CHAPTER 66 Hemispheric Disconnection Procedures
Development
The first anatomic hemispherectomy was done for glioma surgery by Dandy in 1928,1 and Krynauw performed the first resection for drug-resistant epilepsy in 1950.2 A change from anatomic hemispherectomy, frequently used in the 1950s and 1960s and in which one whole hemisphere was removed (with or without the basal ganglia block), to the less invasive disconnection techniques has taken place since the early 1990s. An important step in the evolution from large resection to large disconnection occurred with development of the technique of functional hemispherectomy, introduced by Rasmussen.3 It included removal of two larger brain segments (temporal lobe and central suprasylvian tissue block), combined with callosotomy and disconnection of the frontal, parietal, and occipital lobes. Although the hemisphere as such is no longer totally removed, the effect of this surgery is functionally equivalent to total hemispherectomy. For the aims of epilepsy surgery, it is not important whether parts of the hemisphere are removed or just disconnected because to achieve a seizure-free outcome, it is sufficient to disconnect the ictogenic cortical part of the hemisphere from its connections to the other hemisphere and the long tracts to the basal ganglia. It makes sense to limit use of the term functional hemispherectomy to Rasmussen’s and Mathern’s techniques.4 The latter consists of a large central peri-insular resection, including the basal ganglia block and the temporal lobe, combined with disconnection of the frontal, parietal, and occipital lobes.
After a period of increased use of anatomic hemispherectomy in the 1960s and 1970s, a high rate of late complications was described, such as superficial cerebral hemosiderosis (SCH), occlusion of the foramen of Monro and aqueduct, development of hydrocephalus and its complications,5–8 and even fatal outcomes.3,9,10 In 27 anatomic hemispherectomies performed in Montreal between 1952 and 1968 with long-term follow-up, hydrocephalus developed in 52% of patients, 33% from SCH and 19% from other causes. Three of nine patients with SCH died of hydrocephalus.11 SCH is thought to be caused by recurrent minor intracranial bleeding in the large cavity as a result of the resection surgery. Such bleeding may occur after minor head trauma or intermittent rises in intracranial pressure from physiologic events such as sneezing.
Less Resection—More Disconnection
Because of the considerable mortality from these complications, the use of anatomic hemispherectomy decreased until the 1970s, when Rasmussen described an alternative technique based on the observation that in patients with multilobectomies, these complications did not occur. Because the frontal lobe and the parieto-occipital lobe are left in situ, a lower incidence of hydrocephalus and hemosiderosis was expected and later confirmed. Rasmussen demonstrated in his patient series that seizure-free outcome rates after anatomic and functional hemispherectomies were very similar, 83% and 85%, respectively, but the rate of complications from increased intracranial pressure was reduced from 35% to 7%. A lower rate of SCH has been reported for functional hemispherectomy techniques after a follow-up of 20 years.3,10,12 Another approach to avoid these complications was the technique by Adams, who occluded the foramen of Monro, excised the choroidal plexus, and obliterated the subdural space by folding down the dura toward the midline and over the residual basal ganglia.13 The hemidecortication and hemicorticectomy techniques14–16 approached the problem of these complications by preserving nearly all of the ventricular system and leaving the cerebrospinal fluid (CSF) space uncontaminated.
It also makes sense to consider hemidecortication and hemicorticectomy separately from disconnection procedures (e.g., transsylvian/transventricular keyhole procedure,17 Delalande’s vertical parasagittal hemispherotomy, and perisylvian window techniques18,19), which usually include very little brain resection. Differentiation between mostly disconnective procedures and functional hemispherectomies3,4 or decortications, including larger resections, is useful because the latter procedures appear to have a different set of characteristics and disadvantages.
The modern transition to nearly exclusively disconnective techniques started in 1992 after a brief description of two quite different approaches developed independently by Schramm, Delalande, and their colleagues.20,21 This was followed in the mid-1990s by the first patient series treated via perisylvian techniques.17,19 Closely related to Villemure’s technique is a variation of a perisylvian resective approach by Shimizu and Maehara.18 Among the disconnective procedures, the technique by Delalande and associates (vertical parasagittal hemispherotomy)22 and the transsylvian transventricular keyhole procedures23 include minimal tissue removal and mostly consist of disconnections. The change to less resective procedures during the past 15 years is being made in many centers, and a number of reports have confirmed the initial results indicating that disconnection procedures are associated with shorter operative time, less blood loss, fewer intraoperative complications, and possibly a lower rate of hydrocephalus.
Indications, Patient Selection, and Timing
Causes
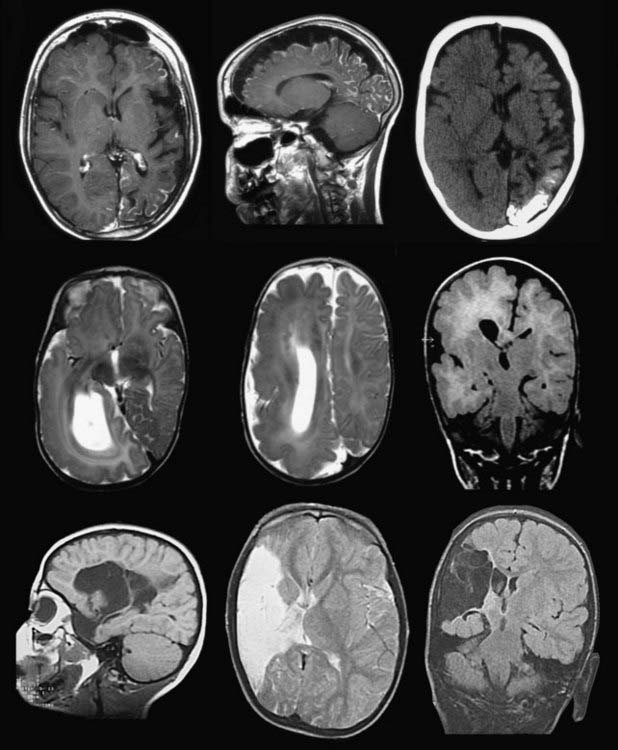
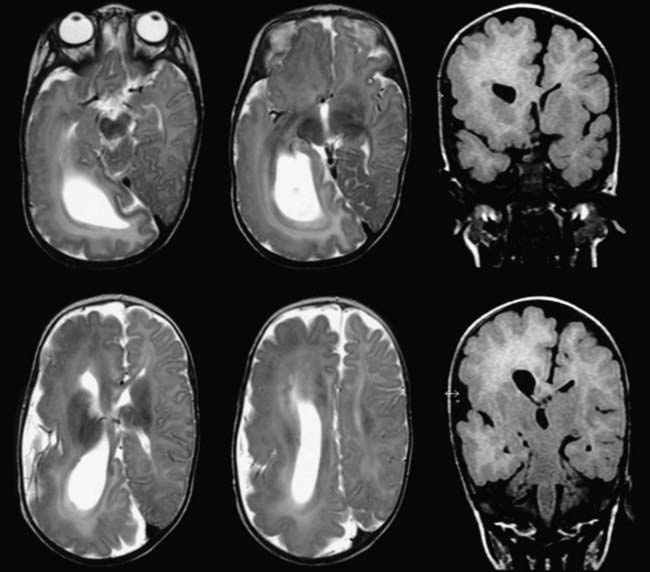
The diagnoses typical in these patients may be grouped into inborn, perinatal, or acquired conditions (Table 66-1) and include Sturge-Weber syndrome (SWS), developmental defects (multilobar cortical dysplasia, polymicrogyria, lissencephaly, hemimegalencephaly [HME]), cystic defects from intrauterine or perinatal infarction or hemorrhage, and hemiplegia-hemiconvulsion-epilepsy syndrome (HHE) (Fig. 66-1). Rasmussen’s encephalitis leads to hemiatrophy, which may also be posttraumatic, postencephalitic, or of unknown origin. The epilepsy syndromes associated with these lesions may include several seizure types occurring with different frequencies. Patients can have up to hundreds of seizures per day or suffer from focal status epilepticus (i.e., epilepsia partialis continua, typical of Rasmussen’s encephalitis).
TABLE 66-1 Etiology of Seizures
| Infarct, ischemia, porencephaly | 54 |
| Cortical dysplasia, migration disorders | 10 |
| Hemimegalencephaly | 12 |
| Sturge-Weber syndrome | 6 |
| Rasmussen’s encephalitis | 15 |
| Hemiatrophy, unclear etiology | 7 |
| Postencephalitic, HHE, or other | 6 |
| Total | 110 |
HHE, hemiplegia-hemiconvulsion-epilepsy syndrome.
Hemimegalencephaly
![]() Patients with HME usually have an enlarged hemisphere displaying different features of abnormal architecture. Parts of the ventricle may be enlarged, whereas others may be compressed by abnormal brain tissue. There may be pachygyria, polymicrogyria, grossly enlarged gyri, and areas of ectopic gray matter around the ventricle in the white matter (Fig. 66-E1).
Patients with HME usually have an enlarged hemisphere displaying different features of abnormal architecture. Parts of the ventricle may be enlarged, whereas others may be compressed by abnormal brain tissue. There may be pachygyria, polymicrogyria, grossly enlarged gyri, and areas of ectopic gray matter around the ventricle in the white matter (Fig. 66-E1).
Sturge-Weber Syndrome
SWS is a rare congenital disorder with a variable natural history. Its characteristic feature is leptomeningeal angiomatosis, and in a proportion of cases it may be associated with a facial nevus, the latter variant occasionally described as encephalotrigeminal angiomatosis. The clinical syndrome is characterized by a progressive neurological disorder consisting of epilepsy, cortical calcifications, cerebral atrophy, and if the epilepsy is untreatable, frequently the development of mental retardation. Seizures usually develop by the end of the first year, may respond to medical treatment initially, but often become resistant to drugs. The cerebral manifestations generally involve the occipital or parietal cortex, or both, but the entire cortex or large parts of it may be involved; however, the pathologic changes remain restricted to one hemisphere. Epilepsy surgery should be considered for patients with refractory seizures, but one has to differentiate between the need for hemispheric deafferentation and a multilobar or more restricted resection. Hemianopia may be present from the beginning or may develop together with hemiparesis during progression of the epileptic encephalopathy, which is associated with cognitive decline and mental retardation. Children with widespread hemispheric involvement are classic candidates for hemispheric deafferentation. The results from three multicenter series have been reported.24–26 Seizure-free rates after hemispherotomy were reported to be 100% in 8, 6, and 5 patients.24,25,27 In three larger series, rates of 80% to 82% were reported in 12, 28, and 70 patients.22,26,28
Rasmussen’s Encephalitis
The origin of Rasmussen’s encephalitis is unknown. The clinical syndrome is characterized by intractable epilepsy and progressive hemiparesis inexorably resulting in hemiplegia, mental decline, and hemispheric atrophy. The median age at onset in a multicenter study of 16 patients was 4.2 years with a range of 2 to 11 years.28 Most of the brain damage occurs during the first 8 to 12 months.29 Progression to hemiplegia or aphasia (or to both) and finally cognitive decline occur invariably. The seizure disorder may begin with generalized seizures, but focal seizures are most frequent and epilepsia partialis continua develops in a large proportion of patients. A characteristic histologic finding is a perivascular infiltrate of T lymphocytes, which is associated with destruction of neurons.30 Seventy-seven percent of 83 patients in that study were seizure free after hemispheric surgery. Once the diagnosis is suspected, the atrophic process may be staged by serial magnetic resonance imaging (MRI).31 A difficult decision on when to operate will arise. These patients are classic candidates for hemispheric deafferentation.
Contraindications
Hemispheric deafferentation is contraindicated if the presurgical evaluation cannot demonstrate that all typical ictal activity originates from the affected hemisphere. A relative contraindication may be seen in patients with independently arising seizures from the so-called healthy hemisphere. Occasional isolated contralateral seizure episodes may be acceptable because they do not automatically result from an independent seizure focus. Bilateral epileptogenic activity may be seen on the electroencephalogram in as many as 75% of patients, but it may be secondary and originate from the diseased hemisphere. Because bilateral involvement may represent an independent seizure focus and not just activity conducted from the diseased hemisphere, it is associated with a somewhat reduced probability of a seizure-free outcome after surgery. Nonetheless, high rates of freedom from seizures32 can still be achieved,33 as demonstrated in a large series in which 77% of patients with suspected bilateral disease were found to either be seizure free or have only “minor events.”34
Occasionally, the presence of incomplete hemianopia may be considered a contraindication, especially in older children. However, it has been our experience that patients who have grown up with hemianopia adjust well to this deficit. Mental retardation is no longer considered a contraindication in our institution and others.35
Surgical Techniques
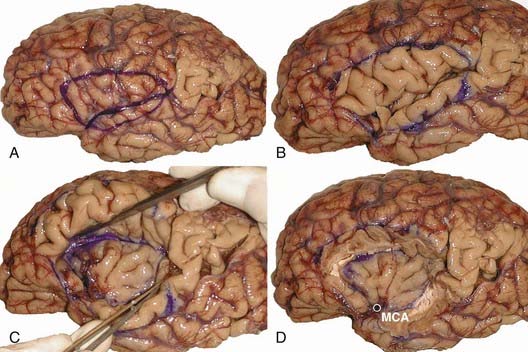
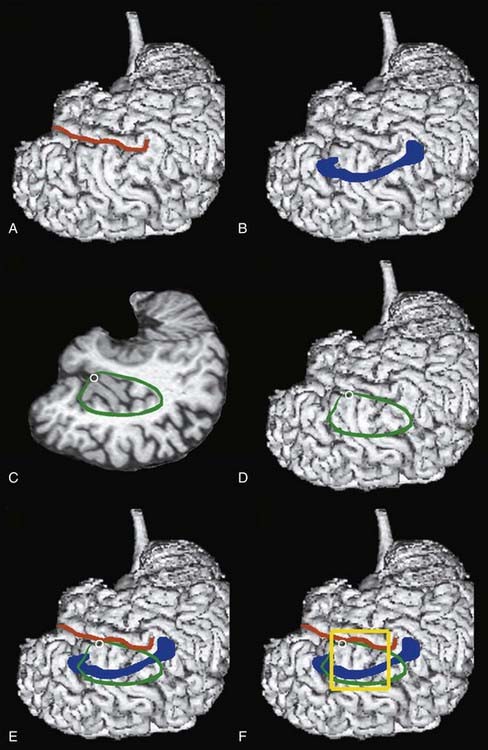
![]() The various forms of hemispheric deafferentation surgery currently in use all evolved from Rasmussen’s functional hemispherectomy technique. The principle behind this surgery is to forgo anatomic removal of brain tissue by instead disconnecting brain regions. One of the early variations of Rasmussen’s method was developed by Mathern’s group in Los Angeles.4 This procedure involved performing a large resection of the operculum, insula, underlying basal ganglia, and temporal lobe to achieve a functional hemispherectomy. An intermediate step short of a pure deafferentation procedure involves perisylvian techniques in which only restricted parts of the opercula (Fig. 66-E2D), corona radiata, or temporal lobe are resected. This is usually combined with disconnection of the frontal and parieto-occipital lobes and callosotomy, which is performed from within the ventricular system.17,18,36 The latest versions of these evolving procedures are two disconnection operations that involve little or no brain removal and extensive disconnections: transsylvian keyhole hemispheric deafferentation21,23 and central vertical hemispherotomy (Fig. 66-2).20,22
The various forms of hemispheric deafferentation surgery currently in use all evolved from Rasmussen’s functional hemispherectomy technique. The principle behind this surgery is to forgo anatomic removal of brain tissue by instead disconnecting brain regions. One of the early variations of Rasmussen’s method was developed by Mathern’s group in Los Angeles.4 This procedure involved performing a large resection of the operculum, insula, underlying basal ganglia, and temporal lobe to achieve a functional hemispherectomy. An intermediate step short of a pure deafferentation procedure involves perisylvian techniques in which only restricted parts of the opercula (Fig. 66-E2D), corona radiata, or temporal lobe are resected. This is usually combined with disconnection of the frontal and parieto-occipital lobes and callosotomy, which is performed from within the ventricular system.17,18,36 The latest versions of these evolving procedures are two disconnection operations that involve little or no brain removal and extensive disconnections: transsylvian keyhole hemispheric deafferentation21,23 and central vertical hemispherotomy (Fig. 66-2).20,22
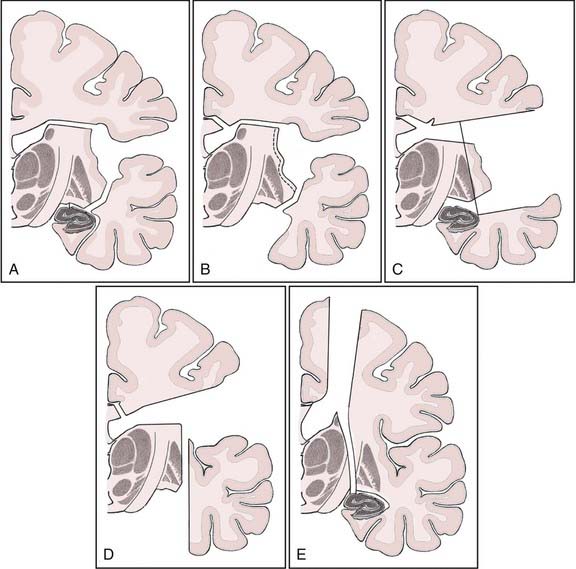
FIGURE 66-2 Schematic coronal views of the perisylvian and disconnection techniques. A, Step 1 of the transsylvian/transsulcal keyhole approach to the ventricle.23 B, Step 2 of the transsylvian/transventricular keyhole approach: facultative temporomesial resection, removal of the insular cortex, and paramedian callosotomy. C, Peri-insular window technique.36 D, Japanese variant of the peri-insular window technique.18 E, Vertical parasagittal hemispherotomy.22
(Modified from Schramm J. Hemispherectomy techniques. Neurosurg Clin N Am. 2001;37:113.)
Hemispheric Deafferentation Techniques
Transsylvian Keyhole Technique
This technique requires limited exposure of the brain and uses disconnection steps almost exclusively. A lateral transsylvian/transventricular approach is performed through a keyhole opening. A 4 × 4 or 4 × 5 cm craniotomy is performed superior to the sylvian fissure to gain access to the underlying insula, which has an average length of 49 to 57 mm,38 and to the corpus callosum, which has a maximal length of 7.5 cm in adults (Fig. 66-3).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree