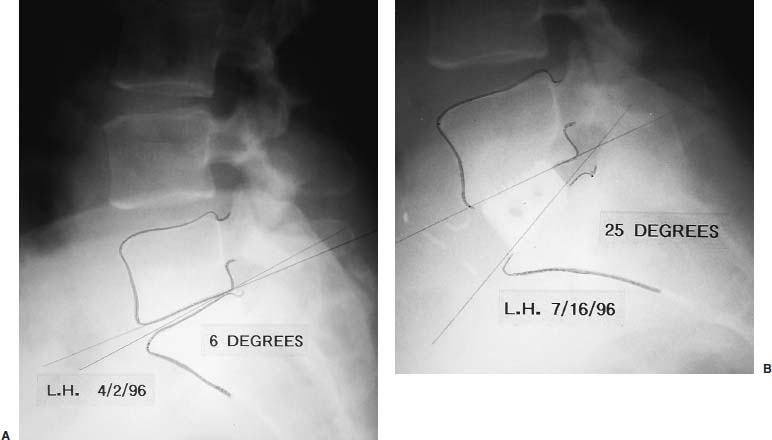
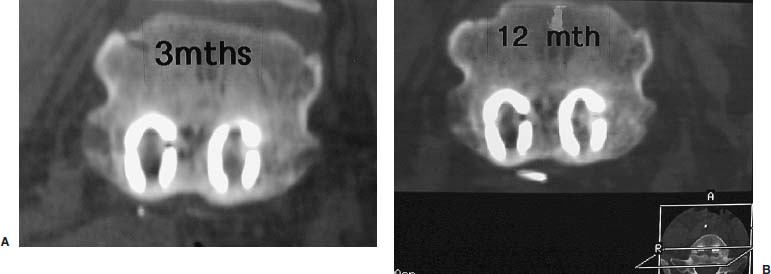
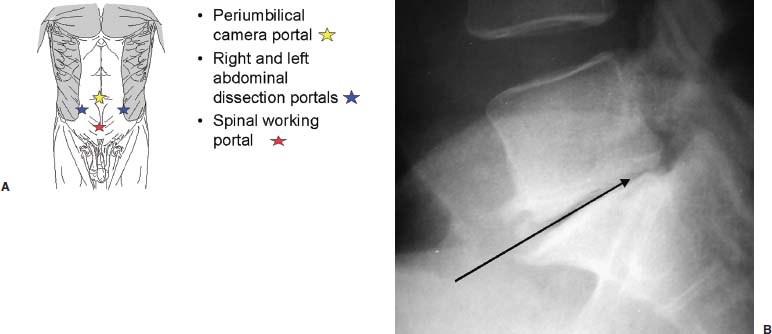
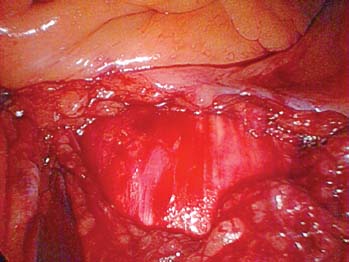
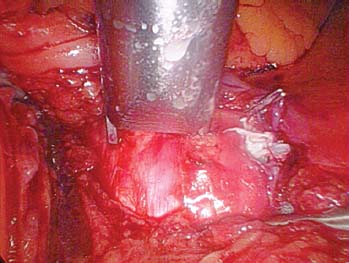
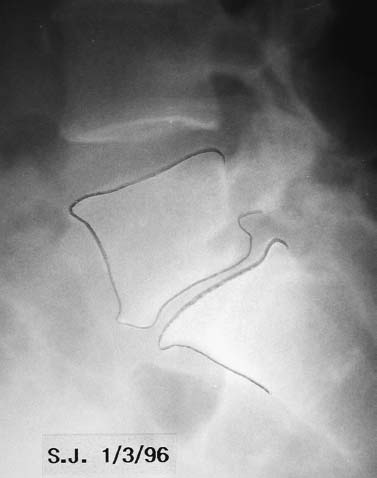
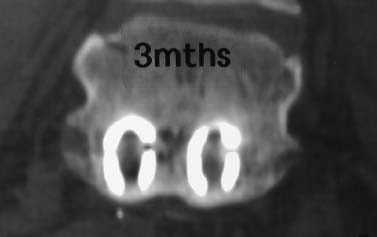
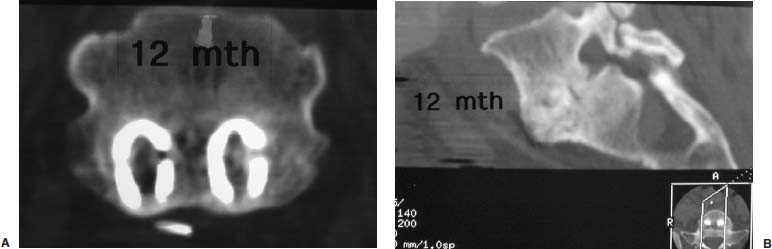
The evolution of threaded interbody fusion cages has presented a significant new form of segmental fixation (interbody fixation devices) for application to the lumbar and lumbosacral spine. These devices serve as carriers for such fusion material as autogenous cancellous bone or, more recently, bone morphogenetic protein, while restoring segmental stability to the involved motion segment. The biomechanics of these devices greatly surpass those of the interbody spacers commonly used (i.e., femoral ring composite grafts, tricortical wedges, and various impacted “containment devices” for cancellous bone), which have not offered significant stability in flexion/extension or torsion. In fact, several biomechanical studies have shown this recent class of construct to compare quite favorably with the segmental pedicular fixation/posterior lumbar interbody fusion (PLIF) constructs.1 Interbody spacers have traditionally been utilized as a component of an overall construct employing segmental posterior instrumentation. In contrast, when applied effectively, the threaded interbody cages are able to confer a degree of stability to the symptomatic motion segment so as to function as stand-alone constructs. The biomechanics of the interbody cage constructs are generally based on significant preoperative disk space narrowing. The act of reconstituting the anterior column height restores the annular tension, which is a key factor in the proper construct mechanics (Fig. 26–1). It is in such an application that the favorable comparison to pedicular fixation/PLIF constructs can be obtained. Inappropriate size and/or placement of the threaded interbody devices may ineffectively create tension in the anulus fibrosus, resulting in a biomechanically compromised stand-alone construct. In these instances, the lack of annular tension results in the cage system functioning as an interbody spacer rather than as an interbody fixation device; supplemental stabilization techniques may be required. This failure to obtain adequate distraction of the anulus fibrosus has been among the most common reasons for failure of the procedure. With regard to the degree of disk space narrowing, the authors generally consider patients with 50% or more disk space narrowing to be appropriate candidates for a stand-alone interbody construct. In addition, radiographic findings of significant end-plate sclerosis and significant traction spurs correlate well with the ability to tension the anulus fibrosis intraoperatively. Increasing the tension of the anulus fibrosus in the interbody construct was found to be associated with higher biomechanical strength in biomechanical studies. Laparoscopic Application of Threaded Interbody Fusion Devices The laparoscopic application of interbody cage devices is simply a variance in the surgical approach to the spine. It is of paramount importance that the surgeon remains cognizant of the biomechanics of stand-alone interbody constructs regardless of the surgical approach selected for their placement. With adherence to these concepts, the authors have found laparoscopic techniques to offer a significant reduction in the operative morbidity associated with the anterior approach. FIGURE 26–1 (A) Plain radiograph showing markedly narrowed preoperative disk space at the L5–S1 disk space. (B) Postoperative radiograph showing restoration of anterior column height and annular tension by interbody fixation device. Initial Clinical Results of Laparoscopic Interbody Fusion The authors have had the opportunity to participate at one of the initial clinical centers for the development of the laparoscopic application of the BAK, threaded femoral cortical bone dowel, and tapered LT Interbody Cage systems (Medtronic-Sofamor Danek, Memphis, TN) since early 1994. The first cases of the laparoscopic technique of interbody cage placement utilized the BAK cage.2 This initial experience demonstrated an important pattern of learning curves associated with this procedure: operative time, length of hospitalization, and complications. In comparing the open and laparoscopic insertion techniques of the BAK cage during the initial learning curve phase of the procedure, the authors were able to demonstrate a significant decrease in operative morbidity by the reduction in the length of hospitalization. With further experience following this initial clinical trial, we were able to further decrease the length of hospitalization to an average of 23 hours for each of these cage systems, as well as the cylindrical Interfix cage. The open application of interbody cages has produced an average 3-day hospitalization over the same time period. More recently, the authors participated at one of the initial pilot trial centers studying the use of recombinant bone morphogenic protein (rhBMP-2; InFuse, Medtronic-Sofamor Danek) with the LT Interbody Cage system (Medtronic-Sofamor Danek). We utilized the laparoscopic application technique for cage/BMP placement while the additional study sites employed a retroperitioneal open approach. The laparoscopic technique provided a remarkable 12-hour average hospitalization compared with the 3-day hospitalization for patients who underwent the open retroperitoneal technique. On the one hand, the further reduction in the length of hospitalization (vs. 23 hours) realized with the use of rhBMP-2 (InFuse) is believed to represent the procedural morbidity of the iliac bone graft harvesting in the earlier clinical trials. On the other hand, the consistent 3-day hospitalization of the retroperitoneal open procedure with either rhBMP-2 (InFuse) or iliac crest bone graft represents the morbidity of the open exposure technique itself. With the possibility of a true same-day discharge, the approach to patient education must address this. Furthermore, the nursing unit staff will need to be educated on both this possibility as well as on the generally much more rapid mobilization of these patients compared with those who undergo an open anterior approach, or certainly the more common posterior instrumented spinal fusion procedures. FIGURE 26–2 Postoperative CT of the LS–S1 interspace of the fusion using the LT Interbody cage system and rhBMP-2 (InFuse). (A) 3 months. (B) 12 months. The radiographic appearance of the pattern of fusion in laparoscopic patients is of even greater experience. Whereas the open patient group underwent complete diskectomies, laparoscopic patients underwent “channel diskectomies”; that is, only the disk material specifically was removed. In all of these patients (i.e., open or laparoscopic) no bone graft material was used. Figure 26–2 demonstrates the postoperative CT images of a patient who underwent an L5–S1 laparoscopic anterior interbody fusion with rhBMP-2 (InFuse) and the LT Interbody Cage system. Ossification within the confines of the disk space is clearly visible, as are the areas where the intervertebral disk had not been excised. This is felt to represent a biological induction of the remaining nucleus pulposus to ossify. Indications The current indications for the use of threaded interbody cages, either metallic or allograft, include: As has been previously mentioned, the “stand-alone” use of the threaded interbody cages requires a significant preoperative narrowing of the symptomatic motion segment (Fig. 26–1). If the application is to a wide disk space, the use of supplementary posterior segmental instrumentation is appropriate. In cases of spondylolytic spondylolisthesis, the lack of integrity of the pars intra-articularis seems to be associated with a greater laxity in the anulus fibrosus—this laxity, in fact, is one of the primary sources of the actual translatory deformity present. This fatigue, or laxity, of the anulus fibrosus can lead to a less definitive end point in disk space distraction with possible implications for fixation. This has led the authors to treat spondylolytic spondylolisthesis with an interbody cage device (as a stand-alone construct) only when there is marked disk space narrowing. The radiographic findings of end plate sclerosis and significant traction spur formation correlate clinically with the reasonable expectation of being able to place tension the anulus fibrosus and thereby produce a stable standalone construct. If distraction does not seem to produce an appropriate annular tension, however, the surgeon must recognize this and consider supplementary posterior fixation. Last, in the instance of L5–S1 spondylolisthesis, the surgeon must ensure that the angle of the disk space does not pass into or below the pubic symphysis on the lateral radiograph. The anterior application of interbody cage devices requires a colinear approach to the disk space. Such an angulation would therefore prohibit appropriate cage placement. Contraindications Commensurate with the importance of significant preoperative narrowing of the involved disk space, a symptomatic motion segment with a well-preserved disk space would be contraindicated for a stand-alone interbody construct. The general contraindications include: Surgical Technique The surgical team is composed of: It is imperative that the general/access surgeon and the spine surgeon truly view themselves as a team. The access surgeon will need to realize the magnitude of – exposure required for the spinal portion of the procedure, particularly the degree to which the great vessels will need to be mobilized. It is especially important that the spine surgeon/access surgeon team first experience the procedure together in a series of open interbody cage fusions prior to progressing to the laparoscopic approach. Preprocedural preparation is of primary importance in ensuring a neutral orientation of the spine and in avoiding difficulties in obtaining intraoperative fluoroscopic images. These are each key factors in the determination of cage placement within the disk space. Whether performed via an open or a laparoscopic approach, this is truly a “procedure of millimeters.” The patient is positioned supine on a radiolucent operating table. The authors prefer to elevate the patient’s pelvis and lumbar spine from the surface of the operating table with folded surgical blankets. This then permits the arms to be positioned along the patient’s side (tucked and padded) without obscuring the lateral fluoroscopic visualization of the symptomatic motion segment (Fig. 26–3). The spine should not be placed into a position of lordosis by way of bolsters or other positioning influences. FIGURE 26–3 Patient positioning. Bucks traction boots help keep the patient from sliding up while in Trendelenburg’s position. The ability to maneuver the C-arm into position and to visualize the pathologic motion segment clearly must also be assessed—the table upright will determine how high on the table the patient must be placed. Doublecheck the fluoroscopy with the requisite Trendelenburg positioning to be used during the procedure. In addition to visualizing the motion segment on the lateral image, the anteroposterior/Ferguson view must show a neutral motion segment rotation prior to proceeding. The bladder is decompressed with a Foley catheter and the stomach with an orogastric tube, with both removed at the completion of the procedure. All routine positioning checks (i.e., padding of neurovascular structures/bony prominences) are also rechecked. It must be recalled that the laparoscopic procedure requires up to 30 degrees of Trendelenburg position throughout its duration, as opposed to the open anterior application of the interbody cages. General Laparoscopic Technique Exposure For L4–L5 and L5–S1, the standard portal placement includes a periumbilical laparoscopic portal, 5 mm right and left of the lower abdominal quadrant dissection/retraction portals, and a suprapubic spinal working portal (Fig. 26–4A). The initial portals placed include the camera and right/left lower quadrant sites. Using these portals, the dissection is performed, and the symptomatic motion segment is exposed. The suprapubic spinal working portal is created only after the involved disk space is verified. This portal must present a colinear approach to the disk space, the line of approach, which will represent the orientation of the interbody cage within the disk space. In other words, if a nonlinear approach from the spinal working portal to the disk space exists, then this is the same orientation the implanted cage will take with the motion segment (Fig. 26–4B). FIGURE 26–4 (A) Laparoscopic portal placement. (B) Determining the placement of the suprapubic spinal working portal. The authors place a percutaneous long spinal needle and fluoroscopically verify the optimal spinal working portal location. In the course of dissection, it is important to avoid the use of monopolar cautery upon opening the posterior peritoneum. This minimizes the risk of retrograde ejaculation. It also avoids extraneous marks on the anterior anulus fibrosus. The midline of the motion segment is determined with fluoroscopy, again ensuring the neutral rotation of the spine as well. The accurate placement of the interbody cages is very much dependent upon this landmark—again, this is truly an “operation of millimeters,” whether performed open or laparoscopically. Cage Insertion Depending on the specific cage system being used, the sequence of cage placement may vary; however, there are several points that are generally not system dependent: Technique for Laparoscopic Placement of LT Interbody Cages FIGURE 26–5 Operative photograph showing the complete exposure of the L5–S1 disk. FIGURE 26–6 Operative photograph showing starting guide being positioned to open the left L5–S1 discal entry site. Complications As stated earlier, the initial laparoscopic BAK series revealed a significant learning curve with regard to complications. It appears that the most significant and highest frequency of complications occur in the first 10 to 15 cases in a surgeon’s experience. The importance of the laparoscopic access surgeon and the spine surgeon working as a team on several open interbody cage procedures prior to evolving to the laparoscopic technique cannot be overemphasized. The complications in the original 118 laparoscopic BAK cases among the five clinical trial sites are shown in Table 26–1. Recall that these complications occurred as each of the study sites were indeed passing through their “learning curve” phase. Also, the initial clinical sites involved in this first laparoscopic clinical series were truly the first to perform the procedure. For that reason, the technique was evolving while the surgical teams progressed through their learning curve. It is for these reasons that the authors feel these may represent a worstcase scenario. With the availability of hands-on surgical technique courses and the opportunity to visit surgical teams well established in the procedure, it is reasonably expected that the current learning curve should be safer. In terms of fusion, each of the cage series utilizing iliac crest bone graft resulted in a 5 to 10% pseudarthrosis rate. No pseudarthroses occurred in the InFuse (rhBMP-2) patients. Case Illustrations A 62-year-old female underwent an L5–S1 laparoscopic interbody fusion with the LT interbody cage and InFuse (rhBMP-2). The preoperative diagnosis was a grade I L5–S1 spondylolytic spondylolisthesis and functionally limiting mechanical low back pain. Preoperative provocative diskography had confirmed the pain origin and also shown L4–L5 to be asymptomatic (Fig. 26–7). The laparoscopic interbody fusion was performed uneventfully, and the patient was discharged to home that afternoon, independent in ambulation and requiring only limited oral analgesics. The preoperative mechanical low-back pain was relieved immediately, and her incisional discomfort resolved over the ensuing 2 weeks as expected. At 3 months a CT scan to assess fusion was performed (Fig. 26–8) and revealed a well-developed interbody fusion mass. FIGURE 26–7 Preoperative lateral radiograph showing narrowing of the L5–S1 disk space. FIGURE 26–8 Coronal CT scan showing well-developed interbody fusion mass at 3 months following surgery. The formation of bone throughout the anatomical confines of the interspace was of particular interest, despite the fact that the disk space was debrided only where the interbody cages were placed. Furthermore, there was no evidence of heterotopic ossification. Repeat CT scans performed at 1 year revealed the further maturation of the interbody fusion without heterotopic extension (Fig. 26–9). The patient recovered and rehabilitated very well clinically. She voiced complete resolution of the preoperative mechanical low-back pain and indicated she was functionally unlimited. In the authors’ experience, the same-day discharge is typical for patients treated with the laparoscopic approach to the spine and InFuse/LT interbody devices. The particular advantages of interbody cages include the beneficial effects on bone healing associated with the anterior column location, the stabilization potential in appropriate motion segments, the ability to restore anterior column height and foraminal volume, the correction of segmental lordosis with the LT interbody device, and the avoidance of the fusion disease phenomenon. These advantages are not dependent on whether the approach of insertion is open or laparoscopic. The specific and demonstrated chief benefit of the laparoscopic approach to interbody cage insertion is the reduction in surgical morbidity. This is especially evident when the laparoscopic technique is combined with the use of bone morphogenic protein (rh-BMP2/In-Fuse). It is our belief that this significant reduction in patient morbidity will drive the further acceptance of the laparoscopic approach in a manner similar to the evolution of arthroscopic orthopedic surgery and laparoscopic general surgery. The main disadvantage of the laparoscopic approach is the initial learning curve of the surgical team with the technique. In addition, the anterior approach to cage placement (open or laparoscopic) is limited, at this time, with regard to the inability to decompress the spinal canal directly. It is recommended that interbody cages be considered as only one option in the stabilization of symptomatic motion segments. With care in patient selection, their ability to function in a stand-alone configuration has been successfully demonstrated. To evolve to the laparoscopic placement technique of interbody cages, the access surgeon and spine surgeon should begin as a team with the open approach, placing the particular instrumentation system they plan to use laparoscopically. Together, the access surgeon and spine surgeon should attend a hands-on laparoscopic course for the specific instrumentation system. In terms of initial case selection, they should begin with nondeformity L5–S1 cases and, most importantly, allow for time for the cases. FIGURE 26–9 Coronal (A) and lateral (B) CT reconstruction images showing interbody fusion at 12 months. Note the absence of heterotopic ossification and the essentially complete ossification of the disk space. REFERENCES
26

Laparoscopic Fusion of the Lumbosacral Spine


< div class='tao-gold-member'>
Laparoscopic Fusion of the Lumbosacral Spine
Only gold members can continue reading. Log In or Register to continue

Full access? Get Clinical Tree








