13
Jugular Bulb Tumors
The jugular foramen at the base of the skull is an anatomically complex region. It is divided into two separate compartments. The posterior lateral compartment is the pars vascularis, which contains the sigmoid sinus, jugular vein, and the posterior meningeal artery. The anterior medial compartment is the pars nervosa, through which the glossopharyngeal, vagal, and accessory cranial nerves, and the inferior petrosal sinus course (Fig. 13.1). Successful treatment of tumors involving the jugular foramen mandates a clear understanding of the anatomy of the jugular foramen and the natural history of the tumors involved.
Since the first reported exploration of the jugular bulb by Seiffert in 1934,1 surgeons have been striving for the optimal approach and technique to facilitate complete extirpation of tumors of the jugular foramen. Earlier treatments focused on limited resection combined with radiation therapy or radiation therapy alone. The relative inaccessibility of the jugular bulb region, the potential for harming the surrounding neurovascular structures, and the extremely vascular nature of these tumors dictated treatment that was, by and large, palliative. As experience was gained, innovative surgical procedures were developed, most notably the combined lateral skull base approaches and the infratemporal approach. In addition to the advancements in surgical approaches, the implementation of adjunctive therapies, such as superselective embolization, gamma knife radiosurgery, and the aggressive treatment of postoperative neurologic deficits, has resulted in dramatic improvements in long-term outcomes.
♦ Historical Overview
The development of surgical techniques aimed at the removal of jugular bulb tumors is intimately tied to the history of the surgical management of glomus jugulare tumors. The seminal work on the glomus jugularis, or jugular body, was reported in 1941 by Guild2 at the American Association of Anatomists meeting in Chicago. He described nests of “blood vessels of capillary or of pre-capillary caliber with numerous epithelioid cells between the vessels” located near or in the wall of the jugular bulb. During the 1950s, several authors communicated their efforts in treating glomus jugulare tumors with mostly discouraging results.3,4 The intricate anatomy of the jugular bulb region and the risk of hemorrhage from the tumor, in combination with the lack of high-definition imaging studies to elucidate tumor margins, were significantly limiting.
In the early 1960s, the introduction of imaging techniques, with advances in diagnostic technology, enabled one to detail the surgical anatomy of the jugular foramen affected by neoplasia. A significant reduction in the morbidity and mortality with improved surgical accuracy resulted.5 Arteriography,6 polytomography,7 and retrograde venography8 allowed the surgeon to “view” the tumor and select the most judicious approach for treatment. Shapiro and Neues9 in 1964 reported their experience with a patient with a recurrent glomus jugulare tumor. They accomplished complete removal of the tumor involving the jugular bulb region with translocation of the facial nerve. Unlike earlier reports, there was minimal blood loss, and neurologically the patient did well. Gejrot10 described a similar procedure in 1965 in a series of four patients. These reports were important in that they established the foundation for current surgical techniques. They proved that tumors in the jugular bulb area could be removed with preservation of neural function. One critical contribution in Gejrot’s approach, which persists as a crucial step of modern surgical treatment of jugular bulb tumors, was that he stressed the importance of maintaining the medial wall of the sigmoid sinus at the level of the jugular bulb to protect the lower cranial nerves.
Further interest and refinement of surgical technique continued through the ensuing years. Gardner et al11 in 1977 detailed an operative technique with a combined lateral skull base approach utilizing a multidisciplinary team. The approach consisted of three phases. Exposure of the skull base through the neck was performed first, followed by bone removal within the temporal bone and the jugular fossa. Lastly, tumor removal was accomplished with wound reconstruction to follow. In 1977, Fisch12 introduced the infratemporal approach to gain complete access to the internal carotid artery within the temporal bone, which was a major limitation of the earlier approaches. The exposure and control of the carotid artery allowed for larger glomus jugulare tumors to be treated with increased safety. Furthermore, Fisch13 added a classification scheme for glomus tumors of the temporal bone, which he revised in 198114 (Table 13.1).
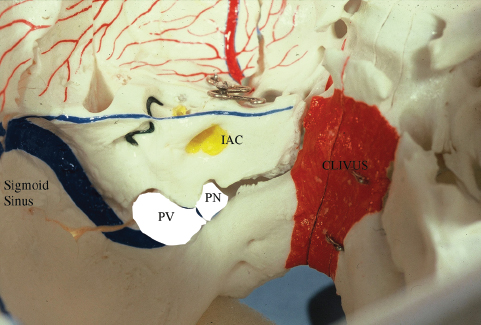
Fig. 13.1 A view of the left jugular foramen region reveals the two compartments: the pars vascularis (PV) and pars nervosa (PN). Note the anterior medial location of the pars nervosa. IAC, internal auditory canal.
A major advancement in the surgical treatment of glomus jugulare tumors occurred with the development of preoperative superselective embolization of the external carotid arterial supply of these highly vascular tumors. First introduced by Hilal and Michelsen15 and Brismar and Cronqvist,16 Simpson et al17 reported in 1979 the use of preoperative embolization in glomus jugulare tumors in an effort to reduce operative blood loss. Murphy and Brackmann18 substantiated the use of preoperative embolization in a 1989 report analyzing 35 patients. They concluded that there was a significant reduction in the operative blood loss and operating time, and embolization led to a higher rate of complete resection of tumors. However, there did not appear to be a reduction in the risk of injury to the lower cranial nerves. As for the morbidity of the embolization procedure, the current state of technology and expertise in interventional radiology has significantly reduced the incidence of stroke and cranial nerve injury experienced during the earlier years of application.
Table 13.1 Fisch Classification of Glomus Tumors of the Temporal Region
| Type A | Tumors limited to the middle ear space |
| Type B | Tumors limited to the middle ear or mastoid without involvement of the infralabyrinthine space of the temporal bone |
| Type C | Tumors involving the infralabyrinthine and apical spaces of the temporal bone, with extension into the apex |
| Type D1 | Tumors with intracranial extension less than 2 cm in diameter |
| Type D2 | Tumors with intracranial extension greater than 2 cm in diameter |
♦ Tumor Types
The most common tumors involving the jugular foramen are paragangliomas, schwannomas, and meningiomas. Although appearing histologically benign, glomus jugulare tumors are characterized by unpredictable biologic behavior. Typically arising from glomus bodies positioned in the dome of the jugular bulb, the neoplasm is intimately associated with critical neurovascular structures at the skull base.
Schwannomas of the jugular foramen arise from cranial nerves IX, X, and XI. These nerves exit the brainstem as a series of rootlets from the retro-olivary sulcus and then traverse to the jugular foramen in an inferolateral direction. Symptoms depend on the cranial nerve involved, the site of origin, and the extent of spread. Jugular foramen schwannomas can be classified according to their growth patterns as described by Kaye et al19 (Table 13.2).
Meningiomas are the third most common tumor of the jugular foramen. Arising from the arachnoid cap cells, they typically originate from the dura overlying the posterior aspect of the petrous bone. Growth often occurs intracranially with subsequent involvement of the jugular foramen. Less commonly, they may originate within the jugular foramen itself. Intracranial and extracranial spread is possible.20
♦ Patient Selection
Achieving an optimal treatment outcome is directly related to the proper choice of a management strategy unique for each patient. A multidisciplinary team utilizes a comprehensive and selective approach in defining the appropriate treatment regimen for a particular lesion. Risk factors considered include the patient’s age and medical condition, the extent of tumor, and the presence of neurologic deficits.
Symptomatic elderly patients or those with significant medical conditions are best treated with palliative radiotherapy, despite the lack of conclusive data establishing its efficacy. In extensive tumors in which it is felt that significant postoperative morbidity will occur, subtotal resection with or without postoperative radiation may be the treatment of choice. The aggressiveness of surgical management has advanced with selective embolization of large vascular tumors and is heavily dependent on the experience and skill of the surgeon. In selected patients with static cranial nerve deficits and no evidence of brainstem compression, observation alone may be warranted.
Table 13.2 Growth Patterns of Jugular Foramen Schwannomas
| Type A | Tumors that are primarily intracranial with only a small extension into bone |
| Type B | Tumors in which the main mass is within bone, with or without an intracranial component |
| Type C | Tumors that are primarily extracranial with only a minor extension into bone or into the posterior fossa |
♦ Preoperative Management
The surgical removal of tumors of the jugular foramen requires a complete and thorough preoperative evaluation involving a multidisciplinary team. Careful attention is given to the medical condition of the patient, tumor size, involvement of the surrounding neurovascular structures, and vascular supply of the tumor. In assessing glomus tumors, one must determine the presence or absence of catecholamine secretion and the presence of multiple tumors.
Plain skull x-rays are of limited benefit, but they may show enlargement of the jugular foramen. A computed tomography scan with thin cuts defines the extent of temporal bone involvement by the tumor and the altered anatomy of the jugular foramen. Glomus jugulare tumors typically result in permeative, erosive margins (Fig. 13.2). Schwannomas of the jugular foramen produce smooth, bony margins through an expansile process (Fig. 13.3). Meningiomas may be calcified, and there can be associated hyperostosis of the bone (Fig. 13.4). Magnetic resonance imaging with and without contrast reveals excellent definition of the soft tissue component of the tumor at the level of the skull base and extracranial space (Fig. 13.5).
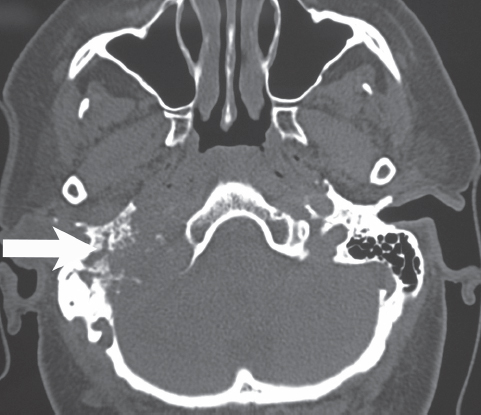
Fig. 13.2 Axial computed tomography scan without contrast reveals a destructive lesion of the left jugular foramen region consistent with a glomus jugulare tumor. The jugular foramen region has been significantly expanded (arrow).
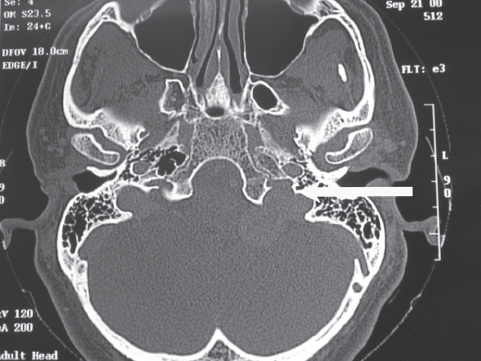
Fig. 13.3 Axial computed tomography scan without contrast reveals an expansile lesion of the left jugular foramen. Note the blunting of the jugular spine (arrow). The patient was found to have a glossopharyngeal schwannoma at the time of surgery.
Four-vessel cerebral angiography should be performed in all cases suspicious for glomus jugulare tumors as a definitive diagnostic study to facilitate proper treatment planning. One often finds rich arterial contributions to the tumor, mostly from the ascending pharyngeal and caroticotympanic arteries (Fig. 13.6). In large tumors, a significant arterial contribution may be found from the internal carotid artery vessel wall when the artery is encased by tumor growth. An examination of the late venous phase is mandatory to determine the patency of the sigmoid-jugular venous system and to identify unusual collateral venous outflow that has developed in response to the tumor. The contralateral jugular vein should be visualized, as it may be congenitally small or absent and blocked in cases of bilateral glomus jugulare tumors. The sigmoid-jugular venous system may be obstructed at any level from compression or intraluminal invasion by tumor. Evaluating the intraluminal extent of the tumor aids in determining the proper level for ligation of the sigmoid sinus and jugular vein to prevent venous tumor embolization when surgically removing a glomus jugulare tumor.

Fig. 13.4 Coronal computed tomography scan without contrast shows prominent calcifications (arrow) seen within this jugular foramen meningioma.
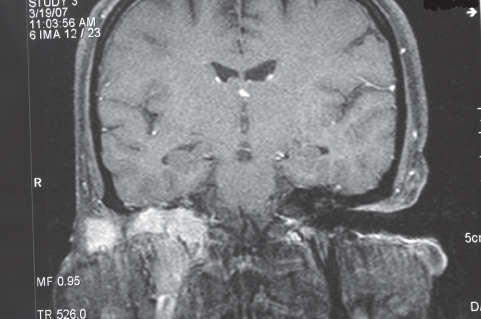
Fig. 13.5 Spread of this glomus jugulare tumor into the high cervical area and through the external auditory canal is well delineated on this coronal magnetic resonance imaging (MRI) with contrast.
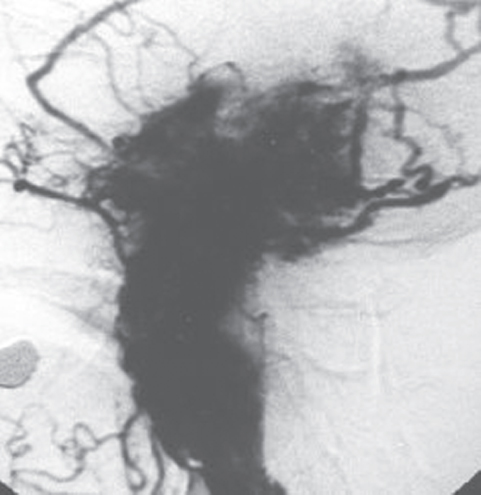
Fig. 13.6 An intense vascular blush reveals the rich arterial supply to the tumor after carotid artery injection.
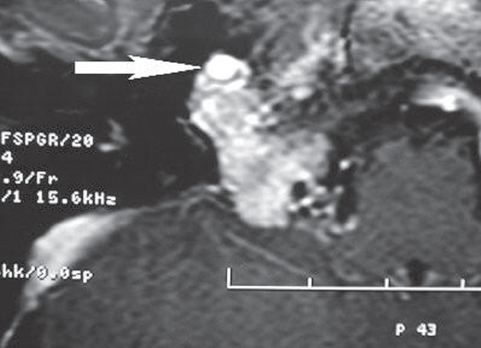
Fig. 13.7 Axial MRI with contrast shows an intensely enhancing, lobulated mass in the right jugular foramen with extraforaminal extension. The carotid artery is seen in the anterior portion of the mass (arrow).
Patients with tumors that encase or displace the carotid artery may be evaluated for the possibility of arterial sacrifice or bypass (Fig. 13.7). An assessment of the collateral circulation during the arterial phase of the angiogram gives one initial insight into whether or not the patient would tolerate permanent occlusion of the internal carotid artery. The gold standard for determination is temporary balloon occlusion, which may be performed during the angiographic study. Temporary occlusion of the internal carotid artery identifies the majority of individuals tolerant of carotid artery sacrifice intraoperatively. However, there is no single test or combination of tests that can absolutely ensure the absence of a cerebrovascular accident following occlusion of an internal carotid artery.21 If the patient fails the test occlusion, an elective arterial bypass prior to tumor removal would be recommended.
The effectiveness of radiosurgery during the past decade for treatment of the majority of the common tumors that occur in the jugular foramen region has resulted in the rare sacrifice of the internal carotid artery to completely remove a benign, slow-growing tumor of the jugular foramen region. In the majority of skull base centers that manage tumors of the jugular foramen that encase the internal carotid artery, surgical subtotal removal of the tumor is the preferred approach followed by radiosurgical treatment of residual tumor involving the internal carotid artery.
A major advancement in the surgical treatment of glomus tumors over the past two decades has been the development and application of preoperative superselective embolization. Performed by the interventional radiologists 24 to 48 hours prior to surgery, embolic materials or coils are used to selectively occlude the arterial feeders of these complex vascular tumors. The current state of technology and expertise in interventional radiology has significantly reduced the incidence of stroke and cranial nerve injury experienced during the early years of application.
In a minority of cases, glomus jugulare tumors secrete catecholamines. An excess of catecholamines can elevate the blood pressure. Manipulation of the tumor during surgery may result in significant intraoperative hypertension that may be uncontrollable. A preoperative 24-hour urine specimen is performed to detect the presence of vanillylmandelic acid, metanephrines, and free catecholamines. If present, a pharmacologic blockade is performed with the use of an alpha-blocker, and this is usually initiated 2 weeks before the operation. A beta-blocker is given approximately 24 hours before surgery to avoid tachycardia.
Other testing completed during the preoperative period includes routine audiometry to establish the function of the hearing apparatus. Routine laboratory studies should include a peripheral blood count and coagulation panel because of the highly vascular nature of these tumors with anticipated blood loss.
♦ Operative Treatment
Anesthesia and Monitoring
The goal of general anesthesia for glomus jugulare tumors is to provide a quiet, surgical field and ample working space for multiple surgical teams. One must be prepared for a lengthy operation involving multiple stages that may involve considerable blood loss and cranial nerve dysfunction. A thorough discussion of the anesthesia plan, to include prolonged intubation and invasive monitoring, should precede surgery.
A smooth induction with a one-time dose of nondepolarizing muscle relaxant facilitates intubation with a reinforced endotracheal tube. After the airway is secured, a Dobbhoff tube (Covidien, Norwalk, CT) is placed. Continuous systemic monitoring is essential and includes placement of an arterial line (in the contralateral radial artery), a central intravenous (IV) line (placed in the femoral vein), a Foley catheter, and a rectal temperature probe. Central IV line insertion into the jugular vein of the neck is avoided because of the unilateral obstruction of the sigmoid-jugular venous system related to the tumor process. Facial nerve monitoring is performed routinely. Glossopharyngeal and vagal nerves are monitored in selective cases only if the size of the tumor suggests a potential for injury. Blood gases and electrolytes are evaluated periodically, as well as blood sugars when indicated. Maintenance of anesthesia is accomplished with a continuous infusion of narcotic (Sufenta) plus a lowered concentration of inhalational agent.
Emergence from anesthesia should include rigid blood pressure control. Strict maintenance of blood pressure control should occur in the first 12 hours in the postoperative period. We commonly use a continuous infusion of the calcium channel blocker Cardene for its ease of titration and short-acting effects.
In tumors involving the jugular fossa, in the absence of preexisting lower cranial nerve deficit, conservative airway management immediately postoperatively with continued intubation during the initial 12 to 24 hours is encouraged. In patients with long-standing cranial nerve deficits, with a surgery ending in less than 8 hours and no comorbidity considerations, a more aggressive approach to extubation may be considered. Patients with brainstem compression should be managed conservatively with regard to extubation.
If uncertainty of lower cranial nerve injury exists, direct fiber-optic visualization of the vocal cords at the time of extubation should be performed. The vocal cords may be swollen and edematous from extended intubation related to a surgical procedure exceeding 8 hours. In such cases, a vocal cord paralysis may not be obvious clinically, and the patient will initially appear to ventilate adequately. Within a 12- to 24-hour period following extubation, aspiration becomes apparent and the patient’s pulmonary status deteriorates.
Positioning (Fig. 13.8)
Positioning of the patient has two main goals: optimal surgical access by multiple surgical teams, and facilitation of venous drainage. We accomplish these goals by placing the patient supine in a flexed position, head up, with the legs slightly bent at the knees. The head is not placed in a fixed headrest; instead, the head and neck are available for turning as the surgeon desires. The arms are tucked at the patient’s side after placing each arm in slight flexion at the elbow with the hands open and flexed. Straps are padded with cushioning and placed across the patient’s chest, hips, and legs to allow for full tilt of the table in any direction.
Operative Approaches
The operative approach is dictated by the location and size of the tumor mass. The size of the tumor and the need for control of the carotid artery mandates individualization of the surgical technique. Three techniques will be described here: the combined lateral skull base approach, the modified lateral skull base approach, and the preauricular infratemporal fossa approach. Modifications of these approaches may be used for tumors with intracranial extension. These include retrosigmoid, presigmoid, retrolabyrinthine, translabyrinthine, and transcochlear approaches.
Operative Procedures
Combined Lateral Skull Base Approach
♦ High Cervical Exposure
This approach is the procedure of choice for small or medium-size tumors that extend up to the level of the petrous internal carotid artery. The first portion of this procedure consists of a postauricular C-shaped incision that begins approximately 3 cm above the pinna and proceeds posteriorly 3 cm behind the helix of the ear. This incision is continued inferiorly onto the neck along the anterior border of the sternocleidomastoid muscle. The greater occipital nerve is often exposed in the operative field and may be harvested to be used later in the operative procedure as a nerve graft. The skin flap, in addition to the musculoperiosteal flap covering the suboccipital bone, is then rotated anteriorly. The external auditory canal is transected at the bony-cartilaginous junction, and the musculoperiosteal flap is used to close the meatus and create a blind pouch. After identification of the sternocleidomastoid muscle, dissection proceeds anteriorly. The facial vein is ligated and transected, and the common carotid artery, internal and external carotid arteries, internal jugular vein, cranial nerves X and XII, and the ansa cervicalis are isolated. These structures are followed superiorly, where cranial nerve XI is identified (Fig. 13.9). Transection of the sternocleidomastoid muscle from the mastoid tip and the posterior belly of the digastric muscle from the digastric groove are then completed. Elevating the tail of the parotid gland, the main trunk of the facial nerve can be identified at the stylomastoid foramen. Removal of the styloid process follows after transection of the stylopharyngeal, styloglossus, and stylohyoid muscles. The base of the styloid is preserved for anatomic orientation, and direct access to the high cervical carotid artery and jugular foramen region is now possible. Removal of the styloid process allows visualization of the high cervical internal carotid artery in the carotid space immediately below the skull base. Cranial nerve IX will be seen crossing over the anterior surface of the internal carotid artery at this level.
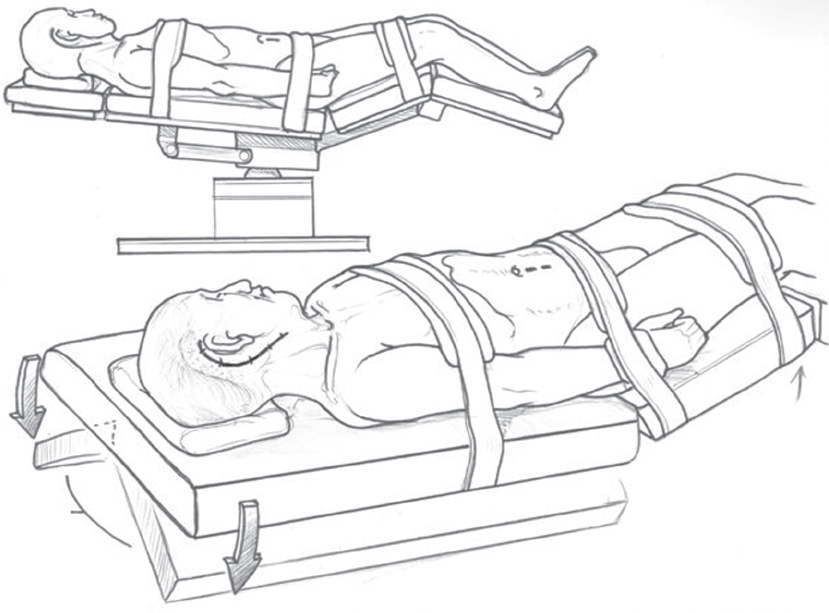
Fig. 13.8 Positioning of the patient is shown. The head is not placed in a fixed headrest so that the surgeon is able to turn the head and neck as desired. Additional rotation in either direction can be accomplished by turning the bed.
Palpation of the transverse process of the first cervical vertebra, the base of the styloid process, and the mastoid tip provides constant bony anatomic landmarks for orientation with respect to the jugular foramen. The jugular vein is found resting on the rectus capitis lateralis muscle, which extends from the transverse process of C1 to the posterior bony margin of the jugular foramen. The transverse process of the first cervical vertebra may be removed for exposure; however, care should be taken to identify and protect the vertebral artery. Cranial nerve XI is usually identified as it crosses over the anterior surface of the jugular vein at or above the level of the transverse process of C1 to enter the posterior margin of the sternocleidomastoid muscle. Soft tissue surrounding the jugular foramen may be removed, allowing a 270-degree control of the jugular vein (Fig. 13.10). The medial margin of the jugular foramen is not violated in the dissection to protect the lower cranial nerves as they exit the skull base.
♦ Temporal Bone Exposure
The initial step involves a simple mastoidectomy. The bone of the external ear canal is removed followed by disarticulation of the incostapedial joint. This facilitates removal of the tympanic membrane, malleus, and incus. Care is taken to protect the stapes. If tumor is present in the middle ear, it is mobilized inferiorly. One must take caution to remain below the labyrinth to avoid loss of hearing. In patients with non-serviceable hearing, removal of the cochlea and labyrinth may be necessary for tumor exposure. Skeletonization of the mastoid and tympanic segments of the facial nerve ensues, and the nerve is traced superiorly to the geniculate ganglion. Fibrous attachments along the course of the nerve are then cut sharply. Depending on the size and extent of the tumor, the greater superficial petrosal nerve and the chorda tympani may be sectioned, and the facial nerve mobilized anteriorly. Drilling of the anterior epitympanum to the soft tissue depth forms a tangential notch where the facial nerve may be placed. The proximal nerve lies within this groove and the distal portion within the parotid gland. Preservation of soft tissue at the stylomastoid foramen is critical to preserving the arterial blood supply from the stylomastoid artery to the facial nerve. Care is taken for the remainder of the procedure to protect the facial nerve for manipulation. If it is found that the nerve is directly involved with tumor during the exposure (Fig. 13.11), the diseased portion may be resected, and an interposition nerve graft (greater occipital nerve) may be used as a cable graft.
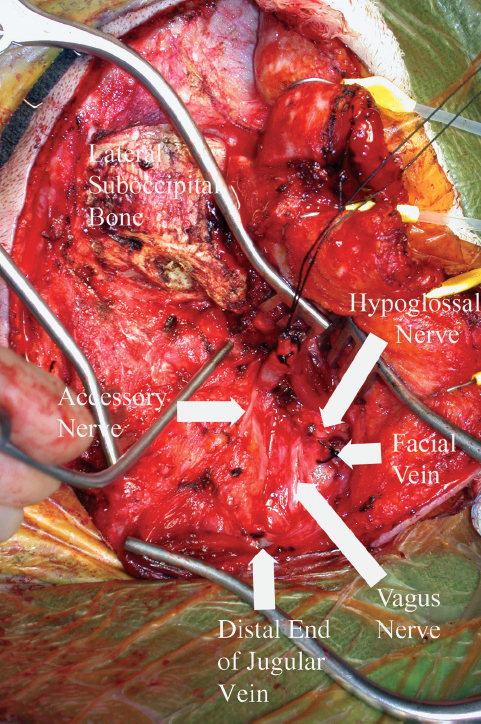
Fig. 13.9 High cervical exposure. The suction device is on the transverse process of the first cervical vertebra.
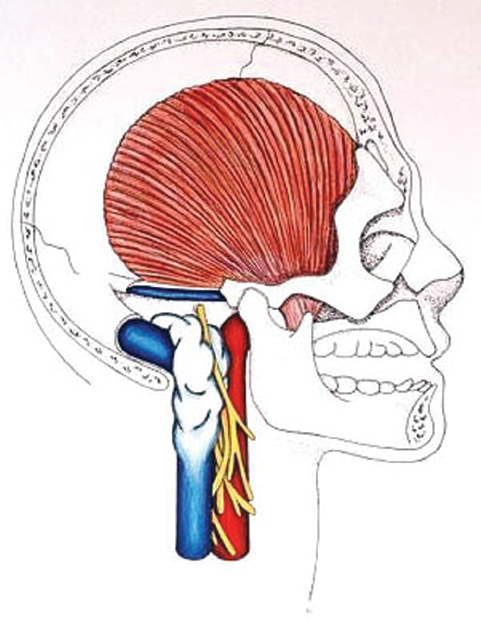
Fig. 13.10 Proximal and distal control of the jugular vein is obtained. A 270-degree exposure of the jugular foramen provides the surgeon excellent visualization of the neurovascular anatomy.
Bone overlying the sigmoid sinus is drilled away, and the presigmoid and retrosigmoid bone is removed to expose the posterior fossa dura. With the skull base cleared of soft tissue attachments (as performed during the high cervical exposure), drilling is continued to reveal the tumor, as well as the jugular bulb. This involves removal of all bone between the posterior end of the digastric groove and the jugular foramen. Attention is then turned to the high cervical carotid artery within the carotid space and the petrous segment of the internal carotid artery within the temporal bone. The bony tympanum, hypotympanum and roof of the glenoid fossa may be drilled away. The mandibular condyle may be displaced anterior-inferiorly or resected, depending on the need for exposure. In our experience, less postoperative pain with little restriction of range of motion of the jaw joint is more common with resection of the mandibular condyle compared with disarticulation of the jaw joint. Drilling continues, beginning at the entrance of the internal carotid artery into the carotid canal. The artery is followed superiorly to the genu adjacent to the eustachian tube and cochlea. If the horizontal segment needs to be exposed, the eustachian tube is divided, inverted, and obliterated with muscle graft, bone wax, and fibrin glue.
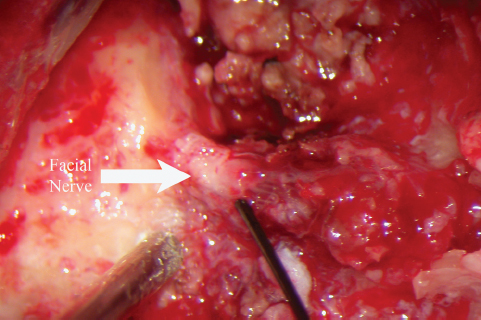
Fig. 13.11 The facial nerve (arrow) is obviously infiltrated with tumor as it descends through the fallopian canal.

Fig. 13.12 The facial nerve has been transposed anteriorly. Ligation of the distal jugular vein is performed inferior to the level of tumor extension.
♦ Tumor Removal
The jugular vein inferior to intraluminal tumor extension is ligated with a silk suture and sharply divided (Fig. 13.12). This maneuver is necessary to prevent embolization of tumor present in the jugular vein distally into the superior vena cava as the tumor is manipulated in its removal. The sigmoid sinus is closed with a silk suture ligature distal to the entry of the superior petrosal sinus into the junction of the transverse-sigmoid sinus (Fig. 13.13). Closure of the sigmoid sinus in this manner enables venous drainage from the superior petrosal sinus in a retrograde fashion. The lateral wall of the sigmoid sinus is resected and mobilized toward the jugular bulb. Working from above and below the bulb, the tumor is removed in a piecemeal fashion. The lower cranial nerves passing through the jugular foramen are protected by the medial wall of the jugular bulb, which must be preserved for the cranial nerves to be protected. Diffuse bleeding can occur within the jugular bulb from the inferior petrosal sinus, which is often represented by multiple small venous channels along the medial wall of the jugular bulb. Gentle packing with Surgicel or soft bone wax controls this venous bleeding. Care must be taken to avoid using bipolar cautery or excessive compressive with cottonoid pledgets to control bleeding along the medial wall of the jugular bulb. Failure to do so may result in a traumatic injury of the lower cranial nerves. During the final steps of tumor dissection, one must directly visualize the carotid artery and the lower cranial nerves to avoid injury (Fig. 13.14).
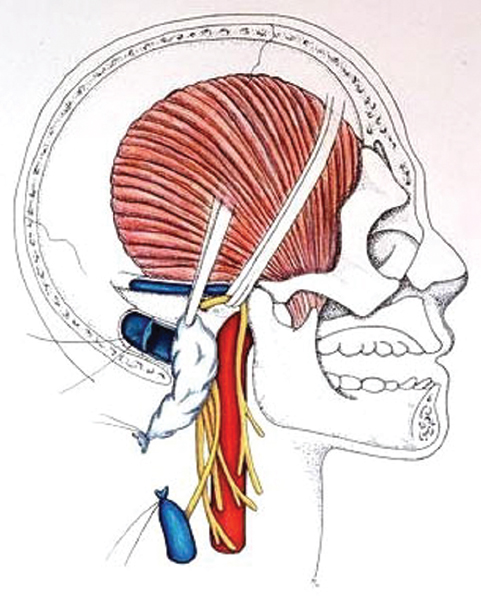
Fig. 13.13 Following ligation of the jugular vein, a 2–0 silk suture is passed circumferential to the sigmoid sinus (distal to the superior petrosal sinus).
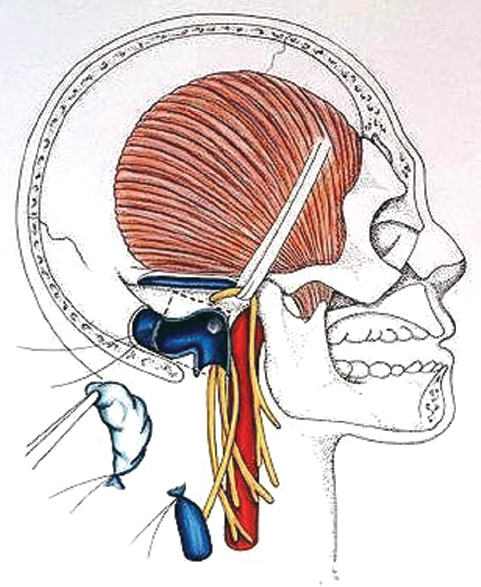
Fig. 13.14 Tumor removal is demonstrated. Care must be taken to preserve the medial wall of the jugular bulb to ensure protection of the lower cranial nerves. The lateral wall of the sigmoid sinus is resected and mobilized toward the jugular bulb.
Most tumors invading the posterior fossa do so in an extradural fashion with displacement of the brainstem and cerebellum. Taking care to maintain this dural layer can help reduce the risk of postoperative spinal fluid leakage. Only with very large tumors will there be absence of a layer of dura between the tumor and the contents of the posterior fossa.
♦ Wound Closure (Fig. 13.15)
If a dural defect has been created by removal of the tumor, this may be repaired primarily with the use of a fascial graft in a watertight fashion. A fat graft harvested from the anterior abdominal wall is placed into the dead space created by the temporal bone dissection. A vascularized musculoperiosteal flap is then rotated inferiorly over the fat graft and tacked down to the soft tissue edges. Incomplete closure of the dural defect requires the use of a lumbar spinal fluid drain postoperatively to reduce the incidence of spinal fluid leakage.
Modified Lateral Skull Base Approach (Fig. 13.16)
This approach is used for patients with small tumors localized to the region of the jugular bulb. Often this select group of patients has functional preoperative hearing, and it is necessary to preserve middle ear function.
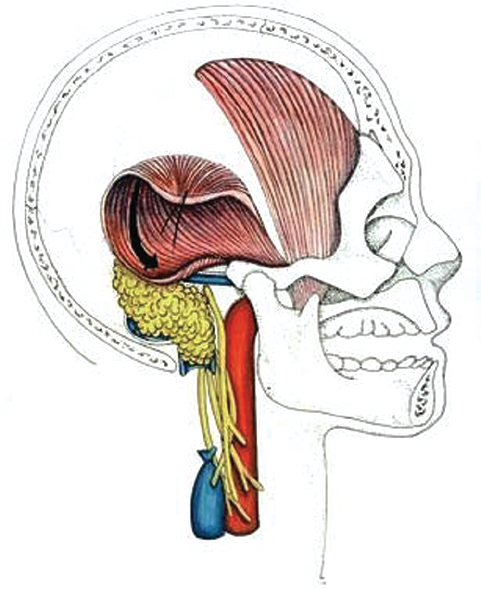
Fig. 13.15 Fat harvested from the abdomen is placed within the dead space of the wound. The vascularized musculoperiosteal flap is then mobilized and placed over the fat. Sutures are then used to affix the flap to the soft tissue edges.
The initial phase of the operation involves the high cervical dissection as described earlier. The temporal bone dissection involves a simple mastoidectomy but limits the bone removal to preserve the posterior bony ear canal wall, tympanic membrane, and the ossicles of the middle ear space. The mastoid segment of the facial nerve rarely would require translocation for tumor removal. Tumor that is present in the middle ear may be removed through the facial recess alone.
Preauricular Infratemporal Fossa Approach
The preauricular infratemporal fossa approach is used for larger tumors with extensive involvement of the petrous internal carotid artery. Further dissection is performed anterior to the ear canal to expose the infratemporal fossa. Disarticulation of the jaw joint is necessary. Bone removal is similar to that in the combined lateral skull base approach, but continued work is focused anteriorly to expose the petrous internal carotid artery to the level of the cavernous sinus.
♦ Management of Major Complications
Cranial Nerve Palsy
Growth of a tumor involving the jugular bulb may result in direct compression or invasion of cranial nerves in the region of the jugular foramen. Most commonly affected are the lower cranial nerves and, less frequently, the facial nerve. Lack of preoperative cranial nerve dysfunction may not correlate with the finding at surgery of tumor invasion of a particular cranial nerve. An acute loss of lower cranial nerve function may be devastating if not properly managed, whereas those patients with chronic loss of function can usually compensate for the deficit. The aggressive management of postoperative cranial neuropathies is absolutely critical to a successful surgical outcome.
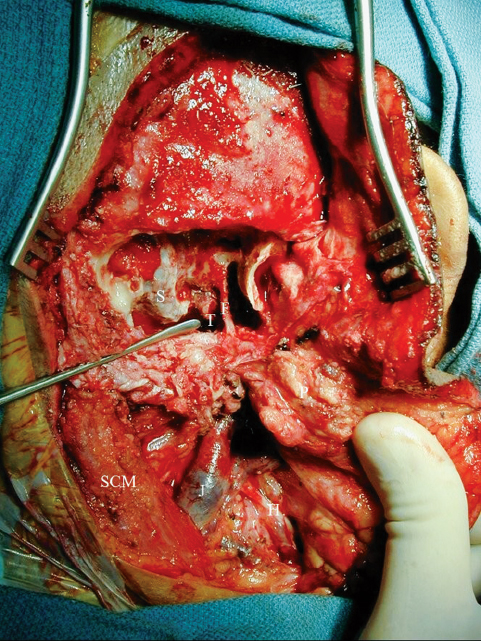
Fig. 13.16 Intraoperative photograph illustrating the modified lateral skull base approach. A simple mastoidectomy has been performed, and the mastoid segment of the facial nerve (F) is skeletonized. The parotid gland is mobilized anteriorly. With the exception of the hypoglossal nerve (H), the lower cranial nerves are hidden deep to the jugular vein (J). Tumor (T) is visualized in the region of the jugular bulb. SCM, sternocleidomastoid.
Injury to the lower cranial nerves is one of the primary risks when operating on tumors of the jugular bulb region. Acute loss of a single lower cranial nerve is usually tolerated, but loss of multiple lower cranial nerves or the vagus nerve may cause a sudden incoordination of swallowing and lead to life-threatening aspiration pneumonia. An acute glossopharyngeal and vagal cranial nerve palsy should be aggressively treated with early placement of a gastrostomy tube. Immediately following the operation, the patient may not manifest symptoms of aspiration secondary to swelling of the vocal cords from the endotracheal tube. However, as the swelling resolves, the patient is at increased risk for aspiration. If the nerve is expected to recover, injection of the vocal cord with Gelfoam will suffice to temporarily protect the patient. If it is not expected to recover, permanent medialization of the vocal cord is recommended. Most patients with preservation of the anatomic continuity of the cranial nerves compensate for swallowing dysfunction and improved phonation over a 3-month period, at which time the gastrostomy tube can be removed. There is a rare patient who presents with regurgitation through the nose secondary to paralysis of the palate, and a palatoplasty effectively corrects this. Persistent dysphagia resulting in recurrent aspiration despite aggressive therapy may require a cricopharyngeal myotomy.
Protection of the eye in a patient with facial cranial nerve palsy is paramount. In the acute postoperative period, a protective eye patch and lubrication is necessary. If the patient’s facial palsy is anticipated to persist beyond 2 months, augmentation of eyelid closure with implantation of a gold weight is recommended. Recovery of facial nerve function postoperatively is almost certain if the anatomic continuity of the facial nerve is preserved during the operative procedure, and the facial nerve responds favorably to electrical stimulation. In patients in whom the continuity of the facial nerve could not be preserved or repaired primarily, a facial-to-hypoglossal anastomosis is performed during the early postoperative period as the patient’s condition permits.
Cerebrospinal Fluid Leak/Wound Healing
Cerebrospinal fluid (CSF) leakage following resection of tumors involving the jugular foramen is related to several factors: the size of the tumor and involvement of the dura, the nutritional status of the patient, and the state of cerebrospinal fluid dynamics.
Careful attention is paid to achieving a watertight closure of the dural with either a primary repair or with a fascial graft. The dural defect is usually present with resection of a large tumor at the level of the jugular foramen. This dural defect is repaired primarily with a fascial graft if possible, placement of a fat graft to obliterate the dead space of the surgical wound, and rotation of a vascularized graft over the adipose tissue of the surgical defect followed by a multilayered tissue closure. A postoperative lumbar spinal drain is advised if dural closure is not achieved. The prevention of spinal fluid rhinorrhea and otorrhea is accomplished by closure of the eustachian tube and external auditory canal during the surgical procedure.
Early nutritional supplementation is mandatory to promote tissue healing. If the patient is not able to tolerate feedings by mouth, a small-diameter feeding tube is inserted within the first 24 hours. Alimentation is immediately begun. If oral feedings are not possible beyond 10 to 14 days postoperatively, a percutaneous endoscopic gastrostomy (PEG) is recommended.
Altered CSF dynamics may lead to postoperative hydrocephalus and result in CSF leakage. The altered spinal fluid dynamics may result from contamination of the wound by blood products, bone dust, or from occlusion of the venous circulation at the level of the jugular bulb. The use of continuous lumbar spinal fluid drainage for 3 to 7 days postoperatively aids in the normalization of intracranial pressure, clearing of the spinal fluid circulation, and prevention of the accumulation of fluid under the wound flap. This promotes sealing of the dural defect and healing of the wound. We have found that the impairment of CSF absorption is usually transient, thus obviating the need for permanent cerebrospinal fluid diversion.
Vascular Injury
In larger tumors that encase the internal carotid artery, there is a risk of injury to the artery. Every patient that demonstrates involvement of the carotid artery undergoes temporary balloon test occlusion to determine the adequacy of collateral circulation. Surgical removal of tumor proceeds with the intent of always preserving the internal carotid artery. If injury to the carotid artery should occur, temporary occlusion of the artery followed by a primary repair is attempted using 9–0 Prolene vascular suture. If this cannot be performed, the results of the balloon occlusion test dictate whether the artery is sacrificed or an arterial bypass is utilized. A saphenous vein interposition graft is recommended if technically possible. A superficial temporal artery–middle cerebral artery bypass may not be an option because of preoperative embolization of the external carotid artery circulation.
♦ Conclusion
Although appearing histologically benign, the majority of tumors involving the jugular foramen are characterized by unpredictable biologic behavior. Tumors originating in the jugular bulb region present a significant surgical challenge. Successful treatment of jugular bulb tumors mandates a clear understanding of the anatomy of the jugular foramen and the natural history of the pathology of the tumor. With the significant advances in surgical approaches to the lateral skull base, surgeons can now accomplish complete extirpation of the majority of jugular foramen tumors with low morbidity rates and good long-term outcomes.
References
1. Seiffert A. Cited in Lundgren N. Tympanic body tumors in the middle ear-Tumors of carotid body type. Acta Otolaryngol 1949;37:366–379
2. Guild SR. A hitherto unrecognized structure, the glomus jugularis in man. Anat Rec 1941;79(Suppl 2):28
3. Albernaz JG, Bucy PC. Nonchromaffin paraganglioma of the jugular foramen. J Neurosurg 1953;10:663–671 PubMed
4. Lundgren N. Tympanic body tumors of the middle ear—tumors of the carotid body type. Acta Otolaryngol 1949;37:366–379
5. William H, Childs DS Jr, Parkhill EM, et al. Chemodectomas of the glomus jugulare (nonchromaffin paragangliomas) with especial reference to their response to roentgen therapy. Trans Am Otol Soc 1955;43:264
6. Riemenschneider PA, Hoople GD, Brewer D, Jones D, Ecker A. Roentgenographic diagnosis of tumors of the glomus jugularis. Am J Roentgenol Radium Ther Nucl Med 1953;69:59–65 PubMed
7. Valvassori GE. Valvassori GE. Laminagraphy of the ear. Normal roentgenographic anatomy. Am J Roentgenol Radium Ther Nucl Med 1963; 89:1155–1167 PubMed
8. Gejrot T. Retrograde jugularography in the diagnosis of abnormalities of the superior bulb of the internal jugular vein. Acta Otolaryngol 1964; 57:177–180 PubMed
9. Shapiro MJ, Neues DK. Technique for removal of glomus jugulare tumors. Arch Otolaryngol 1964;79:219–224 PubMed
10. Gejrot T. Surgical treatment of glomus jugulare tumors. With special reference to the diagnostic value of retrograde jugularography. Acta Otolaryngol 1965;60:150–168 PubMed
11. Gardner G, Cocke EW Jr, Robertson JT, Trumbull ML, Palmer RE. Combined approach surgery for removal of glomus jugulare tumors. Laryngoscope 1977;87(5 Pt 1):665–688 PubMed
12. Fisch U. Infratemporal fossa approach for extensive tumors of the temporal bone and base of the skull. In: Silverstein H, Norrell H, eds. Neurological Surgery of the Ear. Birmingham: Aesculapius, 1977:34–53
13. Fisch U. Infratemporal fossa approach to tumours of the temporal bone and base of the skull. J Laryngol Otol 1978;92:949–967 PubMed
14. Jenkins HA, Fisch U. Glomus tumors of the temporal region. Technique of surgical resection. Arch Otolaryngol 1981;107:209–214 PubMed
15. Hilal SK, Michelsen JW. Therapeutic percutaneous embolization for extra-axial vascular lesions of the head, neck, and spine. J Neurosurg 1975;43:275–287 PubMed
16. Brismar J, Cronqvist S. Therapeutic embolization in the external carotid artery region. Acta Radiol Diagn (Stockh) 1978;19:715–731 PubMed
17. Simpson GT II, Konrad HR, Takahashi M, House J. Immediate postembolization excision of glomus jugulare tumors: advantages of new combined techniques. Arch Otolaryngol 1979;105:639–643 PubMed
18. Murphy TP, Brackmann DE. Effects of preoperative embolization on glomus jugulare tumors. Laryngoscope 1989;99:1244–1247 PubMed
19. Kaye AH, Hahn JF, Kinney SE, Hardy RW Jr, Bay JW. Jugular foramen schwannomas. J Neurosurg 1984;60:1045–1053 PubMed
20. Nager GT, Heroy J, Hoeplinger M. Meningiomas invading the temporal bone with extension to the neck. Am J Otolaryngol 1983;4:297–324 PubMed
21. Origitano TC, al-Mefty O, Leonetti JP, DeMonte F, Reichman OH. Vascular considerations and complications in cranial base surgery. Neurosurgery 1994;35:351–362, discussion 362–363 PubMed
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








