CHAPTER 330 Imaging of Traumatic Brain Injury
TBI lesions can be classified into primary and secondary injuries (Table 330-1). Primary injuries are the direct result of trauma to the head. Secondary injuries arise as a complication of the primary lesions. This classification is important because secondary injuries are potentially preventable, whereas primary injuries, by definition, have already occurred by the time the patient first presents for medical attention. This classification also underscores the fact that TBI is not just a static event, but is rather a progressive injury with varying therapeutic windows. TBI can be further divided according to location (intra-axial or extra-axial) and mechanism (penetrating/open or blunt/closed). Clinical classification of the severity of TBI is currently based on the Glasgow Coma Scale (mild: 13 ≤ GCS ≤ 15; moderate: 9 ≤ GCS ≤ 12; severe: 3 ≤ GCS ≤ 8).1 The goal of neuroimaging is to identify treatable injuries, assist in the prevention of secondary damage, and provide useful prognostic information.
TABLE 330-1 Imaging Classification of TBI
| “Primary” Injury |
| Extra-axial Injury |
| Intra-axial Injury |
| Vascular Injury |
| “Secondary” Injury |
| Acute |
| Chronic |
Imaging Options
Conventional Radiography
Skull fracture, even without clinical signs, is an independent risk marker for a neurosurgically relevant intracranial lesion.2 However, skull films are mainly used for the identification of skull fractures in cases of suspected child abuse, and not for the evaluation of intracranial pathology in children or adults. In fact, conventional radiography is a poor predictor of intracranial pathology and should not be performed to evaluate TBI.3–5 In mild TBI, skull films rarely demonstrate significant findings. In severe TBI, the lack of abnormality on skull films does not exclude major intracranial injury. In one large autopsy series of patients with fatal head injuries, only 75% had skull fractures.6 Negative findings may even mislead medical management. Patients who are at high risk for acute intracranial injury, even without clinical evidence of skull fractures, must be imaged by computed tomography (CT).
Computed Tomography
In the setting of acute head trauma, CT is recommended for patients with moderate and severe TBI (GCS ≤ 12); who are older than 60 years; and have persistent neurologic deficit, headache or vomiting, amnesia, loss of consciousness of 5 minutes or more, depressed skull fracture, penetrating injury, bleeding diathesis, or had anticoagulation therapy.7–13 CT is the modality of choice because it is fast, widely accessible, and highly accurate in the detection of skull fractures and intracranial hemorrhage. Life support and monitoring equipment can be more easily accommodated in the CT scanner suite than in the magnetic resonance imaging (MRI) suite. In addition, CT is superior to MRI in revealing skull fractures and radiopaque foreign bodies. Indeed, MRI is contraindicated in the presence of certain foreign bodies. Noncontrast CT scans can provide rapid and accurate localization of space-occupying hematomas, associated mass effect, and impending complications that would require immediate medical and/or surgical intervention. Intravenous contrast administration should not be performed without a baseline noncontrast examination because the contrast can both mask and mimic underlying hemorrhage.
CT angiography (CTA) uses intravenous iodinated contrast to delineate the vascular structures at high (submillimeter) resolution. CTA is best performed with multidetector CT (MDCT) and rapid bolus contrast injection using a tracking technique. In suspected vascular injury, such as a fracture traversing the carotid canal or venous sinus, CTA can serve as a useful screening method.14
Dynamic perfusion CT is a technique for measuring brain hemodynamics by tracking transient attenuation changes in the blood vessels and brain parenchyma during the first-pass passage of an intravenously injected contrast bolus. Perfusion CT typically involves continuous cine scanning with a scan rate interval of 1 second and a total scanning duration of 40 to 45 seconds.15 Color maps of cerebral blood volume (CBV), mean transit time (MTT), and cerebral blood flow (CBF) can be generated from a voxel-by-voxel analysis of the change in attenuation over time. CT perfusion in patients with severe head trauma has been shown to provide independent prognostic information regarding functional outcome, with normal brain perfusion or hyperemia in patients with favorable outcome, and oligemia in patients with unfavorable outcome.16 One limitation of dynamic perfusion CT is limited anatomic coverage because only a few slices of the brain can be imaged during the 1-second window. Wider coverage can be achieved using a 40-mm wide detector and toggling table technique or a scanner with more multidectors.17,18 Another limitation of CT perfusion is the additional radiation exposure due to cine imaging. Radiation exposure is not an issue with MR perfusion imaging.
Magnetic Resonance Imaging (MRI)
On occasion, MRI may be indicated in patients with acute TBI if the neurologic findings are unexplained by the CT findings (e.g., hemiparesis). However, because of the risks to the patient imposed by the high-strength magnets in modern MRI units and the long scan times, routine MRI during the first week after severe TBI, especially with multiple injuries, is rarely performed. MRI is the preferred imaging modality for subacute and chronic TBI because of its superior sensitivity to both gray and white matter injury. MRI is comparable to CT in the detection of the majority of acute epidural and subdural hematomas.19,20 However, MRI is more sensitive to subtle extra-axial “smear” collections, nonhemorrhagic lesions, and brainstem injuries. It can also be more sensitive to subarachnoid hemorrhage.21,22
Fluid attenuated inversion recovery (FLAIR) imaging increases the conspicuity of focal cortical injuries (e.g., contusions), white matter shearing injuries, and subarachnoid hemorrhage by suppressing the adjacent bright cerebrospinal fluid (CSF) signal typically seen on conventional T2-weighted images. Note, however, that the abnormal high signal in the sulci and cisterns of ventilated patients receiving a high inspired oxygen fraction (>0.60) can be observed in uninjured patients and should not be mistaken for a hemorrhage.23 Sagittal and coronal FLAIR images are particularly helpful in the detection of diffuse axonal injury (DAI) involving the corpus callosum and the fornix, two areas that can be particularly difficult to evaluate on routine axial T2-weighted images.24
Gradient-recalled-echo (GRE) T2*-weighted imaging is highly sensitive to the presence of blood breakdown products, including deoxyhemoglobin, intracellular methemoglobin, ferritin, and hemosiderin. The presence of these molecules alters the local magnetic susceptibility of tissue, resulting in areas of signal loss on GRE T2*-weighted images. Because hemosiderin can persist indefinitely, its detection on GRE T2*-weighted images allows for improved evaluation of remote TBI. Unfortunately, small foci of hemosiderin can sometimes be resorbed; therefore, the lack of hemosiderin on GRE T2*-weighted images does not exclude old hemorrhage.25
Susceptibility-weighted imaging (SWI) can be likened to a supercharged GRE sequence. It amplifies the susceptibility changes among tissues and blood products by combining magnitude and phase information from a high-resolution, full velocity compensated 3-D T2-weighted gradient echo sequence.26 Conventional MRI relies only on the magnitude images and ignores the phase images, the latter of which contain valuable information regarding tissue susceptibility differences. In SWI, phase images are unwrapped and high-pass filtered to highlight phase changes. These are then converted into a mask that is multiplied with the corresponding magnitude images. SWI images are displayed using minimum intensity projection (minIP) reconstruction. The increase in tissue magnetic susceptibility contrast afforded by SWI improves the sensitivity to small TBI hemorrhages, as well as other diseases associated with hemorrhage (e.g., amyloid angiopathy). SWI is 3 to 6 times more sensitive than GRE T2*-weighted imaging in detecting hemorrhagic DAI.27–29
Diffusion-weighted imaging (DWI) measures the random motion of water molecules in brain tissue. Because of its sensitivity to acute shearing injury, DWI has been particularly useful for the detection of DAI.30–33 DWI reveals more DAI lesions than fast spin echo T2-weighted and/or GRE T2*-weighted images in patients imaged within 48 hours of injury. Acute DAI lesions typically show reduced apparent diffusion coefficient (ADC), which measures the magnitude of water diffusion averaged over a three-dimensional space. The fractional anisotropy (FA), which measures the preferential motion of water molecules along the white matter axons, is frequently reduced in chronic DAI. The integrity of the white matter tracts can be further assessed with diffusion tensor imaging (DTI) with 3-D tractography. Unfortunately, the white matter tracts generated with DTI tractography are quite variable and depend on the parameters used to create the tracts. Abnormalities within the tractogram, while visually very appealing, are not specific for TBI. More research is necessary before DTI tractography can be reliably incorporated into the imaging armamentarium.
MR spectroscopy (MRS) measures the relative amount of metabolites in brain tissue. Common neurochemicals that are measured with proton ([1H]) MRS include N-acetylaspartate (NAA), creatinine (Cr), choline (Cho), glutamate, lactate, and myo-inositol. NAA is a cellular amino acid and is a marker of neuronal health. Creatinine is a marker of energy metabolism and cellular density. Creatine is especially abundant in glial cells, and can serve as a marker for posttraumatic gliosis. A reduction in NAA:Cr ratio has been found in patients with history of TBI, and this finding correlated with a worse prognosis.34 An increase in Cho is observed in myelin injury. A reduction of NAA and an elevation of Cho correlate with the severity of injury as measured by the GCS and duration of posttraumatic amnesia.35 MRS can be useful since it can detect abnormalities that are invisible on conventional MRI.35,36
Magnetization transfer imaging (MTI) exploits the longitudinal (T1) relaxation coupling between bound (hydration) protons and free water (bulk) protons. When an off-resonance saturation (radiofrequency) pulse is applied, it selectively saturates protons that are bound in macromolecules. These protons subsequently exchange longitudinal magnetization with free water protons. The magnetization transfer ratio (MTR), a relative measure of the reduction in signal intensity because of the MT effect, provides a quantitative measure of the structural integrity of tissue. A reduction of the MTR correlates with a worse clinical outcome in patients with history of TBI.34
Magnetic Source Imaging (MSI)
Magnetic source imaging (MSI) uses magnetoencephalography (MEG) to localize a weak magnetic signal generated by neuronal electrical activity. Electrical currents flowing within dendrites give rise to a surrounding magnetic field that can be measured by superconducting quantum interfering devices (SQUID). MEG provides a selective reflection of activity in dendrites oriented parallel to the skull surface. MSI integrates anatomic data obtained with conventional MRI and electrophysiologic data obtained with MEG. In two recent studies, MSI showed excessive abnormal low-frequency magnetic activity in mild TBI patients with postconcussive syndromes.37,38 Application of MSI in the evaluation of TBI has been lacking because of the limited availability of MEG, mainly because of cost. Additional research is necessary before MSI is adopted in the clinical setting.
Single Photon Emission Tomography (SPECT)
Single photon emission tomography (SPECT), a nuclear medicine study, uses gamma-emitting isotopes (e.g., [133Xe] and technetium-99-m-hexamethyl-propylamine-oxime [99Tc-HMPAO]) to measure cerebral blood flow (CBF). It can potentially provide a better long-term prognostic predictor in comparison to CT or conventional MRI.39 For example, a worse prognosis has been associated with multiple CBF abnormalities, larger CBF defects, and defects that involve the basal ganglia, temporal and parietal lobes, and brainstem. However, due to its inherent low spatial resolution, SPECT is less sensitive in detecting smaller lesions that are visible on MRI. Therefore, SPECT imaging is complementary to, but not a replacement for, MRI in the evaluation of TBI.
Positron Emission Tomography (PET)
Positron emission tomography, as the name implies, uses positron-emitting isotopes, commonly 15-oxygen (15O) to measure cerebral perfusion and oxygen metabolism, and 2-fluoro-2-deoxy-D-glucose (18F-FDG) to measure cerebral glucose metabolism. PET is relatively expensive and not widely available, serving mainly as a research tool. 15O-PET can define a potential ischemic area after brain injury, which is associated with poor outcome.40–42 Acutely injured brain cells show increased glucose metabolism following severe TBI because of intracellular ionic perturbation.43 Following the initial hyperglycolysis state, injured brain cells show a prolonged period of regional hypometabolism, lasting up to 1 month. Early human studies in TBI have had limited success in demonstrating consistent results regarding regional glucose metabolism. Due to the heterogeneous nature of TBI, studies have found both hypermetabolism and hypometabolism in the same regions across different TBI patients.44 The “metabolic abnormalities” can also be found extending far beyond the lesions, especially with subdural and epidural hematomas.45 The cortical contusion, intracerebral hematoma, and encephalomalacia all tend to show more regional metabolic abnormalities confined to the specific lesions. A more recent study was able to show almost consistent hypermetabolism within the cerebellar vermis of the injured brain.46
Imaging Findings
Scalp and Skull Injury
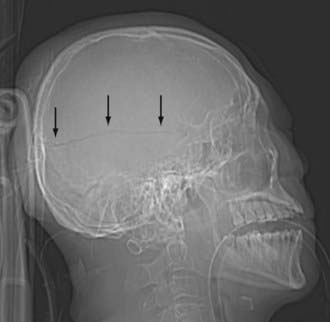
Scalp injury is often the only reliable evidence of head impact, and thus can identify the coup site. When reviewing CT images, first look at the AP and lateral scout images (Fig. 330-1). Sometimes, the scout image can reveal unsuspected upper cervical spine injury or a horizontal skull fracture that will be missed if only the axial CT images are reviewed. When evaluating the CT and MR cross-sectional images, begin by examining the extracranial structures for evidence of scalp trauma (Fig. 330-2; see Figs. 330-6, and 330-15), such as a laceration, subgaleal hematoma, or radiopaque foreign body. The subgaleal hematoma is located beneath the subcutaneous fibrofatty tissue and superficial to the temporalis muscle (see Fig. 330-15).
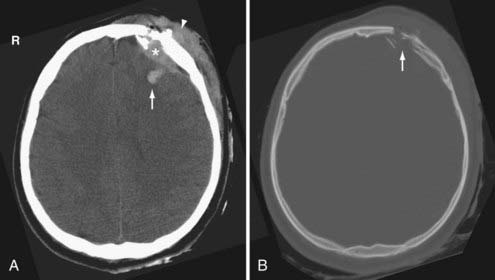
Skull fractures can be classified as linear, depressed, comminuted, compound (open), or diastatic. The linear fracture is the most common type of skull fracture. As mentioned earlier, the nondisplaced linear skull fractures may be difficult to detect, even on CT, if the fracture plane is parallel to the plane of section. Fortunately, isolated linear fractures without associated intracranial pathology are usually clinically insignificant. Depressed and comminuted (i.e., multiple bone fragments) skull fractures are easily detectable on CT (Fig. 330-2). Depressed fractures are often associated with an underlying cortical contusion. When a fracture involves the skull base, communicates with the overlying scalp, or involves the paranasal sinus, it is called a compound fracture. An external compound fracture is typically caused by a penetrating injury and is commonly associated with a depressed, comminuted skull fracture. An internal compound fracture is usually caused by blunt injury and includes those that extend from the mastoid, middle ear, and paranasal sinuses into the cranium. Compound fractures frequently result in pneumocephalus (intracranial air), and they have an increased risk of infectious complications.47 In a compound depressed fracture, the underlying dura is exposed under the fracture site. This is usually a neurosurgical emergency warranting removal of the contaminated bone fragments and dural repair. Diastatic fractures occur from separation of the cranial sutures and are more frequent in children than in adults.
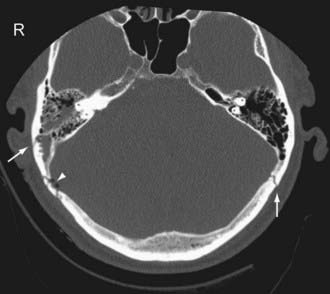
Temporal bone fractures (TBF) are classified either according to their orientation (transverse versus longitudinal) relative to the long axis of the petrous bone (Ulrich’s original classification)48 or based on their involvement of the otic capsule (otic capsule sparing versus otic capsule violating).49,50 The older Ulrich’s classification is still more commonly used, while the newer classification is more predictive of clinical outcome. Longitudinal fractures are oriented parallel to the long axis of the petrous pyramid (Fig. 330-3). They frequently result from direct impact to the side of the head. They represent 70% to 90% of temporal bone fractures.51 Longitudinal fractures can lead to subluxation or fracture of the ossicles, conductive hearing loss, CSF otorhinorrhea, and facial nerve palsy (often partial and delayed). In contrast, transverse fractures are oriented perpendicular to the long axis of the petrous bone (Fig. 330-4). They typically result from impact to the frontal or occipital region. Sensorineural hearing loss, vertigo, nystagmus, perilymphatic fistula, and facial palsy are common complications of transverse fractures. Facial palsy (often complete) occurs in 30% to 50% of transverse fractures.51
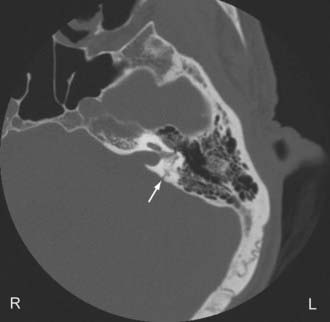
Most temporal bone fractures are oblique and mixed, and the simple classification of fractures as longitudinal or transverse may not be sufficient.52 Otic capsule sparing fractures run anterolateral to the otic capsule, and are usually caused by direct blows to the temporoparietal region. With otic capsule violating fractures, the cochlea and the semicircular canals are damaged. These fractures are the results of direct impacts to the occipital region. Compared with otic sparing fractures, patients with otic capsule violating fractures are 2 to 5 times more likely to develop facial nerve injury, 4 to 8 times more likely to develop CSF leak, and 7 to 25 times more likely to experience hearing loss, as well as more likely to sustain intracranial injuries such as epidural hematoma and subarchnoid hemorrhage.49,50
Primary Extra-Axial Injury
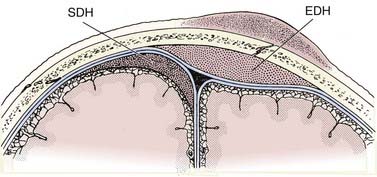
The epidural hematoma (EDH) develops within the potential space located between the dura and the inner table of the skull. The hematoma dissects the dura from the inner table of the skull, forming an ovoid collection that compresses the underlying brain. Since the EDH is subperiosteal, it rarely crosses cranial sutures, where the outer periosteal layer of the dura is firmly attached at sutural margins (Fig. 330-5). At the vertex, however, where the periosteum is not tightly attached to the sagittal suture, the EDH can cross the midline.
EDHs are usually arterial in origin. The vast majority of EDHs occur at the site of impact (i.e., the coup site) and are usually associated with a skull fracture, commonly involving the temporal squamosa region where the fracture disrupts the partially embedded middle meningeal artery.13 In children, EDHs may occur from stretching or tearing of meningeal arteries without an associated fracture. EDHs are less common in children and in the elderly for several reasons. First, the overall incidence of severe head trauma is less in children and the elderly. Second, the skull is more compliant and the meningeal groove is more shallow in children. Third, the dura is more adherent to the inner table of the skull and is therefore not easily displaced in the elderly.
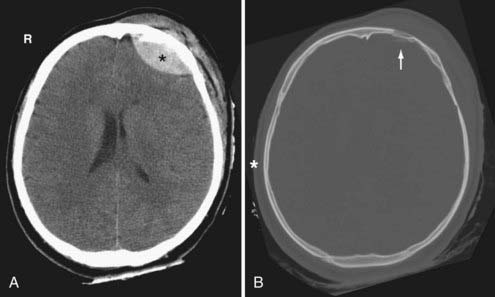
On CT, an acute EDH appears as a well-defined biconvex hyperdense collection within the epidural space that causes focal brain compression (Fig. 330-6). Mass effect, sulcal effacement, and midline shift are frequently seen with large EDHs. The attenuation value of an acute EDH usually ranges between 50 to 70 Hounsfield units (HU).
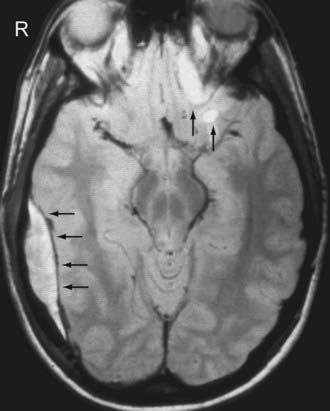
On MRI, a thin dark line is observed at the inner margin of the EDH (Fig. 330-7). This line represents the two layers of displaced dura and confirms the epidural location of the hematoma. Inward displacement of the venous sinuses also serves as a clue that the hematoma is located within the epidural space. As is the case with hematomas elsewhere, the signal characteristics of the EDH are dependent on the age of the blood products.
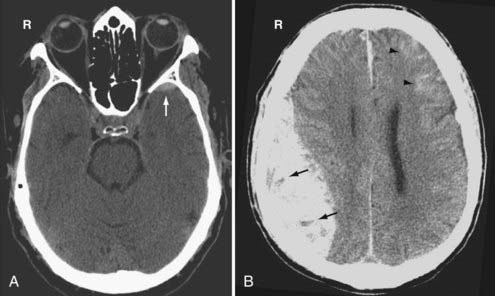
Venous EDHs are less common than arterial EDHs and they tend to occur in three locations: (1) posterior fossa (from rupture of the torcula or transverse sinus), (2) middle cranial fossa (from disruption of the sphenoparietal sinus) (Fig. 330-8A), and (3) vertex (due to injury to the superior sagittal sinus or cortical veins).53 Unlike the arterial EDH, the venous EDH rarely expands beyond its initial size because of the lower pressure imposed by venous extravasation. The venous EDH is less frequently associated with a skull fracture than is the arterial EDH.
Certain imaging characteristics of the EDH are correlated with a worse prognosis and require more aggressive management. One important imaging finding is the presence of low-density areas within the hyperdense hematoma (the so-called swirl sign), thought to represent active bleeding (Fig. 330-8B).54,55 It is an ominous sign that forewarns expansion of an arterial EDH. Patients with this type of EDH tend to present early, with a poorer GCS and a higher mortality rate.56 Contrast extravasation within the low-density areas of the EDH due to active hemorrhage from an underlying dural vessel laceration has also been reported.57 Thus, active extravasation on CT may be another potential biomarker for EDH expansion, and when used in conjunction with other imaging characteristics such as mixed density, it may warrant more aggressive clinical management. Midline shift greater than 1 cm and brainstem distortion are additional imaging findings that often require aggressive management.
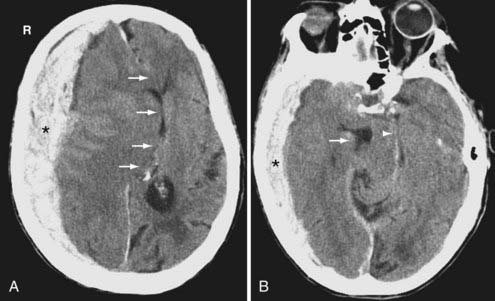
Subdural hematomas (SDH) are located between the arachnoid and the inner meningeal layer of the dura. Because the dura and arachnoid are not firmly attached, the SDH is frequently seen layering along the entire hemispheric convexity from the anterior falx to the posterior falx (see Fig. 330-5). The SDH usually develops from laceration of bridging cortical veins during sudden head deceleration. It can also arise from injury to pial vessels, pacchionian granulations, or penetrating branches of superficial cerebral arteries. Another cause of the SDH is rapid decompression of obstructive hydrocephalus. In this setting, the brain surface recedes from the dura quicker than the brain parenchyma can reexpand after being compressed by the distended ventricles, causing disruption of the bridging cortical veins. The incidence of the SDH is higher in the elderly due to cerebral atrophy because the increase in the extra-axial space allows for increased motion between the brain parenchyma and the calvarium.
On CT, the typical acute SDH is hyperdense, homogeneous, and crescent-shaped (Fig. 330-9). The majority of SDHs are supratentorial and located over the convexity. They are frequently seen along the falx and tentorium (Fig. 330-10). Unlike the EDH, they tend to occur at the contrecoup site. Because the SDH is often associated with parenchymal injury, the degree of mass effect may appear more extensive than the size of the collection.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree