and ischemic stroke.
70.2 Red Blood Cells and Hemoglobin
Red blood cells are biconcave discs with a diameter of about 7.5 µm. The capillary diameter is 3-8 µm, which implies that the red blood cells must possess the ability of deformation to pass the capillary network and deliver oxygen to the tissues. The red blood cells carry hemoglobin, and hemoglobin has four important functions: 1) to facilitate O2 transport; 2) to facilitate CO2 transport; 3) to act as a buffer; 4) to transport NO.
70.2.1 Oxygen (O2) Transport
The total sum of the oxygen delivered to the whole body or to specific organs, is the product of blood flow and arterial oxygen content. The arterial oxygen content is mainly dependent on the hemoglobin level. More than 99% of the oxygen is transported by hemoglobin. Oxygen delivery must be sufficient for the metabolic demand of the tissues. Oxygen is not stored in tissues and must be delivered continuously in adequate amounts for avoidance of anaerobic metabolism and production of lactate. The cerebral metabolic rate in the human brain is on average 3.5 ml/100 g brain/minute. This represents approximately 20% of the total body resting oxygen consumption. Aerobic metabolism is the primary energy source for the brain and the brain is dependent on a continuous delivery of substrate, including glucose and oxygen. The brain is thus extremely sensitive to decreased delivery of oxygen.
The affinity for oxygen to hemoglobin is an important factor for oxygen delivery to the tissues. Increased affinity means that less oxygen is liberated in the tissues.
Important factors affecting the affinity for oxygen in hemoglobin are: pH, temperature, and concentration of 2,3-diphosphoglycerate (2,3-DPG).
2,3-diphosphoglycerate is plentiful in red blood cells and it enhances the ability of red blood cells to release oxygen in tissues. For example, high altitude triggers a rise in 2,3-diphosphoglycerate concentration in red blood cells, which subsequently increases the release and availability of oxygen in tissues.
Factor | Effect |
Decrease in pH | Decrease in oxygen affinity |
Increase in temperature | Decrease in oxygen affinity |
Decrease in 2,3-diphosphoglycerate | Increase in oxygen affinity |
Table 70.1. Factors affecting the affinity for oxygen in hemoglobin.
The uptake of oxygen in the tissue is less in the alcalotic than in the acidotic patient.
A fall in 2,3-diphosphoglycerate is associated with an increased hemoglobin affinity for oxygen, resulting in less oxygen liberated to the tissues. In stored red blood cells 2,3-diphosphoglycerate is decreased within 48 hours of storage.
The delivery of oxygen to a tissue increases with raised hematocrit up to a certain level. Above that level there is a net reduction in oxygen delivery most likely due to the simultaneous increase in blood viscosity. While a normal hematocrit of 35-45% is optimal in healthy individuals, it may vary in critically ill patients due to different pathophysiological conditions depending on type of illness and quality of the blood transfused (see below).
In summary, the brain is dependent on a continuous delivery of oxygen in order to survive. Hemoglobin carries more than 99% of the oxygen. The optimal hematocrit may vary in patients with different pathophysiological conditions.
70.2.2 Carbon Dioxide (CO2) Transport
CO2 is the major end product of oxidative metabolism. Since CO2 is able to convert into carbonic acid when hydrated, and accumulation of carbonic acid, H2CO3, can produce problems in the tissue, an effective elimination of CO2 from the tissue is essential. The focal point of CO2 transport is the hydration reaction; CO2 + H2O=H2CO3. The rate of this reaction is fastest in the red blood cell by the help of carbonic anhydrase. In the red blood cell, H2CO3, dissolves into hydrogen ion, H+ and bicarbonate, HCO3–. The H+ generated in the red blood cell is buffered by hemoglobin and HCO3–, is pumped back into plasma in exchange for chloride. In summary, hemoglobin plays a major role in the elimination of carbon dioxide (CO2) from the tissue.
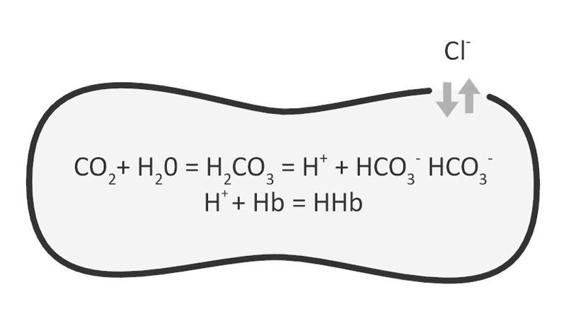
Figure 70.2. Transport and reactions of CO2 in the red blood cell. In the red blood cell, the reaction, CO2 + H20=H2CO3, is facilitated by carbonic anhydrase. Carbonic anhydrase is only present in the red blood cell. Carbonic acid, H2CO3, dissociates into hydrogen, H+, and bicarbonate, HCO3–. The hydrogen ion is buffered by hemoglobin, Hb, and forms HHb. A large fraction of the bicarbonate produced is pumped back into plasma in exchange for chloride, Cl–.
70.2.3 Hemoglobin as a Buffer
Hemoglobin has an important role in CO2 transport. By transporting CO2, hemoglobin acts as an important buffer for the hydrogen ions produced by the hydration of CO2 (see above, CO2 transport). Hemoglobin in blood has six times the buffering capacity of the plasma proteins.
70.2.4 Transport of Nitric Oxide by Hemoglobin
Nitric oxide is a potent vasodilator that is produced in endothelial cells and acts more or less exclusively at its site of synthesis. New evidence suggests that red blood cells distribute nitric oxide (NO), thereby matching blood flow to tissue oxygen demand. This mechanism could provide an explanation for physiological matching of blood flow to tissue oxygen levels, by mediating dilatation of blood vessels under low oxygen conditions, and constriction of blood vessels when oxygen levels are high.
In summary, nitric oxide can be transported by hemoglobin. Hemoglobin can thus be an important factor for physiological matching of blood flow to metabolic demands.
70.2.5 Red Blood Cells and Blood Volume
The red blood cells comprise about 40% of normal blood volume. The red blood cells are, therefore, of importance to maintain normovolemia. In a pathophysiological situation with increased transcapillary leakage it will, therefore, be more difficult to maintain normovolemia at a low than at a normal hematocrit. In summary, the red blood cells contribute to maintenance of normovolemia.
70.3 Transfused Red Blood Cells
A main goal of red blood cell transfusion is to increase the hemoglobin concentration, and thereby improve O2 delivery to tissues.
The efficacy of stored red blood cells to maintain the biological activity of red blood cells has been questioned. Transfusion of large amounts of stored red blood cells can possibly lead to adverse clinical consequences. Transfused red blood cells may be ineffective transporters of oxygen, especially in compromised critically ill patients who have microcirculatory abnormalities from a number of pathologic processes and the content of leukocytes (see below) and other inflammatory components can trigger inflammation in a compromised host.
70.3.1 Red Blood Cells and Storage
During storage, red blood cells undergo changes in morphology, associated with biochemical and biomechanical changes, collectively referred to as storage lesions. There is strong laboratory evidence suggesting that prolonged red blood cell storage may be deleterious and clinical studies have reported an association between prolonged storage and adverse clinical outcome. Storage lesions ultimately include loss of deformability. Loss of deformability in combination with increased interactions with vascular endothelium compromise microvascular flow. The unloading of oxygen to the tissues will be impaired by the well-documented depletion of 2,3-DPG in red blood cells that occur after storage for more than 48 hours.
Characteristic | Effect |
Compromised deformability | Microvascular obstruction |
Depletion of 2,3-diphosphoglycerate | Increased affinity for O2, reduced ability to unload O2 |
Pro-inflammatory effects | Immunodysfunction |
Depletion of NO | Vasoconstriction |
Table 70.2. Possible negative characteristics of stored transfused red blood cells.
70.3.2 Transfused Red Blood Cells and Immunodysfunction
Perhaps the most negative characteristic of transfused red blood cells units is the content of white blood cells. The immunomodulating effects of red blood cell transfusion have mainly been attributed to leukocytes and inflammatory mediators in the supernatant liquid, denoted as transfusion related immunomodulation (TRIM). TRIM is supposed to be mediated by white blood cells in the transfused red blood cell unit. The donated white blood cells may either down-regulate the immune function of the recipient or trigger TRIM effects by soluble mediators released in the red blood cell unit during storage. Randomized controlled trials comparing the risk of postoperative infection or short-term (three months) mortality between non-leukocyte depleted versus leukocyte depleted red blood cell transfusions have reported conflicting results.
A clinical situation where randomized controlled trials clearly have revealed that the use of leukocyte depleted red blood cell transfusion reduces short-term mortality is in cardiac surgery patients. Many blood transfusion services have added universal leukocyte depletion to the routine processing of all blood components and the majority of red blood cell transfusions in Western Europe and North America are today depleted of white blood cells. Noteworthy, the published studies on harmful effects of red blood cell transfusion in critical ill patients have used mainly non-leukocyte depleted blood.
In summary, storage of red blood cells can alter the biological effects negatively. Content of leukocytes in transfused red blood cells units can induce immunodysfunction.
70.4 Red Blood Cell Transfusion in Neurocritical Care Patients
There are studies in critically ill patients indicating that a liberal transfusion policy is ineffective and even potentially harmful. Interestingly, a well-known and world wide accepted treatment approach in severe sepsis and septic shock, i.e. the early goal directed therapy, contains a recommendation of red blood cells transfusion to achieve a hematocrit of at least 30%, if the central venous saturation is under 70%. In the treatment group 68% of the patients received red blood cell transfusion compared to 45% in the control group. The inhospital mortality in the treatment group was 31% versus 47% in the control group. This indicates that critically ill patients with severe sepsis had a lower mortality if transfused to a hematocrit of at least 30% in the presence of hypoxemia.
The applicability of a restricted transfusion policy to neurocritical care patients has been questioned, given that impaired oxygen delivery is an important cause of secondary brain injury. The brain is more than any other organ depending on a continuous supply of oxygen and only moderate reductions in oxygen delivery to an injured brain could lead to cerebral hypoxia.
Most of the studies concerning the level of hemoglobin and outcome after neurocritical care are prospective or retrospective cohort studies and therefore despite multivariate analysis, the relationship between red blood cell transfusion and increased adverse events may still be a result of confounding factors. There are studies that have reported an association between red blood cell transfusion and increased mortality, but it has to be taken into consideration that the amount of blood transfused may reflect injury severity rather than the blood transfusion per se.
No evidence based optimal hemoglobin level exists for patients receiving neurocritical care. Randomized controlled studies regarding the optimal hemoglobin concentration in neurocritical care patients are lacking.
70.4.1 Traumatic Brain Injury
Patients with traumatic brain injury have been thought to be particularly susceptible to injury from anemia, due to the well-documented association of increased mortality and worsening functional outcome in the presence of hypotension and hypoxia. Few studies have investigated the effects of anemia in patients with traumatic brain injury. Retrospective studies have revealed that any period of anemia in traumatic brain injury is associated with a significant increase in morbidity and mortality, but also that red blood cell transfusion in traumatic brain injury patients, whether or not anemia is present, is an independent risk factor for complications and mortality. The Transfusions Requirements in Critical Care, TRICC, trial was a randomized controlled clinic trial investigating liberal (hemoglobin level 10.0 g/dl to 12.0 g/dl) versus restrictive (hemoglobin level 7.0 g/dl to 9.0 g/dl) transfusion policy in critically ill patients. The results from the trial were published in New England Journal of Medicine in 1999. Primary outcome in the TRICC trial was mortality at 30 days. Although overall results from the original TRICC trial suggested that a restrictive red blood cell transfusion strategy was safe, it was suggested that subgroups of patients may exist, who are especially vulnerable to effects of anemia. A subgroup analysis from the TRICC trial in patients with traumatic brain injury was published in 2006, and even though no significant difference in mortality between a restrictive and a more liberal transfusion strategy could be detected, the mortality rate at 30 days was 13% in the liberal transfusion group and 17% in the restrictive transfusion group. Thus, the liberal transfusions strategy seemed to be at least as safe as the more restrictive transfusion policy. The subanalysis of the TRICC trial concerning patients with traumatic brain injury could not recommend a specific transfusion trigger level in those patients, but the authors concluded that the brain could be an organ that is especially vulnerable to adverse effects of anemia.
It may also be of importance that during the TRICC trial, red blood cells units were not universally leukocyte depleted. Perhaps physiological indications of the need for red blood cell transfusion other than the hemoglobin level could be triggers for transfusion of red blood cells in the future. Recent studies have documented that red blood cell transfusion increases brain oxygenation, measured by brain tissue partial pressure of oxygen (PtiO2) catheters, in most patients with traumatic brain injury and subarachnoid hemorrhage. A limitation of these studies is that PtiO2 might not be an accurate tool to demonstrate the effect of transfusion on oxygen uptake by the tissues.
Normalization of the hemoglobin level would theoretically improve the oxygen transport capacity to injured areas of the traumatized brain, in spite of limitations in this capacity in stored red blood cells. Arguments can be given that patients with a traumatic brain injury would benefit from keeping a normal hemoglobin level in order to optimize oxygen delivery to the traumatized brain. This approach has been adopted in the Lund-concept for treatment of severe traumatic brain injury, the outcome data presented with this approach is good, even though of course this can not be attributed only to the hemoglobin level.
More prospective investigations are required to determine the effects of anemia and the potential benefits and optimal indications for red blood cell transfusion in patients with traumatic brain injury. The hemoglobin threshold may not be identified as the treatment goal, rather more sophisticated methods of neuromonitoring may be necessary to guide the need for red blood cell transfusion.
In summary, no evidence based hemoglobin level can be recommended for patients with traumatic brain injury, but the existing data taken together support a hemoglobin level near normal, that is, a target hemoglobin value around 110 g/l.
70.4.2 Subarachnoid Hemorrhage
Patients with subarachnoid hemorrhage, especially those with a manifest cerebral vasospasm, are vulnerable to hypoxic episodes. Limited oxygen delivery due to anemia, could have deleterious effects. The optimal transfusion threshold for patients suffering from non-traumatic subarachnoid hemorrhage is debatable. Current practice most often uses a liberal approach. Anemia after subarachnoid hemorrhage has been identified as an independent predictor of infarction, death and dependency. Retrospective studies have reported that subarachnoid hemorrhage patients with higher initial and mean hemoglobin value had improved outcome. Anemia seems to be a predictor of adverse outcome in subarachnoid hemorrhage patients, even when baseline differences in clinical and radiographic severity are taken into account. However, data suggesting an association between red blood cell transfusion and adverse outcome after subarachnoid hemorrhage have also been reported. The triple-H (hypervolemia, hypertension, hemodilution) therapy, used for treatment of cerebral vasospasm, could be one reason for anemia in patients with subarachnoid hemorrhage.
It remains unclear whether anemia after subarachnoid hemorrhage reflects general illness severity or whether the treatment for anemia, red blood cell transfusion, directly contributes to poor outcome. Randomized trials that compare liberal and restrictive transfusion strategies in patients with subarachnoid hemorrhage are needed.
In summary, no evidence based hemoglobin level can be recommended for patients with non-traumatic subarachnoid hemorrhage, but the existing data supports a near normal hemoglobin level. In patients with non-traumatic subarachnoid hemorrhage, the target hemoglobin value could be 100-110 g/l.
70.4.3 Ischemic Stroke
Patients with ischemic stroke have an injured brain that theoretically would benefit from a constant and normal delivery of oxygen. The effect of red blood cell transfusion on outcome in patients with ischemic stroke has been scarcely investigated. Elevated hematocrit has been associated with greater infarct size, early mortality and major disability in patients with ischemic stroke. Elevated hematocrit could be caused by decreased fluid intake and hypovolemia. Hemodilution on the other side has not been proven to improve survival or functional outcome after ischemic stroke. A too low hematocrit may decrease oxygen transport to the injured brain and a too high hematocrit may increase blood viscosity and thus impair microcirculation.
In summary, no evidence based hemoglobin level can be recommended for patients with ischemic stroke. Theoretically normovolemia and a near normal hemoglobin level would be of value for avoiding secondary insults in the injured brain.
70.5 Conclusion
Red blood cell transfusion is a potentially life-saving therapy in order to maintain oxygen delivery to an injured brain. Transfused red blood cells may not only increase oxygen delivery but also possibly influence tissue oxygenation by the capacity to regulate microvascular blood flow. The efficacy of stored red blood cells to maintain their biological activity has been questioned, due to storage damages.
Patients with traumatic brain injury may, based on the data presented, benefit from hemoglobin levels near normal. In patients with traumatic brain injury, the target hemoglobin value could be around 110 g/l.
Anemia has been identified as an independent risk factor for unfavourable outcome after subarachnoid hemorrhage. The existing data support a near normal hemoglobin level. In patients with non-traumatic subarachnoid hemorrhage, the target hemoglobin value could be 100-110 g/l.
The optimal hemoglobin level in patients with ischemic stroke is barely investigated. Theoretically would normovolemia and a near normal hemoglobin level be of value for avoiding secondary insults in the injured brain.
General References
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree






