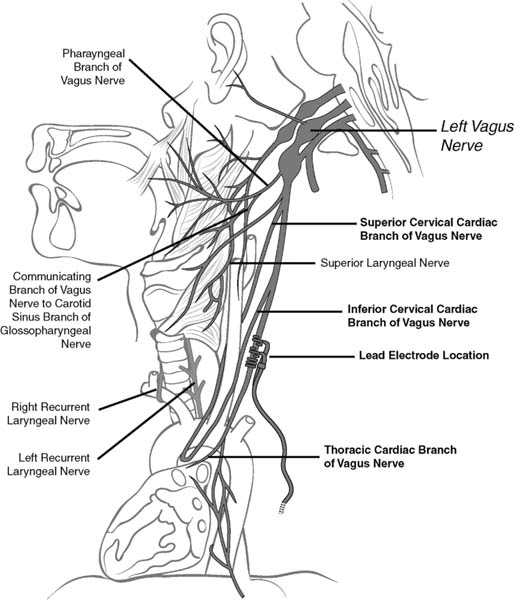
13 Intractable Epilepsy Vagus nerve stimulation (VNS), as delivered by the VNS Therapy System (Cyberonics, Inc., Houston, Texas), is indicated for use as an adjunctive therapy in reducing the frequency of seizures in adults and adolescents over 12 years of age with partial onset seizures that are refractory to antiepileptic medications.1 VNS is not a cure for epilepsy; therefore, patients should consult their physicians before engaging in unsupervised activities.1 It is a good treatment choice for patients who have not experienced success with conventional antiepileptic drugs. VNS does not cause adverse cognitive effects. Furthermore, it can be administered concurrently with antiepileptic drugs, but without additional sedative or behavioral side effects.2,3 Because no formal clinical trials have been conducted in children younger than 12 years when the systems were implanted, the U.S. Food and Drug Administration (FDA) has not approved the use of the device in this age group.4 Although use of VNS for children younger than 12 years is consequently considered “off label,” some physicians do treat patients in this age group with VNS, and such use is according to the individual physician’s judgment. Most often, VNS is prescribed for preadolescent children with partial epilepsy refractory to antiepileptic medications who are not candidates for resective surgery. The safety and efficacy of VNS has not been established for use in patients with arrhythmias, respiratory conditions, preexisting swallowing difficulty, ulcers, vasovagal syncope, only one vagus nerve, other concurrent forms of brain stimulation, preexisting hoarseness, age younger than 12 years, primary generalized seizures, or pregnancy. VNS could affect the operation of a cardiac pacemaker or implanted defibrillator. VNS cannot be used in patients after a bilateral or left cervical vagotomy.1 Special precautions must be taken when a patient undergoes magnetic resonance imaging (MRI). MRI should not be performed with a magnetic resonance body coil in the transmit mode on persons implanted with VNS. The heat induced in the lead by an MRI body scan can cause injury. If an MRI needs to be done, use only a transmit-and-receive type of head coil. Magnetic and radio frequency (RF) fields produced by MRI may change the pulse generator settings or activate the device. The following pulse generator and MRI procedures can be used safely without adverse events:1 The mechanism of anticonvulsant action is not precisely known. It is believed that the VNS disrupts the abnormal pattern of neuronal epileptogenic discharge, increases the threshold of seizures, excites inhibitor pathways, and releases inhibitory neurotransmitters.5 In animal models, VNS prevented seizures or seizure spread in these models: maximum electroshock, pentylenetetrazol (PTZ), 3-mercaptopropionic acid (3-MPA), alumina gel, potassium penicillin, strychnine, and kindling. Localization of vagus-initiated activity in the brain has been observed through animal studies of Fos immunoreactivity, regional brain glucose metabolism, and positron emission tomography (PET) imaging in human patients.1 In a [15O]H2O PET study of 10 patients receiving VNS, increases in blood flow were seen in the rostral medulla, right thalamus, right anterior parietal cortex, bilateral hypothalamus, anterior insula, and inferior cerebellum.6 In the same study, blood flow was decreased bilaterally in the hippocampus, amygdala, and posterior cingulate gyrus. Henry et al6 suggested that VNS brought about acute increases in the synaptic activity of the structures that are directly inner-vated by the central vagal structures, as well as the areas that process left-sided somatosensory information. In addition, VNS seems to alter synaptic activity in the bilateral multiple limbic system structures. The altered synaptic activities at these sites of VNS-induced cerebral blood flow (CBF) changes may indicate areas of therapeutic VNS activity.6 Other studies have shown that VNS can reduce the onset or propagation of seizures. These studies also showed that VNS can cause inhibition of the hippocampus/amygdala, leading to long-term clinical effectiveness.7,8 The electrodes of the VNS device are coiled around the left, rather than the right, vagal nerve because efferent innervations seem to be asymmetrical. The sinoatrial node is innervated by the right vagus nerve, and the left vagus nerve supplies the atrioventricular node. Stimulation of the right vagus nerve therefore has the potential to produce greater cardiac slowing than similar stimulation of the left vagus nerve.9 Preoperative antibiotics are given 30 to 60 minutes before skin incision, depending on antibiotic type. The patient’s neck and chest are prepped and draped in the usual standard sterile fashion (Fig. 13.1). The left carotid sheath is exposed, as it extends along the anterior border of the sternocleidomastoid muscle, and then the left vagus nerve is identified. An exposure of >3 cm of the vagus nerve facilitates placement of the electrodes on the nerve. It is preferable to choose a 3 cm section of nerve halfway up between the clavicle and the mastoid process, where it is clear of nerve branches (Fig. 13.2). Above this area, superior and inferior cervical cardiac branches separate from the vagus nerve. Stimulation of these branches during the system diagnostics (lead test) may cause bradycardia and/or asystole. The nerve usually lies in a posterior groove between the carotid artery and the internal jugular vein.1 A pocket is then created in the left upper chest below the clavicle for the pulse generator. In small children, this preaxillary incision can be made posterior to the lateral border of the pectoralis major to avoid the pulse generator from placing pressure on the incision line.10 A subpectoral implantation may allow better coverage with soft tissue, especially in children with minimal subcutaneous fat, and it may reduce the likelihood of the skin breaking down and damage to the generator.11 The lead is tunneled subcutaneously from the neck to the pulse generator pocket in the chest, and the two electrodes (three helixes) are wrapped around the left vagus nerve. A strain relief bend and loop are then formed (i.e., the lead body is curled around and secured to the fascia with Silastic tie-downs to allow for some movement), and the lead is connected to the pulse generator. System diagnostics (i.e., testing of lead) is then performed. We then place the pulse generator in the chest pocket, with the extra coiled lead next to the pulse generator, and repeat the system diagnostics. Both incisions are closed in the standard fashion.1Figure 13.3 shows the hardware that is implanted. During the 7 to 14 days after implantation, the patient should be seen to confirm wound healing and proper operation of the pulse generator. The pulse generator’s output current for both the magnet and the programmed stimulation should be set at 0.0 mA for the first 14 days after implantation to allow for healing. Because VNS is an adjunctive therapy to existing antiepileptic medications, we generally keep all antiepileptic medications stable for the first 3 months of stimulation before we attempt to reduce or change a patient’s medication. During initial programming, the output current should be programmed to start at nominal parameters (0.0 mA) and then be slowly increased in 0.25 mA increments until the patient feels the stimulation at a comfortable level. Patients who are receiving replacement generators should also be started at nominal parameters, with 0.25 mA-step increases to allow reaccommodation. The average output current used during the clinical studies was ~1.0 mA. Other standard treatment settings were 30 Hz, 500 μsec pulse width, 30 second on time, and 5 minute off time. There is no proven correlation between high output current (mA) and device effectiveness, nor is there a standard treatment level that should be achieved during treatment ramping. Excessive stimulation at an excessive duty cycle has resulted in degenerative nerve damage in laboratory animals.1 Fig. 13.1 The skin incisions are marked. (From Cyberonics, Inc., Houston, Texas. Reprinted with permission.) Several studies have featured VNS in the pediatric population. Many have reported benefit in children with a variety of epilepsy syndromes.12–15 Results from several small observational studies suggest that the therapeutic effect of VNS is better in children than in adults, and the benefit in children is achieved more rapidly.3,9,12,16–26 Parents and care-givers reported an increase in alertness, memory, motor, verbal, and cognitive function.19,20,27,28 Fig. 13.2 Branches of the vagus nerve. The three helixes should be placed around the vagus nerve below the level of the inferior cervical cardiac branch. (From Cyberonics, Inc., Houston, Texas. Reprinted with permission.) Three VNS studies did not note any negative effects on cognition in adults or in children.29–31 Studies have shown that VNS is effective in preadolescent patients.12,15,25,27,28,32,33 In one study, response was more favorable in the younger group compared with children older than 12 years.4 Studies have suggested that children with highly medication-resistant seizures, such as children with Lennox-Gastaut syndrome (LGS), may derive significant benefit from VNS.3,18,21,22,28 A study of 16 children with LGS showed an overall seizure reduction of 26.9% following VNS.21 With LGS, some studies indicate that VNS can produce satisfactory seizure control, whereas other studies have failed to demonstrate significant efficacy.3,18,20,34 In the study by Rychlicki et al, the clinical effectiveness of the VNS seemed greater in the group of patients with partial epilepsy and drop attacks than in the group of patients with LGS.34 In a series of patients with autism and Landau-Kleffner syndrome, improvements in quality of life were seen in up to 76% of pediatric patients at 12 months of follow-up.35 In a series of children with epileptic encephalopathies, a >50% decrease in seizure frequency was seen in 72% of patients at 12 months and in 50% of patients at 18 months.36 Fig. 13.3 Hardware showing pulse generator and lead. (From Cyberonics, Inc., Houston, Texas. Reprinted with permission.) We present highlights of several studies’ findings here, in chronological order: 1. Study E04, an open-label safety study done during the preapproval period, included patients 12 years old and younger. Sixteen patients under age 12, ranging from 3.6 to 12.0 years old, were evaluated. These patients experienced a 17.9% median reduction in seizures, and 31% of the patients experienced a >50% seizure reduction.1 2. Murphy et al reported a reduction of >90% in the frequency of seizures in 3 out of 12 children with intractable epilepsy.27 In a study of 19 children, 6 (32%) had a >90% reduction in the number of seizures, and 10 other patients (52.6%) had a >50% seizure reduction. This study had a follow-up period ranging from 2 to 30 months.3 Murphy et al described the results of at least 3 months of VNS treatment in 60 patients (<18 years of age) enrolled in three different multicenter trails. A median reduction in seizure frequency of 23% at 3 months, 31% at 6 months, 34% at 12 months, and 42% at 18 months was observed. This study included 16 children younger than 12 years of age, enrolled in a compassionate use uncontrolled preapproval trial. These younger children appeared to derive a benefit similar to that of the entire group.12 3. Patwardhan et al’s 2000 retrospective study of 38 consecutive pediatric patients, at a median follow-up period of 12 months, seizure reduction >50% was achieved in 26 of 38 patients (68%). There was no difference in the response observed in the younger (<12 years at implantation; n = 28) as compared with the older pediatric patients. The quality of life was improved in the majority of patients in this study.25 4. Farooqui et al reported six patients with ages ranging from 7 to 18 years, one of whom underwent VNS explantation because of a self-inflicted wound that was secondary to persistent trauma at the site of implantation. The remaining five patients had an average of 73 seizures per month. After an average follow-up time of 6.5 months with VNS, these patients had an average of 14 seizures per month, a 78% reduction.37 5. Helmers et al published a six-center retrospective series in 125 patients younger than 18 years of age who had been treated with VNS for 3 months to 1 year. Average seizure reduction was 36.1% at 3 months and 44.7% at 6 months. The authors did not observe any difference in seizure reduction among the group of 41 patients younger than 12 years at the time of VNS implantation.28 Zamponi et al reported a seizure reduction rate in children >50% in 10 of 13 patients.23 6. Murphy et al examined outcome in the first 100 patients implanted at a single pediatric epilepsy center. Twelve patients were older than 18 years at the time of implantation. At a mean follow-up of 2.7 years, seizure reduction >50% was achieved in 45% of patients. At their last follow-up, 18% of the patients had been seizure free for 6 months. The percentage of seizure-frequency reduction in younger (6–11 years; n = 50) and older (12–18 years at the time of VNS implantation; n = 34) patients was similar in the two groups.38 7. In a study of 13 patients ranging in age from 6 to 28 years, Buoni et al reported 38.4% of patients obtained a >50% reduction in seizure frequency.26 In the Hallbook et al study, 6 of 15 children showed a 50% or more reduction in seizure frequency, 12 showed an improvement in quality of life, and 11 improved in seizure severity and mood. Behavior, mood, and depressive parameters tended to improve over time.39 8. Blount et al studied VNS in six patients younger than 5 years, of whom 83% had a significant decrease in the frequency of their seizures. Two became seizure free (33%), three improved (50%), and one (17%) had no change in seizure status. Age at implantation did not seem to correlate with patient success.32 9. Alexopoulos et al compared the efficacy in children younger than 12 years at the time of VNS implantation versus adolescent children older than 12 years. They studied 46 children (ages 2.3–17.9 years); 21 patients (45.6%) were younger than 12 years at the time of surgery. Median seizure-frequency reduction was 56% at 3 months, 50% at 6 months, 63% at 12 months, 83% at 24 months, and 74% at 36 months. A total of 24 patients experienced a seizure-frequency reduction of >75%, and 19 patients experienced no response (increase in seizures or <50% reduction). Finally, five patients (10.1%) experienced no seizures for >6 months at the time of their last follow-up. Response was more favorable in the younger group.4 10. In the Rychlicki et al study of pediatric patients, ~55% of patients showed a reduction in seizure frequency of at least 50%. Efficacy progressively improved with the duration of treatment up to 24 months postoperatively. Among study patients with partial epilepsy, a small number experienced long-term seizure freedom. The authors noted that during seizure freedom, patients may function better during daily activities and achieve improvements in quality of life and neuropsychological performances.34 11. Large studies of children have described multicenter28 and company database series.13 Most single-center studies range from 1 to 28 patients.12,20,25,26,40 Many studies have not separated younger (<12 years) from older (>12 years) children in their analysis. 12. Saneto et al studied 43 children younger than 12 years. Overall median seizure reduction was 55%, and 37% had at least 90% reduction. VNS was effective in children with generalized, mixed, and partial medically refractory seizures, indicating that VNS may be effective in multiple seizure types and across age ranges.15 Generators eventually require surgical replacement due to battery depletion. Projected battery life decreases as lead impedance increases. Battery life also depends on the pulse generator model, parameter settings, and the frequency of magnet use. In our experience, a battery can last ~5 years. Generator replacement does not, of itself, require lead replacement unless a lead fracture is suspected. Fractures can happen but are infrequent and can often be revealed when performing diagnostic testing. A lead’s lifetime is undetermined at this time, but it can easily outlast adolescent growth spurts and several battery lives. Events that can shorten the life expectancy of the lead are blunt trauma to an area of the body beneath which the lead is implanted and twisting or picking at the lead or generator. Improper surgical implantation of the VNS device, such as inadequate strain relief, suturing directly on the lead or to muscle, and not using tie-downs, can also decrease life expectancy of the lead.1 Hoarseness, the most common complication in all patients, can be caused by device malfunction, nerve fatigue, or vocal cord dysfunction due to trauma at the implantation site. Adverse events reported also include voice alteration (hoarseness), increased coughing, pharyngitis, paresthesias, dyspnea, dyspepsia, nausea and vomiting, laryngismus, ataxia, hypesthesia, infection, and insomnia.1 It has been reported that 54% of pediatric patients have hoarseness, 14% have cough, and 9% have dysphagia.25 During the clinical trials (N = 454 persons ages 12 years and older except for E04, which was a compassionate use trial and included some children), 1.1% of the patients developed infections that required explantation of the device, and 1.8% developed infections that did not require explantation.41 Wound infection has been reported as <3% by DeGiorgio et al.42 The risk of infection in pediatric patients may be higher. Studies of VNS in children10,25,43,44 report infection rates from 2.9 to 12.5%. In the study by Kirse et al, infections seemed to occur more commonly with the axillary incision compared with the cervical incision.10 Kirse et al also found the most common surgical complication leading to generator explantation is skin breakdown and infection at the implantation sites.10 The pediatric population poses particular challenges with regard to this complication because they may tamper with the wound or may experience trauma to the wound or generator.43,45 Manipulation of the pulse generator or lead through the skin may damage or disconnect the lead from the pulse generator and/or possibly cause damage to the vagus nerve.1One developmentally disabled child in the study by Smyth et al caused a lead fracture by rotation of the device in the subcutaneous pocket.43 Vassilyadi and Strawsburg suggested that infections requiring explantation may be more common in younger children, especially those with mental retardation.46 Although device implantation in children can sometimes be risky, VNS is usually not associated with adverse effects that can limit the use of antiepileptic drugs. Adverse effects can include dizziness, ataxia, insomnia, cognitive impairment, and weight gain.4 The VNS discontinuation rate in one study4 was 21.7% (10/46 patients) and primarily reflected the absence of clinical response after 12 months or more of intermittent VNS. Five patients developed a wound infection around the generator, which necessitated device removal in four.4
Vagus Nerve Stimulation
Contraindications and Precautions
 Pulse generator output programmed to 0 mA for the MRI procedure
Pulse generator output programmed to 0 mA for the MRI procedure
 Head coil type: transmit-and-receive only
Head coil type: transmit-and-receive only
 Static magnetic field strength: ≤2.0 tesla
Static magnetic field strength: ≤2.0 tesla
 Specific absorption rate (SAR): <1.3 W/kg
Specific absorption rate (SAR): <1.3 W/kg
 Time-varying intensity: <10 tesla/sec
Time-varying intensity: <10 tesla/sec
Pathophysiology
Operative Procedure
Patient Follow-up
Results
Pulse Generator and Lead Replacement
Complications
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree