Cranial Nerve V (The Trigeminal Nerve)
Anatomy of Cranial Nerve V (Trigeminal Nerve)
The trigeminal nerve is a mixed nerve that provides sensory innervation to the face and mucous membranes of the oral and nasal cavities and motor innervation to the muscles of mastication [9].
Motor Portion
The motor nucleus of the trigeminal nerve is situated at a midpontine level (Fig. 9.1), medial to the main sensory nucleus of the trigeminal nerve, near the floor of the fourth ventricle. It receives its supranuclear control through corticobulbar fibers originating in the lower third of the precentral gyrus. These bilateral connections travel through the corona radiata, internal capsule, and cerebral peduncle and decussate in the pons before supplying the motor nuclei.
The motor root, or portio minor, exits from the motor nucleus, passes forward in the substance of the pons, and emerges from the anterolateral aspect of the pons anterior and medial to the larger sensory root (the portio major). The motor root then passes through the posterior fossa and pierces the dura mater beneath the attachment of the tentorium to the tip of the petrous portion of the temporal bone. It then enters a cavity in the dura mater overlying the apex of the petrous bone (Meckel’s cave), travels beneath the trigeminal (gasserian) ganglia, and leaves the skull through the foramen ovale. After leaving the skull, the motor root joins the mandibular (third) division of the trigeminal nerve to form the mandibular nerve, which supplies the masticatory muscles: the masseter, temporalis, and medial and lateral pterygoid muscles. In addition, motor fibers are given off to the tensor tympani, tensor veli palatini, and mylohyoid muscles, and to the anterior belly of the digastric muscle.
Sensory Portion
The pseudounipolar perikarya of the sensory portions of the trigeminal nerve are in the semilunar or gasserian ganglion, which is situated near the apex of the petrous bone in the middle cranial fossa. From this ganglion, the fibers of the sensory root (portio major) enter the substance of the pons, course dorsomedially, and terminate in three major nuclear complexes (Fig. 9.1) within the brainstem: the nucleus of the spinal tract of the trigeminal nerve, the main (or principal) sensory nucleus, and the mesencephalic nucleus.
On entering the pons, many of the sensory fibers descend as a bundle, the spinal tract of the trigeminal nucleus, to the caudal end of the medulla and into the spinal cord (as far as the third or fourth cervical level), where it becomes continuous with Lissauer’s tract. As the spinal tract descends, it gives off fibers to the medially located nucleus of the spinal tract of the trigeminal nerve, which also descends into the upper cervical cord. This nucleus is divided into a pars oralis (which extends from the midpons to the inferior olive), a pars interpolaris (which extends from the rostral third of the inferior olive to the obex of the fourth ventricle), and a pars caudalis (which extends to and is continuous with the dorsal horn gray matter of the cervical spinal cord). The fibers of the ophthalmic division of the trigeminal nerve travel in the most ventral part of the spinal tract and extend most caudally (i.e., terminate in the trigeminal nucleus in series with the second cervical sensory level). The fibers of the mandibular division of the trigeminal nerve travel in the most dorsal part of the spinal tract and terminate in the most rostral level of the spinal nucleus of the trigeminal nerve. The rostral trigeminal nuclei are important in intraoral and dental sensation [39]. In another possible sensory somatotopic spinal nucleus representation, the midline facial areas (nose and mouth) are represented rostrally in the spinal nucleus, whereas the more lateral facial sensation fibers terminate in more caudal spinal nucleus regions. This pattern of termination may account for the onionskin pattern of facial sensory loss with intramedullary lesions and the perioral numbness that occurs with more rostral spinal nucleus and tract lesions.
The spinal nucleus of the trigeminal nerve receives fibers that convey the sensations of pain, temperature, and soft touch from the face and mucous membranes. From the spinal nucleus, ascending fibers travel mainly ipsilaterally in the trigeminothalamic tract to terminate in the ventral posteromedial (VPM) and intralaminar nuclei of the thalamus.
Other fibers from the portio major enter the pons and ascend and enter the main sensory nucleus of the trigeminal nerve. This nucleus is located in the lateral pons, posterolateral to the motor nucleus of the trigeminal nerve. Fibers entering this nucleus are concerned with tactile and proprioceptive sensation. The main sensory nucleus gives off ascending fibers that terminate in the thalamus. These fibers travel in the ventral crossed trigeminothalamic (quintothalamic) tract or trigeminal lemniscus, which ascends with the medial lemniscus, and in the uncrossed dorsal trigeminothalamic tract. Both these fiber tracts terminate predominantly in the ventral posterior medial (VPM) nucleus of the thalamus.
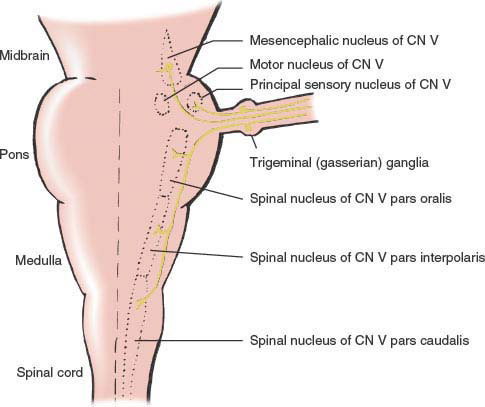
FIG. 9.1. Schematic diagram of the trigeminal system. CN = cranial nerve.
The third sensory trigeminal nucleus, the mesencephalic nucleus, extends cephalad from the main sensory nucleus to the superior colliculus of the mesencephalon. This nucleus receives proprioceptive impulses from the masticatory muscles and from muscles supplied by other motor cranial nerves.
The sensory root (portio major) of the trigeminal nerve leaves the pons along with the motor root (portio minor) and expands in the Meckel’s cave to form the trigeminal (gasserian) ganglion. This ganglion lies near the cavernous sinus and internal carotid artery and gives rise to three nerve trunks: the ophthalmic, maxillary, and mandibular divisions of the trigeminal nerve.
This division (V1) (Fig. 9.2) lies in the lateral wall of the cavernous sinus in close association with the third, fourth, and sixth cranial nerves. Along with these three nerves, the ophthalmic division enters the orbit through the superior orbital fissure. Before leaving the cavernous sinus, this division divides into tentorial, lacrimal, frontal, and nasociliary branches. The tentorial branch supplies the dura of the cavernous sinus, sphenoid wing, anterior fossa, petrous ridge, Meckel’s cave, tentorium cerebelli, posterior falx cerebri, and dural venous sinuses. The frontal branch divides into the supraorbital nerve—supplying the medial upper lid and conjunctiva, the frontal sinuses, the forehead, and the scalp—and the supratrochlear nerve, which supplies the conjunctiva, medial upper lid, forehead, and side of nose (Fig. 9.3). The lacrimal nerve, through its lateral palpebral branch, innervates the conjunctiva and skin in the area of the lacrimal gland. The lacrimal nerve also carries postganglionic parasympathetic fibers for reflex lacrimation. The nasociliary nerve divides into nasal nerves, which innervate the mucosa of the nasal septum, the lateral nasal wall, and the inferior and middle turbinates, and an external nasal branch, which innervates the skin of the top of the nose. The infratrochlear branch of the nasociliary nerve supplies the lacrimal sac, the caruncle, and the conjunctiva and skin of the medial canthus. Two long ciliary nerves carry sensation from the ciliary body, the iris, and the cornea and also carry sympathetic innervation to the dilator of the pupil. Multiple short ciliary nerves transmit sensory fibers from the globe, which pass through the ciliary ganglion to join the nasociliary nerve; these short ciliary nerves also carry postganglionic parasympathetic fibers from the ciliary ganglion to the constrictor of the pupil and the ciliary muscle. The parasympathetic fibers reach the ciliary ganglion through the inferior division of the oculomotor nerve destined to innervate the inferior oblique muscle.
The ophthalmic division therefore supplies the skin of the nose, the upper eyelid, the forehead, and the scalp (as far back as the lambdoidal suture in the midline and for 8 cm lateral to the midline) (Fig. 9.3); the upper half of the cornea, conjunctiva, and iris; the mucous membranes of the frontal, sphenoidal, and ethmoidal sinuses and the upper nasal cavity and septum; the lacrimal canals; and the dura mater of the anterior cranial fossa, falx cerebri, and tentorium cerebelli.
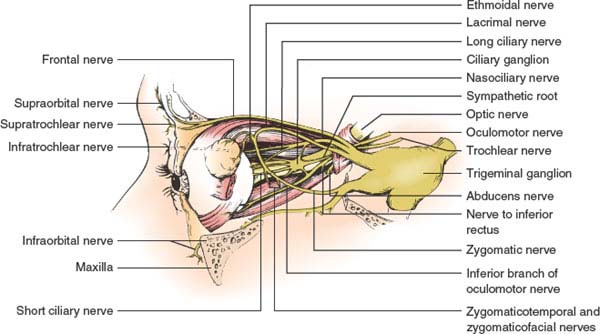
FIG. 9.2. The branches of the ophthalmic and maxillary divisions of the trigeminal nerve.
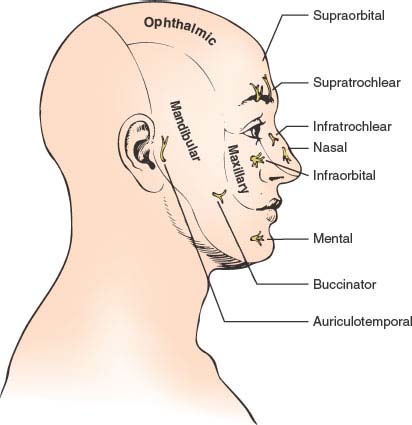
FIG. 9.3. Areas of skin supplied by the three major trigeminal nerve divisions.
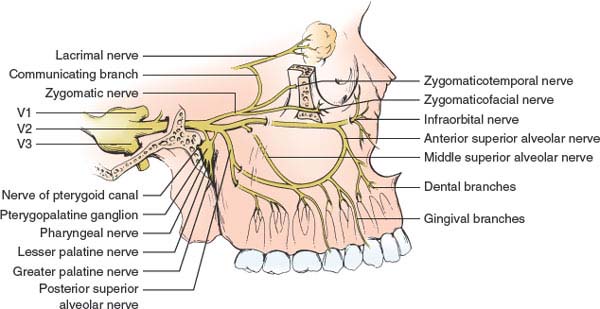
FIG. 9.4. The branches of the maxillary division of the trigeminal nerve.
MAXILLARY DIVISION
This division (V2) passes (Figs. 9.2 and 9.4) through the inferolateral portion of the cavernous sinus and then leaves the skull through the foramen rotundum to enter the sphenopalatine fossa. Next, it enters the orbit through the inferior orbital fissure (as the infraorbital nerve) and, after traveling through the infraorbital canal, reaches the face by way of the infraorbital foramen. Within the sphenopalatine fossa and infraorbital canal, palatine nerves and middle, posterior, and anterior superior alveolar nerves arise, which supply the upper teeth, maxillary sinus, nasopharynx, soft palate, roof of the mouth, and tonsils. After exiting from the infraorbital foramen, the nerve divides into an inferior palpebral branch to the lower lid, a nasal branch to the side of the nose, and a superior labial branch to the upper lip. A zygomaticofacial branch innervates the cheek.
The maxillary division therefore supplies the skin of the lower eyelid, the lateral nose, upper lip, and cheek (Fig. 9.3); the lower half of the cornea, conjunctiva, and iris; the mucous membranes of the maxillary sinus, lower nasal cavity, hard and soft palates, and upper gum; the teeth of the upper jaw; and the dura mater of the middle cranial fossa (through the middle or recurrent meningeal nerve).
MANDIBULAR DIVISION
The mandibular division (V3) (Fig. 9.5) joins the motor root of the trigeminal nerve to form the mandibular nerve. This nerve leaves the skull through the foramen ovale and travels in the infratemporal fossa, dividing finally into several terminal branches. The motor branches supply the eight muscles noted in the preceding text, whereas the lingual nerve conveys sensation from the lower gums and the papillae and mucous membrane of the anterior two-thirds of the tongue. Inferior dental branches supply the lower gums and teeth of the mandible; mental branches, after emerging from the bone at the mental foramen, supply the skin of the chin and the skin and mucous membrane of the lower lip.
In addition to the muscles listed previously (see Motor Portion), the mandibular nerve supplies the skin of the lower lip, lower jaw, chin, tympanic membrane, auditory meatus, and upper ear (Fig. 9.3); the mucous membranes of the floor of the mouth, the lower gums, and the anterior two-thirds of the tongue (not taste sensation, which is carried by the facial nerve); the teeth of the lower jaw; and the dura mater of the posterior cranial fossa.
Clinical Evaluation of Cranial Nerve V Function
Sensory Evaluation
Exteroceptive sensation (pain, light, touch, heat, and cold) is tested on the face and mucous membranes. Each of the three trigeminal divisions is tested individually and compared with the opposite side. Lesions of individual divisions (distal to the gasserian ganglion) result in sensory loss confined to the cutaneous supply of that division (Fig. 9.3) with relatively little overlap into the cutaneous area of another division. Lesions at or proximal to the gasserian ganglion result in sensory loss that affects the whole ipsilateral face. Lesions within the brainstem or upper cervical cord may result in an onionskin distribution of sensory loss, whereas dissociation of sensation on the face (pain and temperature vs. touch sensation) differentiates lesions affecting the spinal tract and nucleus of the trigeminal nerve from lesions affecting the main sensory nucleus.
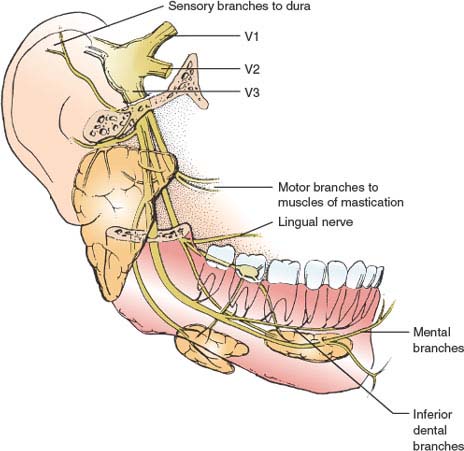
FIG. 9.5. The branches of the mandibular division of the trigeminal nerve.
The cutaneous area over the angle of the mandible is supplied by the second and third cervical roots (by way of the great auricular nerve) and not by the trigeminal nerve. Therefore, a hemifacial sensory loss that spares the angle of the jaw is probably organic, whereas one that includes this area may be of functional origin or related to an intramedullary lesion.
Motor Evaluation
The trigeminal nerve supplies the muscles of mastication. These are tested by having the patient clench the jaw (masseters and temporalis), move the jaw from side to side against resistance (lateral pterygoids), and protrude the jaw. With nuclear or infranuclear lesions of the motor division of the trigeminal nerve, the temporalis and masseter muscles on the side of the lesion do not contract when the jaw is clenched, the jaw deviates to the paralyzed side when the mouth is opened (due to contraction of the contralateral intact lateral pterygoid muscle), and the jaw cannot be deviated toward the nonparalyzed side (due to ipsilateral lateral pterygoid paresis). Atrophy and fasciculation of the masticatory muscles may also be evident. Trismus (inability to open the jaw) may be seen with acute dystonic reactions (e.g., due to neuroleptic medications), polymyositis, tetanus, trauma to the muscles of mastication, infection of the pterygomandibular space, neoplasm, radiation therapy, tryptophan-associated eosinophilic connective tissue disease, nemaline myopathy, and psychogenic factors [14,77,92]. Acute trismus has also been described due to Foix–Marie–Chavany syndrome, a clinical disorder characterized by facio-pharyngo-glosso-masticatory weakness and loss of voluntary control of facial movements with preserved automatic and emotional motility classically associated with bilateral anterior opercular lesions [32].
Other muscles supplied by the trigeminal nerve (mylohyoid, anterior belly of the digastric, tensor, tympani, tensor veli palatini) are difficult to evaluate clinically. However, flaccidity of the floor of the mouth due to mylohyoid and digastric paralysis may be evident on palpation, and paralysis of the tensor tympani may result in difficulty in hearing high notes.
Reflex Evaluation
The important reflexes conveyed by the trigeminal nerve include the corneal reflex and the jaw jerk (masseter reflex). The afferent arc of the corneal reflex travels through the ophthalmic (upper cornea) and maxillary (lower cornea) divisions of the trigeminal nerve. The efferent arc moves through the ipsilateral (direct reflex) and contralateral (consensual reflex) facial nerve to the orbicularis oculi muscles. Lesions of the trigeminal nerve result in loss of the ipsilateral and contralateral responses. These lesions may involve the peripheral or pontomedullary trigeminal pathways; however, a suprasegmental modulation of this reflex also exists, because a parietal lobe lesion (involving the perisylvian portion of the postcentral gyrus) may result in a contralateral loss of the corneal reflex. The jaw jerk or masseter reflex involves contraction of the masseter and temporalis muscles when the patient’s lower jaw is tapped. The afferent arc is through the 1a motor fibers in the mandibular division of the trigeminal nerve that run to the mesencephalic nucleus of the trigeminal nerve. The efferent arc also travels through mandibular fibers that originate in the motor nucleus of the trigeminal nerve. Lesions anywhere along this reflex arc result in depression of the ipsilateral jaw reflex, whereas bilateral supranuclear lesions result in an accentuated response.
Trigeminal sensory neuropathy may result in significant disability due to impaired intraoral sensation associated with a disturbance of mastication and swallowing [3]. Disturbed intraoral sensation, combined with impaired trigeminal reflexes (especially the masseter inhibitory reflex), interferes with the neural mechanisms that regulate chewing.
Another reflex mediated partly by trigeminal pathways is the blink reflex (glabellar reflex, orbicularis oculi reflex). Percussion over the supraorbital ridge results in bilateral contraction of the orbicularis oculi muscles. The afferent arc of this response is likely mediated by the ipsilateral main sensory nucleus of the trigeminal nerve and the ipsilateral and contralateral spinal nuclei of the trigeminal nerve. The spinal nuclei of the trigeminal nerve (bilateral) make motor connections through the corresponding facial nuclei, which innervate the orbicularis oculi muscles. By studying the blink reflex electrically, subtle peripheral and central lesions of the trigeminal and facial nerves may be uncovered.
The corneomandibular reflex consists of bilateral eye blink and a brisk anterolateral jaw movement induced by corneal stimulation [78]. A spontaneous palpebromandibular (eyelid jaw) synkinesia (SPMS), in which jaw movements similar to those in the corneomandibular reflex regularly accompany spontaneous eye blinks without an external corneal stimulation, has been described in patients with upper brainstem (bilateral lesions above the midpons) or bilateral cerebral lesions [78]. SPMS is pathophysiologically the same as the eyelid jaw synkinesia of the corneomandibular reflex, and both synkinesias originate centrally, probably in the pons. In the corneomandibular reflex, the jaw movement is primarily related to the blink rather than the corneal stimulus, but corneal stimulation may be necessary to overcome a higher threshold for expression of the synkinesia in patients with SPMS [78].
Localization of Lesions Affecting Cranial Nerve V
Supranuclear Lesions
Supranuclear control of trigeminal motor function is bilateral; however, the contralateral hemisphere exerts predominant control on the voluntary activity of the masseter [17]. Corticobulbar fibers originate in the lower frontal motor cortex, descend through the corona radiata, internal capsule, and cerebral peduncle, and then decussate in the pons to supply the motor nucleus of the trigeminal nerve. Lesions interrupting this pathway may result in contralateral trigeminal motor paresis (e.g., deviation of the jaw “away from” the lesion), but because of the bilateral innervation, paresis may be mild. Bilateral upper motor neuron lesions (pseudobulbar palsy) result in profound trigeminal motor paresis, often with an exaggerated jaw reflex. Mastication is then markedly impaired. Thalamic lesions may result in anesthesia of the contralateral face. Parietal lesions may be associated with depression of the contralateral corneal reflex, even when facial sensation is otherwise intact.
Nuclear Lesions
The motor and sensory nuclei of the trigeminal nerve may be involved by lesions (e.g., primary or metastatic tumors, arteriovenous malformation, demyelinating disease, infarction, hemorrhage, syringobulbia) that affect the pons, medulla, or upper cervical cord. These nuclear lesions involve other brainstem structures, and therefore brainstem lesions of the trigeminal nuclei are diagnosed by “the company they keep” (e.g., long tract signs, and other cranial nerve involvement).
Lesions affecting the dorsal midpons may affect the motor nucleus of the trigeminal nerve. Congenital anomalies of the motor component of the trigeminal nerve are rare, and may be associated with the involvement of cranial nerve VII or XII. Ipsilateral paresis, atrophy, and fasciculations of the muscles of mastication therefore occur. A pontine localization of this masticatory paresis is suggested by associated findings that may include contralateral hemiplegia (due to affection of the basis pontis), ipsilateral hemianesthesia of the face (due to affection of the main sensory nucleus of the trigeminal nerve), contralateral hemisensory loss of the limbs and trunk (due to spinothalamic tract affection), and ipsilateral tremor (due to affection of the brachium conjunctivum). Internuclear ophthalmoplegia (secondary to medial longitudinal fasciculus damage) and an ipsilateral Horner syndrome (due to involvement of descending sympathetic fibers) may also occur. Pontine syndromes are more thoroughly discussed in Chapter 15. Lateral pontine tegmental hemorrhage may rarely present as an isolated trigeminal sensory neuropathy, with numbness and paresthesias of half of the face, scalp, ear, and tongue [45], perhaps due to the involvement of the main sensory nucleus of the trigeminal nerve. A patient with small left dorsolateral pontine infarct presented with isolated orofacial sensory deficits (left upper face, tongue, and buccal mucosa numbness and paresthesias) without trunk or limb sensory findings indicating exclusive involvement of the pontine trigeminal sensory complex, including the principal sensory nucleus and the pars oralis of the spinal trigeminal nucleus and tract [48]. Unilateral trigeminal pain and numbness in a V1 through V3 distribution with decreased corneal reflex has also been described with a pontine abscess [5].
Patients with dorsal pontine lesions (usually tumors) may develop unilateral spasm and contracture of the masseter muscle, impairing the ability to open the jaw and forcing the patient to “speak through the teeth” [94]. Hemimasticatory spasm is a rare disorder of the trigeminal nerve that involves one or more of the jaw-closing muscles (masseter, temporalis, and medial pterygoid) on one side of the face and produces involuntary jaw closure due to paroxysmal unilateral muscle contraction [2,18,28,51,75,97]. The spasms may be sudden and brief or may last several minutes and cause intense pain. They are often triggered by voluntary jaw closure or other movements of the jaw and are sometimes relieved by voluntary jaw opening. Trigeminal function is otherwise normal. Electrophysiologic findings in hemimasticatory spasm suggest ectopic excitation of the trigeminal motor root or its nucleus, an abnormality that is analogous to ectopic excitation of the facial nerve in hemifacial spasm [2,18]. Some authors suggest that the nerve to the masseter and temporalis muscles may be entrapped at a point in its course between the lateral pterygoid muscle and the skull, causing focal demyelination and spontaneous discharges [18]. Hemimasticatory spasm may be associated with localized scleroderma [53] or may be seen with facial hemiatrophy [18,28,51,53,75,97].
The nucleus of the spinal tract of the trigeminal nerve extends from the caudal end of the pons to the third or fourth cervical spinal cord level. Therefore, lesions affecting the caudal pons, lateral medulla, or upper cervical cord result in ipsilateral facial analgesia, hypesthesia, and thermoanesthesia. Because the lateral spinothalamic tract lies in close proximity to the trigeminal spinal nucleus, the hemifacial sensory disturbance is often associated with contralateral trunk and extremity hypalgesia and thermoanesthesia. However, isolated orofacial pain and sensory deficit over the ipsilateral face, neck, tongue, and oral cavity may occur with small pontine or medullary lesions [45,72]. Caudal pontine lesions may damage the rostral spinal trigeminal nuclei and result in diminished intraoral sensation for all modalities but spared facial sensation [15,39]. Patients with isolated ventral pontine infarction may present with prominent ipsilateral midfacial sensory signs (hypesthesia and numbness of the midline facial areas) associated with dysarthria and contralateral hemiparesis [65]. The clinicoanatomic basis of the ipsilateral midfacial sensory loss is unknown but may be through the involvement of the dorsal trigeminothalamic tract or fibers related to the central regions of the face located medially. With upper (rostral) medullary spinal nuclear lesions, the entire trigeminal cutaneous distribution is affected. Lower medullary or upper cervical spinal nuclear lesions result in a sensory disturbance that affects the peripheral (lateral) forehead, cheek, and jaw (onionskin pattern of sensory loss). This onionskin segmental distribution reflects the rostral–caudal somatotopic arrangement of the cutaneous distribution of the spinal nucleus (e.g., perioral area—rostral; lateral face—caudal).
The spinal nucleus of the trigeminal nerve is characteristically affected in the lateral medullary (Wallenberg) syndrome, which is most often secondary to brainstem infarction due to intracranial vertebral artery occlusion [55,56]. This syndrome is described in Chapter 15. Currier et al. divided the trigeminal sensory loss in patients with Wallenberg syndrome into four clinical groups [20]:
1. In the typical syndrome, pain and temperature sensation are lost over the entire side of the face.
2. In the ventral syndrome, the first and second divisions of the trigeminal area are involved. This distribution follows damage to the ventral aspect of the descending tract and nucleus of V, where the ophthalmic and maxillary fibers travel.
3. In the dorsolateral syndrome, only the second and third divisions of the descending tract are affected.
4. In the superficial syndrome, all portions of the ipsilateral face are involved initially, but symptoms are mild and improve rapidly.
In a study of 50 patients with sensory dysfunction from lateral medullary infarction by Kim et al. [56], the findings were as follows:
1. Thirteen patients (26%) had a “classic” ipsilateral trigeminal–contralateral body and limb pattern of sensory loss with lesions affecting the most posterolateral part of the caudal-middle medulla.
2. Twelve patients had a bilateral trigeminal pattern associated with large ventrally extending lesions usually at the middle-rostral medulla.
3. Nine patients had a contralateral trigeminal pattern with lesions sparing the most posterolateral area of the medulla.
4. Ten patients had isolated body and limb sensory involvement.
5. Four patients had isolated trigeminal involvement.
6. Two patients had no sensory signs.
In these patients, trigeminal sensation was usually inhomogeneously involved among the three trigeminal divisions and was more often of an onionskin pattern than a divisional pattern. Therefore, in this study, the so-called classic dissociated sensory pattern of lateral medullary infarction was actually uncommon, whereas sensory patterns previously thought of as atypical were relatively frequent [56]. Patients with contralateral face-arm-trunk-leg sensory loss with lateral medullary infarction often have retro-olivary lesions in the ventrolateral tegmentum with preservation of the lateral medulla [99]. The mediolateral lesion in these patients likely involves the crossed lateral spinothalamic tract and the ventral trigeminothalamic tract, corresponding to the contralateral arm, face, and leg sensory loss. The ventrolateral extension of the infarct damages the far lateral part of the spinothalamic tract, corresponding to sensory loss in the contralateral lower trunk and leg [99]. Patients with lateral medullary infarction may develop diminished facial pain and temperature sensation, sparing intraoral structures (because the rostral spinal trigeminal nuclei in the caudal half of the pons conveying intraoral sensation is spared) [39].
The sensory sequelae in patients with lateral medullary infarcts include facial numbness, burning, or coldness; these sensory symptoms are often of delayed onset (up to 6 months after the infarct) [52]. Also, rare patients with Wallenberg syndrome may develop neurotrophic ulcerations in the territory of the trigeminal nerve [27,44]. Such ulcerations have also been described following alcohol injection of the gasserian ganglion, postencephalitic parkinsonism, syringobulbia, and trigeminal rhizotomy [27,44].
Lesions affecting the mesencephalic nucleus of the trigeminal nerve cause no apparent neurologic signs and symptoms, except perhaps depression of the ipsilateral jaw jerk (masseter reflex).
Lesions Affecting the Preganglionic Trigeminal Nerve Roots
In its cisternal course, the preganglionic trigeminal nerve root may be damaged by tumor (meningioma, schwannoma, metastasis, nasopharyngeal carcinoma), infection (granulomatous, infectious, or carcinomatous meningitis), trauma, or aneurysm. Preganglionic trigeminal nerve involvement is suggested by the involvement of the neighboring cranial nerves (especially cranial nerves VI, VII, and VIII). Trigeminal damage is manifested by ipsilateral facial pain, paresthesias, numbness, and sensory loss. The corneal reflex is depressed and a trigeminal motor paresis may occur. An idiopathic, isolated, self-limited trigeminal sensory neuropathy with transient abnormalities on magnetic resonance imaging (MRI) has been described [82]. Some patients with “idiopathic” trigeminal neuralgia have enhancement of the cisternal segment of the trigeminal nerve on MRI studies [90]. This enhancement usually resolves if the pain resolves.
The trigeminal roots may be involved by extension of pathologic processes (usually acoustic neuroma or meningioma) located in the cerebellopontine angle. Ipsilateral facial pain, paresthesias, sensory loss, masticatory paresis, and a depressed corneal reflex are then associated with ipsilateral tinnitus, deafness, and vertigo (due to involvement of cranial nerve VIII). Facial nerve paralysis, ipsilateral ataxia, and nystagmus (due to involvement of the cerebellar peduncles and cerebellum), ipsilateral lateral rectus paralysis (due to abducens nerve involvement), and, rarely, affection of cranial nerves IX through XII may also occur.
Trigeminal neuralgia (tic douloureux, Fothergill’s disease) refers to a distinctive syndrome of sudden, excruciating, lancinating, paroxysmal, and usually unilateral pains in the distribution of one or more of the divisions (often the maxillary or mandibular) of the trigeminal nerve [35]. This syndrome is more common with advancing age, affects women more often than men, and affects the right side more than the left. It is exceedingly rare for a patient to have bilateral trigeminal neuralgia during the same period of time, except in cases of multiple sclerosis. Typically, the paroxysms of pain are brief, usually lasting less than a minute. In severe cases, the pain may recur several times a day. The attacks are most frequent during the day, but they may awaken the patient at night. The painful paroxysms are often triggered by non-nociceptive facial stimulation and are often associated with facial contortions. Pain arising from the maxillary division (V2) is often referred to the upper lip, nose, and cheek. Pain originating in the mandibular division (V3) is often referred to the lower lip. Tic pain confined to the ophthalmic division (V1) is distinctly uncommon. Although often called idiopathic, this painful facial syndrome may be seen with pathology affecting the brainstem, preganglionic root, gasserian ganglion, and peripheral trigeminal nerve [24]. Many cases are probably due to compression or irritation of the entry zone of the trigeminal nerve root (e.g., by a multiple sclerosis plaque, brainstem infarction, cerebellopontine angle tumor, cavernous malformation, or an aberrant blood vessel, most frequently the superior cerebellar artery) [23,36,43,49,54,67,85]. Among 2,972 patients with trigeminal neuralgia in one series, 296 had tumors causing the facial pain [13]. The patients with tumors causing trigeminal neuralgia were younger than the patients with idiopathic pain, but gender and pain distributions were similar. Meningiomas and posterior fossa tumors were the most common tumors causing trigeminal pain [13]. Distortion of the trigeminal sensory root secondary to brainstem displacement may cause trigeminal neuralgia in patients with Chiari malformation or basilar invagination (e.g., due to osteogenesis imperfecta) [74, 79]. However, a patient with a Chiari’s type I malformation presented with trigeminal neuralgia thought to be because of compression of the trigeminal nucleus [83]. Trigeminal neuralgia owing to pontine infarction, with the lesion transecting the central trigeminal pathways, has also been described [76]. In another case, trigeminal neuralgia was due to pontine infarct affecting the intramedullary portion of the left trigeminal nerve [47].
In some patients with multiple sclerosis, trigeminal neuralgia paroxysms may be triggered by auditory stimuli [43]. Lesions in these patients were in the pons affecting the ipsilateral lateral lemniscus and trigeminal pathway. The lateral spread of the impulse within the demyelinating pontine lesion is the likely explanation for this phenomenon.
Occasionally, patients who are destined eventually to develop trigeminal neuralgia may have prodromal pain of toothache or sinusitis character lasting up to several hours (pretrigeminal neuralgia) [31]. This pain may be triggered by jaw movements or by drinking hot or cold liquids; typical trigeminal neuralgia then develops days (or even years) later in the same distribution.
Lesions Affecting the Gasserian Ganglion
Lesions of the middle cranial fossa (e.g., tumor, herpes zoster, sarcoidosis, syphilis, tuberculosis, arachnoiditis, trauma, abscess) may directly damage the gasserian ganglion in Meckel’s cave [19]. Pain, often severe and paroxysmal, is the most characteristic finding and may be hemifacial or involve only select divisions of the trigeminal nerve (especially the maxillary and mandibular divisions). Paresthesias and numbness may also occur, often starting close to the midline on the upper lip and chin and progressing laterally to involve the anterior ear. Sensory loss occurs in the division or divisions affected, and unilateral pterygoid and masseter paresis may occur. Other cranial nerves (especially the abducens nerve) may also be affected. Vascular compromise of the ganglion, causing isolated facial numbness, has been described with a spontaneous dural external carotid-cavernous sinus fistula [80]. Multiple cranial neuropathies, including variable affection of the trigeminal divisions, may occur with primary amyloidosis [98], and bilateral trigeminal neuropathies associated with bilateral abducens nerve palsies have been described with Tangier disease [91].
A unilateral or bilateral trigeminal sensory neuropathy may be seen with Sjögren’s syndrome, rheumatoid arthritis, systemic sclerosis, mixed connective tissue disease, systemic lupus erythematosus, Churg–Strauss syndrome, and dermatomyositis [29,42,60,89]. Facial numbness with or without paresthesias, often associated with facial pain, is most often seen in a maxillary distribution. Occasionally, symptoms are bilateral. The trigeminal symptoms develop before symptoms of the connective tissue disease in 7% and concurrent with the symptoms of connective tissue disease in 47% of the patients [42]. Numbness, often associated with facial pain, is most often noted in a maxillary distribution; oropharyngeal involvement may be prominent, and occasionally symptoms may be bilateral. Trigeminal sensory neuropathy may be distinguished from other conditions associated with facial numbness by its sparing of the muscles of mastication, frequent bilaterality, occasional disregard for trigeminal boundaries, and negative neuroimaging studies. Half of the patients complain of altered or absent taste, but when tested, primary gustatory sensibility is present. The lesion is suspected to involve the trigeminal ganglion or proximal part of the main trigeminal divisions and is perhaps related to the capillaries of the trigeminal ganglion being more permeable than the brain capillaries (blood–brain barrier) to abnormal proteins [60].
Trigeminal injury may occur with penetrating or blunt head trauma [50]. For example, a blow to the auriculotemporal area may rarely cause an isolated, complete sensory and motor trigeminal neuropathy [87].
Raeder’s Paratrigeminal Syndrome
This syndrome is composed of two essential components: unilateral oculosympathetic paresis and evidence of trigeminal involvement on the same side [69]. The former consists of miosis and ptosis but differs from the typical Horner syndrome in that facial anhidrosis is absent because the sudomotor fibers to the face that travel extracranially with the external carotid artery are spared. The unilateral head, facial, or retro-orbital pain related to trigeminal dysfunction may be associated with evidence of involvement of other cranial nerves (e.g., cranial nerves IV and VI). This syndrome is usually due to lesions in the middle cranial fossa, especially in the region between the trigeminal ganglion and the internal carotid artery, near the petrous apex. It may also be caused by lesions of the gasserian ganglion. The usual etiologies include tumor, aneurysm, trauma, and infection (e.g., Lyme disease) [71].
Gradenigo’s Syndrome
Lesions located at the apex of the temporal bone, especially metastasis, osteitis, or leptomeningitis associated with otitis media, may cause damage to the ophthalmic division of the trigeminal nerve and the nearby abducens nerve (Gradenigo’s syndrome) [22]. Pain and sensory disturbance in the upper part of the face (ophthalmic distribution) are then associated with ipsilateral lateral rectus palsy. Oculosympathetic paresis (without anhidrosis) may also occur ipsilaterally if the lesions extend to involve sympathetic fibers. Other etiologies for this syndrome include trauma and tumor.
The Cavernous Sinus Syndrome
Lesions within the cavernous sinus (e.g., tumor, carotid aneurysm, trauma, carotid-cavernous fistula, infection) may damage the ophthalmic and maxillary divisions of the trigeminal nerve and the abducens, trochlear, and oculomotor nerves. Total unilateral ophthalmoplegia, usually starting with abducens nerve involvement, if the lesion originates laterally, or oculomotor palsy, if the lesion proceeds from the sella, is then associated with pain, paresthesias, and sensory loss in the distribution of the ophthalmic and, less often, the maxillary divisions of the trigeminal nerve. Occasionally, oculosympathetic paresis (without anhidrosis) may also occur. Because the mandibular nerve is spared, no masticatory paresis is evident.
The Superior Orbital Fissure Syndrome
The abducens, trochlear, and oculomotor nerves as well as the ophthalmic division of the trigeminal nerve pass through the superior orbital fissure. Therefore, lesions at the superior orbital fissure (e.g., tumor, trauma, aneurysm, infection) may cause complete (external and internal) ophthalmoplegia associated with pain, paresthesias, and sensory loss in the ophthalmic cutaneous distribution. Occasionally, oculosympathetic paresis (without anhidrosis) may occur because of the involvement of the sympathetic fibers. Exophthalmos, due to blockade of the ophthalmic veins, and blindness, due to extension of the pathologic process to involve the optic canal, may also occur.
Except for the occasional instance of involvement of the maxillary division of the trigeminal nerve in the cavernous sinus syndrome, the superior orbital fissure syndrome and the cavernous sinus syndrome usually cannot be differentiated clinically without the use of neuroradiologic procedures.
Lesions Affecting the Peripheral Branches of the Trigeminal Nerve
The ophthalmic division of the trigeminal nerve may be damaged in the middle cranial fossa, at the temporal bone apex, at the lateral wall of the cavernous sinus, in the superior orbital fissure, or distally in the face. Localization of ophthalmic branch lesions in the former regions is made by associated cranial nerve findings, whereas very distal (e.g., facial) lesions result in sensory disturbances that are confined to the cutaneous supply of the ophthalmic division or its branches (e.g., the nasociliary, frontal, and lacrimal nerves) [100]. Some degree of corneal hypesthesia may occur in 45% of patients with diabetes [7] and, indeed, bilateral corneal erosions and complete corneal anesthesia may be the presenting feature of diabetic neuropathy [21].
The maxillary division of the trigeminal nerve may be damaged at the lower lateral wall of the cavernous sinus, at the foramen rotundum, in the pterygopalatine fossa, in the floor of the orbit, at the infraorbital foramen, or in the face. Numbness or discomfort in a maxillary distribution may be the initial presentation of a nasopharyngeal tumor [93], as these tumors often arise in the lateral nasopharyngeal wall (fossa of Rosenmüller) and extend through the foramen lacerum to involve the region of the middle cranial fossa and cavernous sinus. Lesions affecting this nerve in the cavernous sinus usually affect other cranial nerves as well. More distal lesions (e.g., infraorbital nerve damage secondary to maxillary fracture) result in sensory disturbances that are confined to the cutaneous supply of the maxillary nerve. Lesions in the infraorbital foramen may cause the numb cheek syndrome [11,100], in which numbness involves one cheek and the upper lip in an infraorbital nerve distribution. The hypesthesia in this syndrome may also involve the medial and lateral upper incisors and canine teeth and adjacent gingiva but spare more posterior teeth and gums (e.g., the molar and premolar teeth and gums that are innervated by the posterior and middle superior alveolar nerves). In two-thirds of patients, the numb cheek syndrome heralded recurrent squamous cell carcinoma of the skin [11]. Because of the proximity of distal branches of the facial nerve to the infraorbital nerve, lesions of the face, especially squamous cell carcinoma, may cause paresis of the muscles of the upper lip and angle of the mouth with ipsilateral lower lid droop accompanied by cheek numbness (the numb cheek–limp lower lid syndrome) [8]. Infraorbital nerve-distribution pain may also be a complication of laser in situ keratomileusis, probably because of manipulation of the eyelid speculum causing nerve injury [66].
Musicians who play brass instruments (trumpet, French horn, trombone, tuba) exert force on the lip with the mouthpiece of the instrument. This pressure may injure the anterior superior alveolar nerve resulting in upper lip numbness and pain (trumpet player’s neuropathy) [33,61].
The mandibular division of the trigeminal nerve may be damaged in the foramen ovale, in the zygomatic fossa, or in the face. Lesions affecting these regions result in sensory disturbances confined to the cutaneous supply of the mandibular division associated with ipsilateral masticatory paralysis. A syndrome of isolated mental neuropathy (the syndrome of the numb chin or Roger’s sign) [1,4,10,25,34,46,63,100] consists of pain, swelling, and numbness in the jaw (lower lip, chin, and mucous membrane on the inside of the lip). This syndrome is usually seen in patients with systemic cancer (especially lymphoreticular neoplasms and carcinoma of the breast and lung) and may be due to compression of the mental or the inferior alveolar nerves by metastases to the jaw [10,63,81], intracranial involvement of the mandibular nerve by base-of-skull lesions [40,41], leptomeningeal seeding [84], or neoplastic perineural infiltration of the mental nerve [58]. A numb chin, due to inferior alveolar nerve damage, may herald a relapse of multiple myeloma [68] or be the presenting sign of Burkitt’s lymphoma in human immunodeficiency virus infection [6]. Proximal versus distal origin of the syndrome depends on the presence or absence of any other cranial nerve (e.g., cranial nerve VI or VII) involvement; with a proximal lesion, involvement of other cranial nerves or signs related to leptomeningeal seeding may be expected rather than the isolated finding of hypesthetic chin associated with a distal lesion [63]. The pattern of oral numbness may also help differentiate between distal and proximal involvement of the trigeminal system [10]. The incisive nerve, which continues in the inferior alveolar canal beyond the mental foramen, innervates the incisor, canine, and bicuspid teeth, and their numbness probably indicates distal involvement of the mandibular nerve; on the other hand, lesions within the central nervous system may produce dissociation of sensory modalities and may spare dental sensation [39]. The estimated frequency of the numb chin syndrome in breast cancer is approximately 4% and is usually associated with a poor prognosis [46]. Therefore, patients who develop a nontraumatic mental neuropathy should undergo a careful search for malignancy, and chin numbness that occurs in a patient known to have cancer may indicate relapse and metastasis. Another study found that the numb chin syndrome was most often due to dental causes, including following dental anesthesia or as a complication of a dental procedure, pressure from ill-fitting dentures in an edentulous atrophic mandible in an elderly patient, infection of the root of a tooth, acute or chronic osteomyelitis of the mandible, or odontogenic or nonodontogenic tumors or cysts of the mandible [4]. The numb chin syndrome has also been described as the first sign of giant cell arteritis [37].
In a retrospective evaluation of 42 patients with cancer and numb chin syndrome, breast cancer comprised 64% of the primary tumors, and lymphoproliferative neoplasms comprised 14% [63]. Fifty percent of the patients had mandibular metastases, 14% had base-of-skull bone lesions, and 22% had leptomeningeal seeding. The numb chin syndrome was a late manifestation of malignancy, associated with disease progression in 67% of the patients or heralding a relapse, which was often confined to the leptomeninges, in 31%. Median survival after its diagnosis was 5 months when due to bone metastases and 12 months if associated with leptomeningeal seeding [63]. Bilateral numb chin syndrome has been described as the initial symptom of Burkitt’s cell acute lymphoblastic leukemia; postmortem examination revealed direct infiltration of the mandibular nerves by leukemic cells [58]. A burning sensation of the lower lip followed later by numbness may also occur with sickle cell disease, probably because of infarction of the inferior alveolar nerve or the mental nerve in its canal [30].
The peripheral branches of the trigeminal nerve are most often damaged in isolation by tumors or by fractures of the facial bones or skull. Subacute facial numbness may be the heralding symptom of an expanding tumor that involves the trigeminal nerve fibers [57]. Three patients have been described who developed subacute facial numbness as the heralding symptom of malignancy: an isolated mental neuropathy as a result of metastatic bone destruction from a renal cell carcinoma, a sensorimotor trigeminal neuropathy caused by direct compression of the semilunar ganglion by a cavernous hemangioma of Meckel’s cave, and facial numbness as the presenting manifestation of a primary brainstem lymphoma. Cutaneous carcinomas of the face (e.g., squamous cell carcinoma, basal cell carcinoma) and some nasopharyngeal carcinomas may present with facial dysesthesias in the distribution of any branch of the trigeminal nerve [96]. These reports demonstrate that it is difficult, initially, to differentiate a “benign” trigeminal neuropathy from serious conditions associated with a poor prognosis.
Tongue numbness (unilateral or bilateral), often of sudden onset, may be seen in temporal arteritis [12]. It is likely that ischemia of the brainstem or lingual nerve is responsible. Also, tongue numbness may be part of the neck-tongue syndrome, in which sudden turning of the head results in pain in the upper neck and occiput accompanied by numbness of the ipsilateral half of the tongue [59,73]. This syndrome is thought to be because of irritation of the second cervical dorsal root, which carries proprioceptive fibers from the tongue through the hypoglossal nerve, and its communications with the second root. Lingual pseudoathetosis may occur with the neck-tongue syndrome, presumably because of lingual deafferentation [73].
Periodic hemilingual numbness may occur in attacks associated with simultaneous submandibular swelling and transient profuse salivation at the termination of the event [86]. This periodic numbness is presumably due to intermittent compression of the lingual nerve due to sialolithiasis.
Numbness of half of the tongue may also occur with lingual nerve trauma [70] and with trigeminal sensory neuropathy related to collagen vascular diseases [60] (see preceding text). Lingual neuropathy may result in hemilingual sensory loss, pain, dysesthesia, paresthesia, and dysgeusia [38]. Lingual neuropathy may follow lower wisdom teeth extraction and other dental procedures, surgery of the mandibular ramus, mandibular block anesthesia, endotracheal intubation, or temporomandibular joint disc displacement [38,62]. The lingual nerve may also be entrapped in the lateral pterygoid muscle [62]. Bilateral anterior lingual hypogeusia and hypesthesia has been described following a dental procedure [88]. In this case, branches of the lingual nerve were damaged, as were the chorda tympani branches of the facial nerves, which convey taste from the anterior two-thirds of the tongue. Ageusia accompanied by numbness of both sides of the anterior tongue and perioral region (the latter due to trigeminal dysfunction confined to the lingual branches) may be the initial manifestation of Guillain-Barré syndrome [16].
Patients may rarely develop neuromyotonia of the floor of the mouth after irradiation of the motor branch (V3) of the trigeminal nerve [26,64]. The neuromyotonia manifests as episodic or sustained muscle contraction due to peripheral nerve dysfunction. The episodic involuntary contraction may also affect the lower facial and masticatory (masseter) muscles [64].
Jaw Drop
Preferential weakness of the jaw-closure muscles (temporalis and masseter muscles) with preservation of jaw-opening muscles (pterygoid muscles), when severe, causes the jaw to hang open or be “dropped.” Patients have fatigue with chewing and the need to manually support the jaw. This neurologic sign has been most often described in myasthenia gravis but may also occur in amyotrophic lateral sclerosis (ALS) or myotonic dystrophy [95]. In ALS, jaw drop is rarely an early feature; rather, the tongue muscles are frequently the earliest and most severely involved, suggesting that the hypoglossal motor nerve cells are particularly susceptible in ALS. Some patients with Kennedy’s disease may present with jaw drop or isolated jaw-closure weakness [95]. Kennedy’s disease, or spinal and bulbar muscular atrophy, is an inherited X-linked degenerative disease of sensory and motor neurons caused by a trinucleotide (CAG) repeat expansion in the first exon of the androgen receptor gene. The distinguishing clinical features of the disease include slowly progressive proximal greater than distal limb weakness, bulbar weakness involving primarily facial and tongue muscles, perioral fasciculations, sensory involvement, elevated creatine kinase level, and signs of androgen insensitivity (gynecomastia and testicular atrophy) [95].
References
1. Acarin N. Roger’s sign. Chin neuropathy. Medicina Clin (Barcelona) 1985;84:546.
2. Auger RG, Litchy WJ, Cascino TL, et al. Hemimasticatory spasm: clinical and electrophysiological observations. Neurology 1992;42:2263–2266.
3. Auger RG, McManis PG. Trigeminal sensory neuropathy associated with decreased oral sensation and impairment of the masseter inhibitory reflex. Neurology 1990;40:759–763.
4. Bar-Ziv J, Slasky BS. CT imaging of mental nerve neuropathy: the numb chin syndrome. Am J Radiol 1997;168:371–376.
5. Bekar A, Kocaeli H, Yilmaz E, et al. Trigeminal neuralgia caused by a pontine abscess: case report. Neurosurgery 2004;55:E1450–E1452.
6. Benito-León J, Simón R, Miera C. Numb chin syndrome as the initial manifestation of HIV infection. Neurology 1998;50:511–512.
7. Ben Osman N, Jeddi A, Sedai JA, et al. La cornee du diabetique. J FR Ophtalmol 1995;18:120–123.
8. Brazis PW, Vogler JB, Shaw KE. The “numb cheek-limp lower lid” syndrome. Neurology 1991;41:327–328.
9. Brodal A. Neurological anatomy in relation to clinical medicine, 3rd ed. New York: Oxford University Press, 1981:508–532.
10. Calverley JR, Mohnac A. Syndrome of the numb chin. Arch Intern Med 1963;112:819.
11. Campbell WW. The numb cheek syndrome: a sign of infraorbital neuropathy. Neurology 1986;36:421–423.
12. Caselli RJ, Hunder GG, Whisnant JP. Neurologic disease in biopsy-proven giant cell (temporal) arteritis. Neurology 1988;38:352–359.
13. Cheng TMW, Cascino TL, Onofrio BM. Comprehensive study of diagnosis and treatment of trigeminal neuralgia secondary to tumors. Neurology 1993;48: 2298–2302.
14. Clauw DJ, Naghel DJ, Umhau A, et al. Tryptophan-associated eosinophilic connective-tissue disease. A new clinical entity? JAMA 1990;263:1502–1506.
15. Combarros O, Berciano J, Oterino A. Pure sensory deficit with crossed oro-crural topography after pontine haemorrhage. J Neurol Neurosurg Psychiatry 1996;61: 534–535.
16. Combarros O, Pascual J, de Pablos C, et al. Taste loss as an initial symptom of Guillain-Barré syndrome. Neurology 1996;47:1604–1605.
17. Cruccu G, Fornarelli M, Manfredi M. Impairment of masticatory function in hemiplegia. Neurology 1988;38:301–306.
18. Cruccu G, Inghilleri M, Berardelli A, et al. Pathophysiology of hemimasticatory spasm. J Neurol Neurosurg Psychiatry 1994;57:43–50.
19. Cuneo HM, Rand CW. Tumors of the gasserian ganglion. Tumor of the left gasserian ganglion associated with enlargement of the mandibular nerve. A review of the literature and case report. J Neurosurg 1952; 9:423.
20. Currier RD, Giles CL, Dejong RN. Some comments on Wallenberg’s lateral medullary syndrome. Neurology 1961;11:778–791.
21. Daniels CS, Fraser SG, Dart JKG. An unusual presentation of diabetic neuropathy. Br J Ophthalmol 2001; 85:625.
22. Davé AV, Diaz-Marchan PJ, Lee AG. Clinical and magnetic resonance imaging features of Gradenigo syndrome. Am J Ophthalmol 1997;124:568–570.
23. Deshmukh VR, Hott JS, Tabrizi P, et al. Cavernous malformation of the trigeminal nerve manifesting with trigeminal neuralgia: case report. Neurosurgery 2005;56:E623.
24. Deshpande S, Kaptain GJ, Pobereskin LH. Temporal glioblastoma causing trigeminal neuralgia. Case illustration. J Neurosurg 1999;91:515.
25. Diago MP, Sebastian JVB, Giner AA, et al. Mental nerve neuropathy in systemic cancer. Report of three cases. Oral Surg 1990;69:48.
26. Diaz JM, Urban ES, Schiffman JS, et al. Post-irradiation neuromyotonia affecting trigeminal nerve distribution: an unusual presentation. Neurology 1992;42: 1102–1104.
27. Dick MT, Gonyea E. Trigeminal neurotrophic ulceration with Wallenberg’s syndrome. Neurology 1990; 40:1634–1635.
28. Ebersbach G, Kabus C, Schelosky L, et al. Hemimasticatory spasm in hemifacial atrophy: diagnostic and therapeutic aspects in two patients. Mov Disord 1995;10:504–507.
29. Förster C, Brandt T, Hund E, et al. Trigeminal sensory neuropathy in connective tissue disease: evidence for the site of the lesion. Neurology 1996;46: 270–271.
30. Friedlander AH, Genser L, Swerdloff M. Mental nerve neuropathy: a complication of sickle-cell crisis. Oral Surg 1980;49:15–17.
31. Fromm GH, Graff-Radford SB, Terrence CF, et al. Pretrigeminal neuralgia. Neurology 1990;40:1493–1495.
32. Frontera JA, Palestrant D. Acute trismus associated with Foix-Marie-Chavany syndrome. Neurology 2006; 66:454–455.
33. Frucht S. Anterior superior alveolar neuropathy: an occupational neuropathy of the embouchure. J Neurol Neurosurg Psychiatry 2000;69:562–567.
34. Furukawa T. Numb chin syndrome in the elderly. J Neurol Neurosurg Psychiatry 1990;53:173.
35. Gardner WJ. Trigeminal neuralgia. Clin Neurosurg 1968;1:1–56.
36. Gass A, Kitchen N, MacManus DG, et al. Trigeminal neuralgia in patients with multiple sclerosis: lesion localization with magnetic resonance imaging. Neurology 1997;49:1142–1144.
37. Genereau T, Lortholary O, Biousse V, et al. Numb chin syndrome as first sign of temporal arteritis. J Rheumatol 1999;26:1425–1426.
38. Graff-Radford SB, Evans RW. Lingual nerve injury. Headache 2003;43:975–983.
39. Graham SH, Sharp FR, Dillon W. Intraoral sensation in patients with brainstem lesions: role of the rostral spinal trigeminal nuclei in pons. Neurology 1988;38:1529–1533.
40. Greenberg HS, Deck MD, Vikram B, et al. Metastasis to the base of the skull: clinical findings in 43 patients. Neurology 1981;31:530–537.
41. Hall SM, Budzar AU, Blumenschein GR. Cranial nerve palsies in metastatic breast cancer due to osseous metastasis without intracranial involvement. Cancer 1983;52:180–184.
42. Hagen NA, Stevens JC, Michet CJ. Trigeminal sensory neuropathy associated with connective tissue disease. Neurology 1990;40:891–896.
43. Hartmann M, Rottach KG, Wohlgemuth WA, et al. Trigeminal neuralgia triggered by auditory stimuli in multiple sclerosis. Arch Neurol 1999;56:731–733.
44. Hendes AM, Thiebot B, Laurent PM. Neurotrophic ulceration in the area of the trigeminal nerve in Wallenberg’s syndrome. Ann Dennatl Veneral 1988;115: 143–149.
45. Holtzman RN, Zablozki V, Yang WC, et al. Lateral pontine tegmental hemorrhage presenting as isolated trigeminal sensory neuropathy. Neurology 1987;37: 704–706.
46. Horton J, Means ED, Cunningham TJ, et al. The numb chin in breast cancer. J Neurol Neurosurg Psychiatry 1973;36(2):211–216.
47. Iizuka O, Hosokai Y, Mori E. Trigeminal neuralgia due to pontine infarction. Neurology 2006;66:48.
48. Ishii K, Tamaoka A, Shoji S. Dorsolateral pontine segmental infarction presenting as isolated trigeminal sensory neuropathy. J Neurol Neurosurg Psychiatry 1998;65:702.
49. Jannetta PJ, Rand RW. Arterial compression of the trigeminal nerve at the pons in patients with trigeminal neuralgia. J Neurosurg 1967;26(Suppl.):159.
50. Jefferson G, Schorstein J. Injuries to the trigeminal nerve, its ganglion, and its division. Br J Surg 1955; 62:561.
51. Kaufman MD. Masticatory spasm in facial hemiatrophy. Ann Neurol 1980;7:585–587.
52. Kim JS, Choi-Kwon S. Sensory sequelae of medullary infarction. Differences between lateral and medial medullary syndrome. Stroke 1999;30:2697–2703.
53. Kim HJ, Jeon BS, Lee K-W. Hemimasticatory spasm associated with localized scleroderma and facial hemiatrophy. Arch Neurol 2000;57:576–580.
54. Kim JS, Kang JH, Lee MC. Trigeminal neuralgia after pontine infarction. Neurology 1998;51:1511–1512.
55. Kim JS, Lee JH, Choi CG. Patterns of lateral medullary infarction. Vascular lesion-magnetic resonance imaging correlation of 34 cases. Stroke 1998; 29:645–652.
56. Kim JS, Lee JH, Lee MC. Patterns of sensory dysfunction in lateral medullary infarction. Clinical-MRI correlation. Neurology 1997;49:1557–1563.
57. Kuntzer T, Bogousslavsky J, Rilliet B, et al. Herald facial numbness. Eur Neurol 1992;32:297–301.
58. Kuroda Y, Fujiyama F, Ohyama T, et al. Numb chin syndrome secondary to Burkitt’s cell acute leukemia. Neurology 1991;41:453–454.
59. Lance JW, Anthony M. Neck-tongue syndrome on sudden turning of the head. J Neurol Neurosurg Psychiatry 1980;43:97–101.
60. Lecky BRF, Hughes RAC, Murray NMF. Trigeminal sensory neuropathy. A study of 22 cases. Brain 1987; 110:1463–1485.
61. Lederman RJ. Trumpet player’s neuropathy. JAMA 1987;257:1526.
62. Liguori R, Cevoli S, Montagna P. Electroneurographic investigation of the mandibular nerve in lingual neuropathy. Muscle Nerve 1998;21:410–412.
63. Lossos A, Siegal T. Numb chin syndrome in cancer patients: etiology, response to treatment, and prognostic significance. Neurology 1992;42:1181–1184.
64. Marti-Fàbregas J, Montero J, López-Villegas D, et al. Post-irradiation neuromyotonia in bilateral facial and trigeminal nerve distribution. Neurology 1997;48: 1107–1109.
65. Masjuan J, Barón M, Lousa M, et al. Isolated pontine infarction with prominent ipsilateral midfacial sensory signs. Stroke 1997;28:649–651.
66. McCulley TJ, Eifrig CWG, Schatz NJ, et al. Infraorbital nerve palsy: a complication of laser in situ keratomileusis. Am J Ophthalmol 2002;134: 264–265.
67. Meaney JFM, Eldridge PR, Dun LT, et al. Demonstration of neovascular compression in trigeminal neuralgia with magnetic resonance imaging. Comparison with surgical findings in 52 consecutive operative cases. J Neurosurg 1995;83:799–805.
68. Miera C, Benito-León J, de la Fuente M, et al. Numb chin syndrome heralding myeloma relapse. Muscle Nerve 1997;20:1603–1606.
69. Mokri B. Raeder’s paratrigeminal syndrome—original concept and subsequent deviations. Arch Neuro 1982;39:395–399.
70. Mozsary PG, Middleton RA. Microsurgical reconstruction of the lingual nerve. J Oral Maxillofac Surg 1984;42:415–420.
71. Murphy MA, Szabados EM, Mitty JA. Lyme disease associated with postganglionic Horner syndrome and Raeder paratrigeminal neuralgia. J Neuro-Ophthalmology 2007;27:123–124.
72. Nakamura K, Yamamoto T, Yamashita M. Small medullary infarction presenting as painful trigeminal neuropathy. J Neurol Neurosurg Psychiatry 1996;61: 138.
73. Orrell RW, Marsden CD. The neck-tongue syndrome. J Neural Neurosurg Psychiatry 1994;57: 348–352.
74. Papanastassiou A, Schwartz RB, Friedlander RM. Chiari I malformation as a cause of trigeminal neuralgia: case report. Neurosurgery 2008;63:E614–E615.
75. Parisi L, Valente G, Dell’Anna C, et al. A case of facial hemiatrophy associated with linear scleroderma and homolateral masseter spasm. Ital J Neurol Sci 1987; 8:63–65.
76. Peker S, Akansel G, Sun I, et al. Trigeminal neuralgia due to pontine infarction. Headache 2004;44: 1043–1045.
77. Powers JM, Young GF, Bass EB Jr, et al. Atypical nemaline myopathy with temporomandibular ankylosis. Neurology 1980;30:971–975.
78. Pullicino PM, Jacobs L, McCall WD Jr, et al. Spontaneous palpebromandibular synkinesia: a localizing clinical sign. Ann Neurol 1994;35:222–228.
79. Reilly MM, Valentine AR, Ginsberg L. Trigeminal neuralgia associated with osteogenesis imperfecta. J Neurol Neurosurg Psychiatry 1995;58:665.
80. Rizzo M, Bosch EP, Gross CE. Trigeminal sensory neuropathy due to dural external carotid cavernous sinus fistula. Neurology 1982;32:89–91.
81. Rohrer MD, Colyer J. Mental nerve paresthesia: symptom for a widespread skeletal metastatic adenocarcinoma. J Oral Surg 1981;39:442–445.
82. Rorick MB, Chandar K, Colombi BJ. Inflammatory trigeminal sensory neuropathy mimicking trigeminal neurinoma. Neurology 1996;46:1455–1457.
83. Rosetti P, Oulad Ben Taib N, Brotchi J, et al. Arnold Chiari type I malformation presenting as a trigeminal neuralgia: case report. Neurosurgery 1999;44:1122–1124.
84. Rubenstein MK. Cranial mononeuropathy as the first sign of intracranial metastases. Ann Intern Med 1969; 70:49.
85. Rushton JG, Olafson RA. Trigeminal neuralgia associated with multiple sclerosis. Report of 35 cases. Arch Neurol 1965;13:383–386.
86. Sadler RM, Curran T, Pryse-Phillips WEM. Numbness of half of the tongue. Can J Neurol Sci 1986;13: 107–108.
87. Schecter AD, Anziska B. Isolated complete post-traumatic trigeminal neuropathy. Neurology 1990;40: 1634.
88. Schwankhaus JD. Bilateral anterior lingual hypogeusia hypesthesia. Neurology 1993;43:2146.
89. Sehgal M, Swanson JW, DeRemee RA, et al. Neurologic manifestations of Churg-Strauss syndrome. Mayo Clin Proc 1995;70:337–341.
90. Seidel E, Hansen C, Urban PP, et al. Idiopathic trigeminal sensory neuropathy with gadolinium enhancement in the cisternal segment. Neurology 2000;54:1191–1192.
91. Siao PTC, Cros DP, Lees RS. Case records of the Massachusetts General Hospital. N Engl J Med 1996; 334:1389–1394.
92. Singer PA, Chikarmane A, Festoff BW, et al. Trismus—an unusual sign of polymyositis. Arch Neurol 1985;42:1116–1118.
93. Smith JL. The eye in nasopharyngeal tumors. Neuro-ophthalmology Audio J 1986;8:9.
94. Sogg RL, Hoyt WF, Boldrey E. Spastic paretic facial contracture: a rare sign of brainstem tumor. Neurology 1963;13:607–612.
95. Sumner CJ, Fischbeck KH. Jaw drop in Kennedy’s disease. Neurology 2002:59:1471–1472.
96. ten Hove MW, Glaser JS, Schatz NJ. Occult perineural tumor infiltration of the trigeminal nerve. Diagnostic considerations. J Neuro-Ophthalmology 1997;17:170–177.
97. Thompson PD, Obeso JA, Delgado G, et al. Focal dystonia of the jaw and the differential diagnosis of unilateral jaw and masticatory spasm. J Neurol Neurosurg Psychiatry 1986;49:651–656.
98. Traynor AE, Gertz MA, Kyle RA. Cranial neuropathy associated with primary amyloidosis. Ann Neurol 1991;29:451–454.
99. Vaudens P, Bogousslavsky J. Face-arm-trunk-leg sensory loss limited to the contralateral side in lateral medullary infarction: a new variant. J Neurol Neurosurg Psychiatry 1998;65:255–257.
100. Warden KF, Parmar H, Trobe JD. Perineural spread of cancer along the three trigeminal divisions. J Neuro-Ophthalmol 2009;29:300–307.
< div class='tao-gold-member'>









