INTRODUCTION
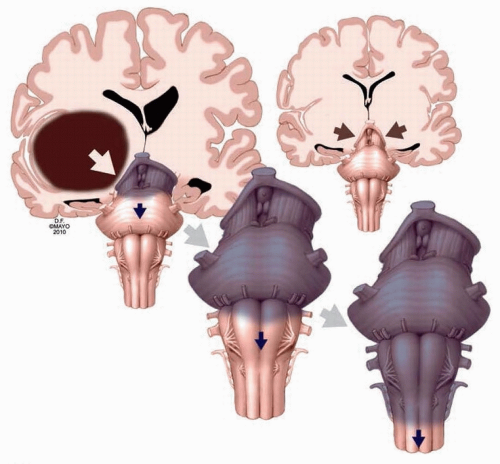
Brain death is the preferred term to summarize an apneic patient with not only irreversible coma from a massive brain injury but loss of all brain stem reflexes and uncontrolled diuresis and hypotension from loss of vascular tone. Nothing else should explain this condition and characteristically, it is due to an acute catastrophic bihemispheric and diencephalic injury. Brain death remains uncommon because such progression would require not only involvement of both hemispheres of the brain but also loss of brain stem function. Typically, this clinical situation would be an acute massive hemispheric lesion (e.g., cerebral hemorrhage) compressing and sequentially damaging the mesencephalon, pons, and medulla oblongata (
Fig. 19.1). The brain stem is very resilient to injury and it would take a substantial shift (from mass effect) or poor perfusion (from basilar artery occlusion or massively increased intracranial pressure) for it to get permanently damaged. This core neurologic principle—the brain stem to be the last brain structure to go out of function—is the most important attribute to our understanding of brain death. Once brain stem function is lost, breathing stops first and the heart soon thereafter. If in the acute phase the patient can be intubated, placed on a mechanical ventilator, sufficiently oxygenated, fluid resuscitated, and vasopressors and vasopressin added, this agonal sequence can potentially be prevented.
Once an untreatable catastrophic neurologic structural injury has been proven and the brain stem reflexes have disappeared, recovery does not occur, and there is no known effective medical or surgical intervention. Irreversibility is determined by this diagnosis and involves testing of absent motor responses, loss of all brain stem reflexes, and observation of apnea after a CO2 challenge in a patient temporarily detached from the ventilator.
The determination of brain death is a fundamental skill for the neurologist but experience could be waning. In many regions, the incidence of brain death has declined over the last decade. It is possible that family members may decide to withdraw when the situation is already hopeless and do not want to wait any further. Improved neurosurgical care, in particular early decompressive craniotomy, may also have contributed.
Brain death determination is relatively straightforward. Physicians should work through a set of criteria and not be swayed by supposedly quicker options. This chapter provides ways on how to proceed with this evaluation and how to prevent common pitfalls.
THE CLINICAL DIAGNOSIS OF BRAIN DEATH
Declaring a patient brain dead can be considered in a comatose patient with a major destructive brain injury, loss of at least three brain stem reflexes, and no evidence of a breathing effort. A more formal assessment, however, may only proceed when the patient worsens even more, when all medical or neurosurgical interventions are futile, and when there are no confounding factors or alternative explanations. The clinical examination starts when
a patient with a massive acute brain injury has no motor response to pain, fails to grimace to pain, has absent brain stem reflexes, and does not trigger the ventilator. Commonly, the patient has become hypotensive and polyuric from diabetes insipidus and has been placed on vasopressors. Using this starting point, many patients will fulfill the criteria of brain death after detailed neurologic examination. With any other (earlier) starting point, there is a greater chance that there will be retained brain stem function and even spontaneous breathing when the patient is briefly disconnected.
DETERMINE THE CAUSE OF BRAIN DEATH
Brain death evaluation involves several sequential steps. First, there is nothing more important than to be very certain that all possible confounding factors have been excluded. This implies there can be no lingering effects of prior sedation, other confounding medications, or prior use of illegal drugs or alcohol. A reasonable guideline is to calculate five to seven times the drug’s elimination half-life in hours and allow that time to pass before clinical examination is performed. Examples of long elimination half-life drugs are phenobarbital (100 hours), diazepam (40 hours), amitriptyline (24 hours), primidone (20 hours), and lorazepam (15 hours). A commonly used short-acting benzodiazepine is midazolam, but the elimination may still take 3 hours. Prior use of therapeutic hypothermia may substantially slow down the metabolism of medications such as lorazepam and fentanyl used to support the procedure. Substantial alcohol levels should be excluded, but alcohol content below the legal alcohol limit for driving (blood alcohol content 0.08%) is acceptable to determine brain death. Absence of neuromuscular blockade (defined by the presence of four twitches with a train of four with maximal ulnar nerve stimulation) should be demonstrated but is likely if the patient has tendon reflexes (or breathes). Furthermore, absence of severe electrolyte, acid-base, or endocrine disturbances (defined by marked acidosis or any substantial deviation from the normal values) should be documented. A core temperature greater than 32°C must be present, but preferably, there should be near normothermia (36°C to 37°C), which can be achieved with a warming blanket—assuming the patient is not a victim of environmental severe hypothermia. Systolic blood pressure should be greater than 90 mm Hg because pupil size and light reflex can fully disappear with lower values. Only after these confounders have been addressed should a more formal examination proceed.
IMAGING CONFIRMATION OF BRAIN DEATH
Next, the computed tomography (CT) scan should be carefully reviewed and expectedly demonstrate massive brain destruction. Abnormalities may include a large mass with brain tissue shift, multiple hemorrhagic lesions, or diffuse cerebral edema with obliteration of basal cisterns. CT scan can be initially normal if the patient has been imaged very early after cardiopulmonary arrest. However, in the patients with anoxic-ischemic encephalopathy who eventually fulfill these criteria, brain edema or marked hypodensities in thalami, caudate nuclei, and basal ganglia are typically seen on a repeat CT scan. A normal CT should under no circumstances be acceptable. In some cases of cardiac arrest, a magnetic resonance imaging can be helpful to demonstrate the full extent of ischemic tissue damage compatible with brain death.