Preparations with the least abuse potential and side effects should be utilized. Agents containing an anti-inflammatory/acetaminophen, to decrease the dose of opioid required for analgesia, should be considered first-line drugs. Careful consideration must be given to the total daily dosage of acetaminophen/NSAIDS when using a combination drug. Common side effects of opioids include respiratory depression, sedation, cognitive impairment, liver dysfunction, and testosterone deficiency in men with long-term use. Tolerance, addiction potential, and withdrawal from sudden cessation are also common.
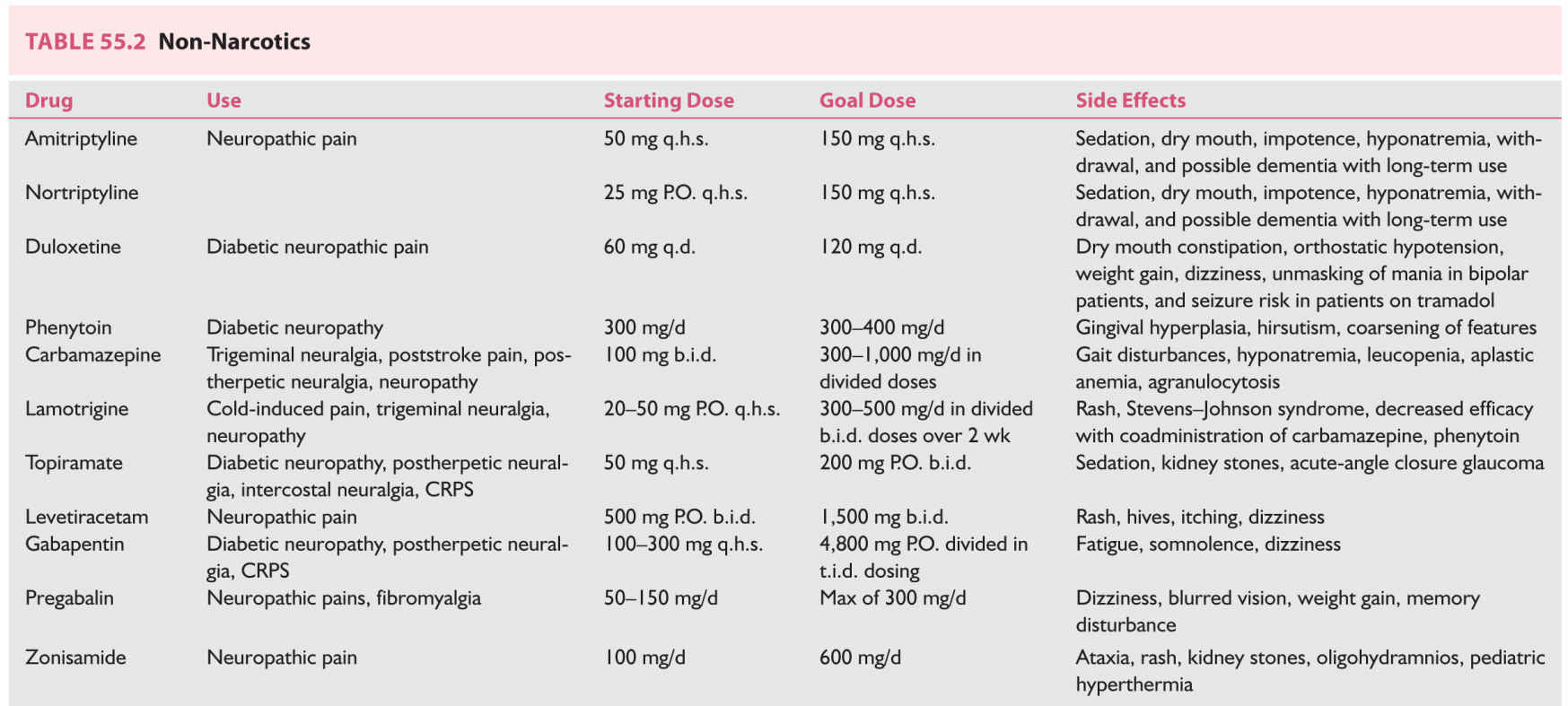
3. Antidepressants. Antidepressants are also utilized in chronic pain management. Mechanisms of action include a direct antidepressant effect, a decrease in synaptic transmission, and enhancement of the action of endogenous opioids (Table 55.2). Antidepressants are classified on the basis of their mechanism of action. Tricyclic antidepressants include amitriptyline and nortriptyline. Common side effects include dry mouth, sedation, sexual dysfunction, hyponatremia, and risk of withdrawal if discontinued. Serotonin norepinephrine reuptake inhibitors (SSNRI), another class of antidepressants, include fluoxetine and duloxetine. Duloxetine is a dopamine, serotonin, and norepinephrine reuptake inhibitor. These drugs can be used for diabetic neuropathy and are contraindicated in patients with concomitant monoamine oxidase inhibitor (MAOI) therapy, hypersensitivity, and narrow angle glaucoma. Common side effects of SSNRI include dry mouth, constipation, orthostatic hypotension, weight gain, dizziness, unmasking of mania in bipolar patients, and risk of seizures in patients receiving tramadol. Other agents in this class include milnacipran, levomilnacipran, and desvenlafaxine. Lower doses of antidepressants are usually used for analgesia.
4. Antiepileptic drugs (AEDs). AEDs are also used in the treatment of chronic pain. AEDs increase inhibitory neurotransmission, decrease excitatory transmission, and block ion channels. AEDs are most effective for managing neuropathic pain (Table 55.2).
Phenytoin is used to treat diabetic neuropathy; treatment is initiated at 300 mg/day, with increased dosage as needed. Side effects include gingival hyperplasia, hirsutism, acne, and coarsening of facial features. Phenytoin activates the P-450 enzyme system in the liver, resulting in a decreased efficacy of mexiletine, haloperidol, and carbamazepine. Coadministration with antidepressants may lead to increased blood levels of phenytoin.
Carbamazepine is used for trigeminal neuralgia (tic douloureux), poststroke pain, postherpetic neuralgia, and diabetic neuropathy. Carbamazepine is believed to act via central and peripheral mechanisms, selectively targeting actively firing C and A delta fibers. Treatment is started at 100 mg twice a day and titrated to effect; typical dose range is 300 to 1,000 mg/day in divided doses. Side effects include gait imbalance, hyponatremia, leukopenia, aplastic anemia, and agranulocytosis. Because of these serious, though rare, side effects, carbamazepine is seldom used as a first-line agent for chronic pain. As a result of these potential hematologic alterations, blood tests are recommended every 2 to 4 months. Oxcarbazepine, an analog of carbamazepine, is less likely to cause CNS/hematologic alterations. Side effects include hyponatremia, sedation, and dizziness.
Lamotrigine prevents the release of glutamate in addition to blocking active sodium channels. Lamotrigine is used for cold-induced discomfort, trigeminal neuralgia, and diabetic and HIV neuropathy. Starting dose is 20 to 50 mg at bedtime, slowly increased to 300 to 500 mg/day in twice daily divided doses (over 2 weeks). A rash, the most common side effect, is commonly seen in pediatric patients, those receiving valproic acid, or patients receiving rapid titration of lamotrigine. The rash may also lead to Stevens–Johnson syndrome. Concomitant administration of carbamazepine and phenytoin leads to a decreased efficacy of lamotrigine.
Topiramate acts at both sodium and calcium channels, enhancing the action of gamma-aminobutyric acid (GABA) and inhibiting AMPA receptors. Topiramate is used in diabetic neuropathy, postherpetic and intercostal neuralgia, and complex regional pain syndrome (CRPS) (see Chapter 56). Initial dosing is begun at 50 mg at bedtime, increasing to 200 mg twice daily. Side effects include sedation, kidney stones, and acute-angle closure glaucoma. Another channel-blocking agent, levetiracetam, is started at 500 mg P.O. twice daily and adjusted to a target of 1,500 mg twice a day. Side effects include rash, hives, itching, and dizziness.
5. Local anesthetics. Lidocaine, available in salve and patch (5%) forms, is used for postherpetic neuralgia, postthoracotomy pain, intercostal neuralgia, and fibromyalgia. The patch is used 12 hours/day and may be cut to size and shape. Lidocaine side effects include bradycardia, dizziness (at plasma level of 10 mg/mL), cardiac depression (at 20 to 25 mg/mL plasma level), blurred vision, and seizures. Mexiletine, an oral analog of lidocaine, is used for poststroke pain, myotonia, spasticity, and diabetic neuropathy. Treatment is initiated at 150 mg/day up to a goal of 300 to 450 mg/day. The side-effect profile is similar to lidocaine.
6. Calcium channel blockers. Gabapentin is a membrane stabilizer that binds at the alpha-2-delta subunit of the L-calcium channel (Table 55.2). Gabapentin is used for many neuropathic pain states including diabetic neuropathy, postherpetic neuralgia, and CRPS. Treatment is started at 100 to 300 mg at bedtime, increasing to 3,600 mg/day divided in three doses. Common side effects include fatigue, somnolence, and dizziness.
Pregabalin also binds to the alpha-2-delta subunit at voltage-dependent calcium channels. Pregabalin is used for neuropathic pain states and for fibromyalgia. The starting dose is 50 to 150 mg/day titrated to 300 mg/day for neuropathic pain states; for fibromyalgia, the maximum daily dose is 300 mg/day. Side effects include dizziness, blurred vision, weight gain, and diminished cognition.
Zonisamide acts at T calcium and sodium channels. This drug increases GABA release. Zonisamide is started at 100 mg daily and increased after 2 weeks by 200 mg/week for a goal of 600 mg/day. Common side effects include ataxia, rash, kidney stones, oligohydramnios, and pediatric hyperthermia.
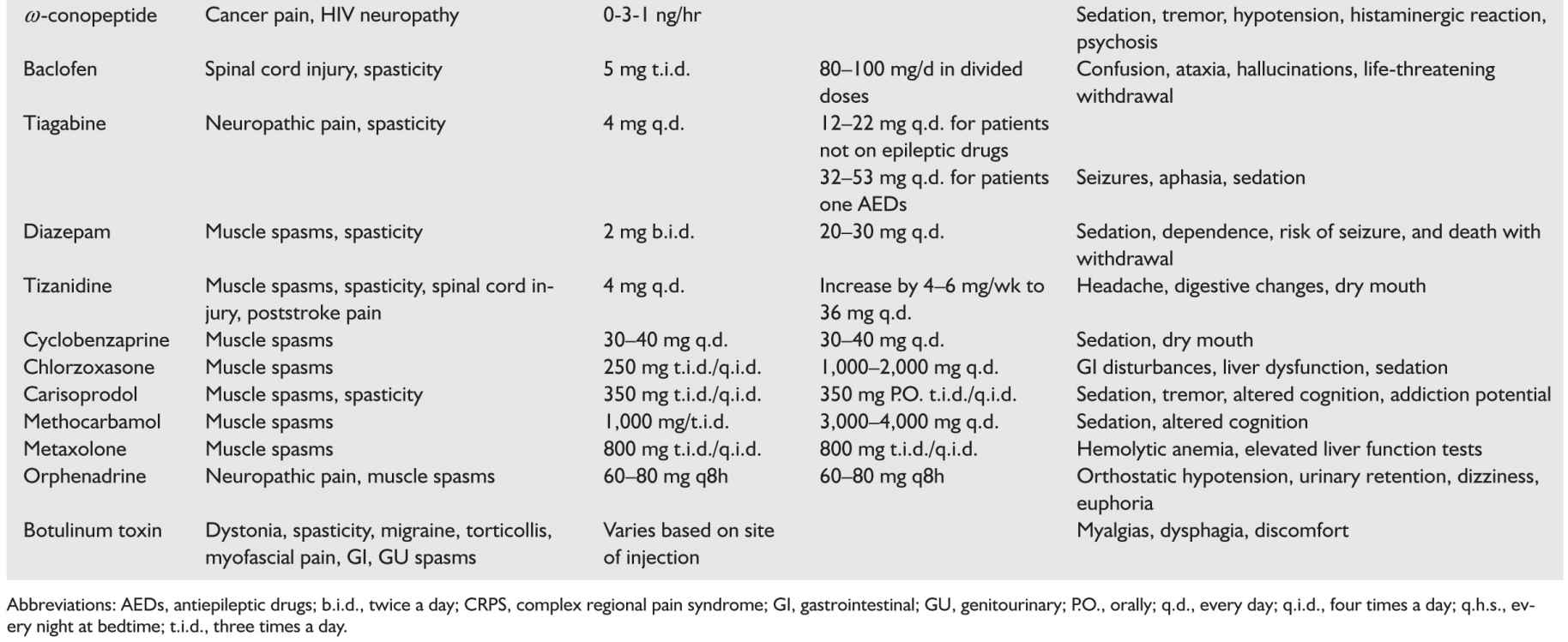
ω-Conopeptides (ziconotide) acts on N-type calcium channels. Used intrathecally at 0.3 to 1 ng/kg/hour, the drug has been studied in patients with cancer and HIV/AIDS. Side effects include sedation, tremor, hypotension, a histaminergic reaction, and psychosis. Nimodipine, diltiazem, verapamil, and nifedipine are calcium channel blockers that may have a role in association with other agents in pain management.
7. Gabaergic agents. Baclofen, a derivative of GABA with activity at GABA-b channels, has both spinal and supraspinal activity (Table 55.2). Baclofen is used for spinal cord injury and spasticity. Initial therapy begins at 5 mg three times a day with titration to a daily dose of 100 mg if necessary. Baclofen may also be given intrathecally. Common side effects include confusion, somnolence, ataxia, and hallucinations. Sudden cessation of therapy may lead to a potentially life-threatening withdrawal syndrome, particularly in the setting of chronic intrathecal administration.
Tiagabine, a GABA reuptake inhibitor, is started at 4 mg daily and increased by 4 to 8 mg/day to a final goal of 12 to 22 mg/day for patients not on AEDs, or 32 to 52 mg/day for patients on AEDs. Common side effects include risk of seizures in patients without history of seizure disorder, aphasia, and sedation.
Diazepam is a benzodiazepine and muscle relaxant that enhances the inhibitory action of GABA-A receptors especially in patients with spinal cord disease and muscle spasms. Diazepam is usually started at 2 mg twice daily to a total dose of 20 to 30 mg/day. Side effects include sedation and dependence; a withdrawal or sudden cessation may lead to seizures and death.
8. Muscle relaxants. Tizanidine is a centrally active alpha-2 adrenergic agonist used for spasticity, spinal cord injury, and poststroke pain (Table 55.2). Dosing starts at 4 mg/day and is increased by 4 to 6 mg/week to a total of 36 mg/day. Side effects include headache, indigestion, and dry mouth. Concomitant use of other alpha-2 agonists increases the risk of arterial hypotension. Cyclobenzaprine is a muscle relaxant that primarily acts at the brainstem, although it is not effective for centrally mediated spastic states. The dose is 30 to 40 mg/day and side effects include sedation and dry mouth. Liver function tests are recommended with long-term use.
Chlorzoxazone is centrally acting with a target dose of 1,000 to 2,000 mg/day. Side effects include GI disturbances, sedation, and liver dysfunction. Carisoprodol is a muscle relaxant that acts centrally at the reticular activating system and the spinal cord. The treatment goal is 350 mg three to four times a day. Side effects include sedation, tremor, altered cognition, and possible addiction potential. Methocarbamol is used at 3,000 to 4,000 mg/day. Metaxolone is dosed at 800 mg three to four times a day; this drug may lead to hemolytic anemia and abnormal liver function tests.
Orphenadrine, an NMDA receptor antagonist with anticholinergic effects, acts via central and peripheral mechanisms. This drug is used for neuropathic pain and muscle spasms. It potentiates the analgesic effects of opioids. Orphenadrine is started at 60 to 80 mg every 8 hours. Side effects include orthostatic hypotension, urinary retention, dizziness, and euphoria.
Botulinum toxin acts at the neuromuscular junction and inhibits the release of acetylcholine presynaptically. Its effect lasts for approximately 3 months. This agent is used for dystonias, spasticity, chronic migraine, hyperhidrosis, myofascial pain, and GI and genitourinary (GU) spasm. Botulinum toxin should not be used in patients with neuromuscular junction or motor neuron disorders such as myasthenia gravis (MG), Lambert–Eaton myasthenic syndrome (LEMS), and amyotrophic lateral sclerosis (ALS). Side effects include myalgia, dysphagia, and local discomfort. The amount injected is tailored to the site of injection, degree of spasm and musculature being injected.
9. Other agents. Clonidine, an alpha-2 agonist that potentiates the analgesic action of opioids, is useful for managing neuropathic pain. A transdermal patch is started at a dose of 0.1 mg/day and changed every 7 days. Side effects include sedation, dry mouth, and orthostatic hypotension. Capsaicin, an extract of chili pepper, is thought to cause analgesia by depletion of substance P. Recent approval by the US FDA for an 8% topical patch has shown some success in the treatment of postherpetic neuralgia. Its application may lead to discomfort and irritation before analgesia.
Chronic pain states often differentially engage different types of glutamate receptors as the source, time course, and quantities of released glutamate and co-transmitters are different. Glutamate is released from central terminals in the spinal cord upon noxious stimulation activating amino-3-hydroxy-5-methyl-4-isoxazole propionic acid (AMPA). Prolonged activation of nociceptors from tissue damage evokes continuous release of glutamate that, in combination with co-released neuropeptides like substance P, causes long-lasting membrane depolarization. Presynaptic GLuR’s localized on central terminals of primary afferents also play a role in segmental nociceptive transmission. These include kainate, NMDA, and some metabotropic GLuR. There are a great variety of GLuR’s that can be used as targets for pharmacological intervention for the treatment of pain. The NMDA receptor has been considered an important target for the treatment of chronic pain. However, the use of clinically available NMDA antagonists is limited because of unacceptable side effects like psychotomimesis, ataxia, and sedation. These side effects appear to be related to the mechanism of action as they are observed with both channel blockers and competitive agonists. With channel blockers, the neurotoxicity is lower with low-affinity compounds (memantidine) compared to high-affinity drugs (MK 801). One of the most promising strategies to dissociate the analgesia from side effects is the development of the subtype NMDA antagonists ifemprodil and eliprodil that are highly selective for the NMDA receptor containing NR2B sub units. The reason for the low side-effect profile of NR2B selective NMDA antagonists is unclear. However, they have a restricted CNS localization pattern and are present in structures specifically involved in nociceptive transmission implying a stronger inhibition of the transmission involving greater receptor activation for chronic pain versus normal physiologic transmission.
Ketamine has also been used to treat various chronic pain syndromes, especially those with a neuropathic component presumably by inhibition of NMDA receptors, though other mechanisms could be involved including enhancement of descending inhibition and anti-inflammatory effects at central sites. Short-term infusions of ketamine produces potent analgesia during administration only, while prolonged infusions (4 to 14 days) showed long-term effects for up to 3 months. Side effects of ketamine include psychedelic symptoms, memory deficits, nausea, vomiting, somnolence, and cardiovascular stimulation. The recreational use of ketamine is increasing and additional risks including bowel and bladder and renal complications. In clinical settings, ketamine is well tolerated, especially when used with benzodiazepines. Close monitoring of patients receiving ketamine is mandatory, particularly aimed at CNS, hemodynamics, renal, and hepatic symptoms. Further research is needed to determine if benefits outweigh risks and costs. Until definite proof is obtained, ketamine use should be restricted to patients with resistant severe neuropathic pain.
AMPA receptor antagonists are a new class of drugs seemingly devoid of the typical neurotoxic effects of NMDA antagonists. A recent finding notes that antinociceptive doses of gabapentin, known to be useful for the treatment of neuropathic pain, selectively inhibit AMPA responses of dorsal spinal horn neurons. The use of selective glutamate receptor antagonists and NMDA blockers has been demonstrated to have high analgesic efficacy. The major problem with previous generations of these drugs has been side effects. The new classes of glutamate modulation have positive side-effect profiles, and continued research into these therapies is promising.
B. Nonpharmacologic treatments.
Physical therapy and occupational therapy. The use of heat, ultrasound, electrical stimulation, and deep tissue massage may reduce the discomfort associated with chronic pain states. Goals of physical therapy include: decreasing pain, improving range of motion, improving strength, and improving functional status. Patients should focus therapy in the plane of comfort: meaning if it is painful to flex the lumbar spine then extension-based exercises should be utilized, and vice versa. Therapy should also improve the pain rather than worsen it.
1. Electrical stimulation involves the use of either transcutaneous electrical nerve stimulation (TENS), spinal cord (dorsal column), or thalamic stimulators. Patients with neuropathic pain states, muscular pain, central pain, and axial low back pain may benefit from these therapies. The classical “gate control” theory postulates that stimulation of large fiber (a beta) neurons closes the gate that has been opened or initiated by the smaller diameter nociceptors.
2. Spinal cord stimulation has become more refined and more widely used and is particularly useful in the setting of chronic neuropathic pain not responsive to more conservative options.
3. A trial initially performed before permanent implantation affords the patient the experience of the stimulation sensation and also to determine the level of pain relief before the more invasive permanent implantation. Most of the time, the trial is performed percutaneously during an outpatient procedure and left in for a period varying from 3 to 7 days.
4. Transmagnetic stimulation is a form of neuromodulation that potentiates or inhibits the transmission of nerve signals, but it is not the actual means of transmission itself. Transcranial magnetic stimulation is a noninvasive method enabling the stimulation of specific cortical areas by an electric current induced by a coil placed on the scalp. A rapidly varying electric current (1 ms) flows through a wiring system and creates an electromagnetic field that produces a current a few centimeters inside the brain parenchyma. This focused electrical current may depolarize neurons and creates evoked responses or changes neuronal plasticity. This repetitive transcranial magnetic stimulation (rTMS), especially when applied to the dorsolateral prefrontal cortex, has been found to be effective in treating major depression. Other studies have shown it to be effective in reducing pain especially if rTMS is performed at a high frequency of 10 Hz over the M1 and the current is delivered in a posterior/anterior direction. rTMS has been shown to be effective in reducing neuropathic pain in some individuals; the greatest effect has been noted with a figure 8 coil over the hand or face area of M1. There has also been a therapeutic effect of rTMS for fibromyalgia pain with treatment to the right M1 for 10 consecutive days, producing a long-term analgesic effect. rTMS has also been noted to be effective in reducing pain associated with CRPS. Patients are treated with pharmacologic and rehabilitation treatment as per standard practice with rTMS applied as additional treatment.
Mounting evidence suggests that stimulation of M1 and prefontal cortex activates distant brain areas. Electrophysiologic studies have shown that motor cortex stimulation has inhibitory effects on thalamic and spinal nociceptive neurons. M1 stimulation decreases the availability of opioid receptors in the periaqueductal gray area and the magnitude of pain reduction correlates with the availability of μ-opioid receptors. High frequency of >10 Hz stimulations seem to be the most effective in reducing pain. New rTMS paradigms have been proposed to increase the size and duration of long-term potentiation. One such treatment is theta-burst stimulation (TBS). This is based on studies suggesting that this pattern of stimulation is able to induce long-term synaptic changes. Various protocols of TBS are used consisting of a short burst of 3 TMS pulses with inner high frequency (50 Hz within the gamma range) that are delivered at 5 Hz (in the theta range). These different stimulation protocols have been noted to decrease overall pain at a higher percentage than standard rTMS techniques. The total number of pulses per session seems to be related to the net analgesic effects, but it is not clear whether a minimum number of pulses is required to obtain this effect and if there is a ceiling effect to this therapy. The major limitation of rTMS is the short duration of analgesia achieved with transcranial stimulation, and therefore the efficacy of this modality is not totally established in patients with chronic pain. Multiple sessions of rTMS and increased number of pulses per session have cumulative analgesic effects with long-lasting pain control in patients with chronic pain syndrome.
5. Interventional modalities can occasionally be useful in the setting of chronic pain. The modalities chosen need to be tailored to the individual patient and treatment goals. Constant evaluation/monitoring of the responses to these interventional modalities also needs to be undertaken, as repeated procedures can expose the patient to increasing risk.
6. Several types of steroid injections can be performed, depending on the suspected diagnosis. In general, whatever the injection being considered, it should be clear to patients that steroid injections do not cure the underlying pathology. Reasons to consider injections is to potentially help ameliorate the pain and to get the patient back to a reasonable level of function.![]()
While there is conflicting evidence on the efficacy of epidural injections in treating low back pain alone, the evidence does point to short-term benefits for relief in the setting of radicular leg pain associated with lumbar disc herniations. Advanced imaging is recommended before epidural injections, MRI being the preferred modality, though computed tomography (CT) scans of the lumbar spine can also be appropriate.![]()
Several other types of injections can be performed, depending on the diagnosis and the underlying pathology (see Video 55.1). Injections for low back pain include epidural injections (see Video 55.2), sacroiliac joint injections, lumbar facet injections, piriformis injections, quadratos lumborum injections, trigger point injections, and intaarticular hip joint injections (see Video 55.3). Again, similar to epidural injections, these injections do not cure or heal the underlying pathology. ![]()
Sympathetic blocks have the potential to offer pain relief in specific subsets of patients. They also have the added benefit of being a diagnostic procedure in that they help differentiate sympathetically maintained pain from sympathetically independent pain (see Video 55.4). In general, sympathetic blocks are more effective in the long run if they are combined with a course of physical therapy or a home exercise program. ![]()
7. Peripheral nerve blocks can have some benefit in certain patients, particularly when combined with physical therapy or a home exercise regimen.
8. It should be noted that while most of the above-mentioned procedures are performed with the use in fluoroscopy as the imaging modality of choice, there is a gradual movement toward ultrasound as a modality used in conjunction with or independently from fluoroscopy. Ultrasound technology does show promise in the arena of interventional pain as it has the ability to show superior soft tissue structures including blood vessels, nerves and nerve roots. It also has the added advantage of not exposing the patient and practitioner to radiation during the procedure.
9. Psychological treatment is also an important component of the patient’s overall pain treatment plan. A support network for patients, involving family and friends, may be conducive to the healing process. Additionally, the use of biofeedback along with adjunctive physical therapy may help a patient with their discomfort.
Finally, the ultimate goal in the management of patients with chronic pain is the precise diagnosis with appropriate treatment for the painful condition. Referral to an interventional pain physician may benefit patients after conservative therapy has failed. Disease processes difficult to treat, such as CRPS, diabetic neuropathy, and peripheral neuropathies, may be addressed with a specialist versed in providing interventional modalities along with pharmacotherapy. Physical therapy is also vital to the patient’s treatment in decreasing discomfort. It is crucial to have honest communication with the patient about treatment goals, expectations, and the possibility of achieving those expectations.
10. Hypnosis. Training patients in self-hypnosis is an attractive component in pain therapy. Hypnosis incorporates relaxation, focused attention, imaging, interpersonal processing, and suggestion. Response to hypnosis treatment is highly variable. Recent studies use a 30% reduction in average daily pain intensity to represent clinically meaningful improvement in chronic pain states. Certain pain types respond to hypnosis better than others. In most instances, neuropathic pain seems to respond to hypnosis better than non-neuropathic pain. There is also a hypnotizability factor, which reflects a person’s tendency or trait to respond positively to a variety of different suggestions following a hypnotic induction. One of the most important findings from recent neurophysiologic studies of pain is that there is no single “pain center” in the brain responsible for processing pain. Cortical areas most often activated during pain are the thalamus, ACC, insular cortex (IC), primary and secondary sensory cortices and prefrontal cortex. Hypnotic suggestion to reduce pain and unpleasantness influences activity in the ACC but not in other brain areas including the sensory cortex. It is also noted that cortical neurons fire at different frequencies and the speed at which they fire is associated with different brain states. Pain is associated with more neuron firing at relatively fast beta (13 to 30 Hz) frequencies and fewer neurons firing at slow alpha (8 to 13 Hz) frequencies. Hypnotic suggestion result in changes in brain activity consistent with those observed in individuals who experience pain relief (decrease in beta activity and an increase in alpha activity). Therefore, hypnotic analgesia may influence pain both by altering activity in specific areas and by facilitating shifts in general brain states. There are two current theories as to how hypnosis affects chronic pain. One is neodissociation where a state of effortlessness is thought to be associated with a shift in the control of responses from higher executive function to cognitive subsystems that have a direct influence on the behavioral responses with the usual layer of judgment or critical screening. Dissociation theories hypothesize that hypnosis involve a qualitative shift in the nature of cognitive processes. Hypnosis creates a shift from an active to a passive form of attention. These attentional shifts are associated with a reduction in the monitoring of control and the censoring experience. The sociocognitive model of hypnosis argues that the concept of an altered state is not required to understand hypnosis but that it is best explained by the same sociopsychological factors that explain all behaviors. Several mechanisms have been offered as important elements of hypnosis. These include relaxation, use of distracting imagery, focused attention, and expectancy. Patients who manage their chronic pain through mindfulness meditation training and therapies incorporating mindfulness are becoming very popular. In these approaches, efforts to directly resist or reduce chronic pain are thought to contribute to suffering, and having a direct goal of a reduction in chronic pain might decrease the quality of life in some patients. Hypnosis can be used to reduce pain; however, in many instances it serves as a tool to help patients better cope and accept their pain rather than seek to change their experience.
Hypnosis for chronic pain states has few negative side effects. In fact, with hypnosis most patients report positive side effects such as improved sense of well-being, greater sense of control, improved sleep, and increased satisfaction with life, all independent of whether they report reductions in pain. Hypnotic suggestion can target specific pain domains and outcomes as well as activities in specific brain regions.
• Chronic pain is any pain lasting 12 weeks or more which may be secondary to traumatic injury, but in many instances there may be no clear cause.
• Pain may be somatic, visceral, and/or neuropathic. Somatic pain is typical pain that occurs after an injury. It is sharp and well localized. Visceral pain is often associated with organ pain. It is poorly localized, dull, and achy. Neuropathic pain is the most difficult to treat, could be sharp, stabbing but also described as achy, throbbing, squeezing, itching, or crawling.
• Chronic pain can be managed by medications. The most common of which are opioids which have side effects with possible tolerance, addiction, or withdrawal with discontinuation.
• Other medications such as NSAIDs, AEDs, SNRIs, GABAergic agents, and muscle relaxants can be used in combination with opioids or other medications to produce analgesia.
• Nonpharmacologic therapies to treat chronic pain include electric stimulation either by transcutaneous methods (TENS), spinal cord (dorsal column stimulator), or thalamic stimulation. TMS of cortical areas can also help control pain. All of these effect pain by neuromodulation.
• Hypnosis may also benefit patients with chronic pain by incorporating a number of components including relaxation techniques, focused attention, imaging, and suggestions to help modulate pain.
• Interventional modalities can be useful in the setting of chronic pain. The use of epidural steroid injection to treat chronic back pain can be coupled with local anesthetic injections of sacroiliac joints, lumbar facet, and trigger points to reduce pain.
• Sympathetic blocks have the ability to reduce certain types of pain, especially neuropathic. These blocks can be used for diagnosis and treatment of both upper and lower extremity pain.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree