CHAPTER 388 Microsurgical Management of Arteriovenous Malformations
General Considerations
In principle, AVMs are arteriovenous shunts composed of feeding arteries that connect into an abnormal vascular nest (nidus), which in turn is drained by abnormal veins. Consequently, there is no or only an abnormal capillary bed. The so-called nidus is the network of channels that is interposed between the feeding arteries and draining veins. A compact nidus is characterized by a well-circumscribed vascular network, whereas a diffuse nidus refers to an abnormal vascular network that is more widespread within normal brain parenchyma.1 In both cases the abnormal vessel conglomerate is intermingled with gliotic brain tissue. This pathology needs to be differentiated from a dural AVM, or fistula, which has no nidus but a more direct communication between enlarged arteries and veins. AVMs are thought to be congenital in nature, but they frequently grow during childhood, adolescence, and young adulthood. It is rare for them to grow significantly in more mature adults. Therefore, it is important to stress that these lesions are not neoplastic and thus the term “angioma” should be avoided because it is inappropriate. Yet it is becoming more and more evident that these lesions also exhibit dynamic features that make them change over time, a fact that could influence even “successfully treated” AVMs.
Table 388-E1 ![]() isa compilation of the natural history studies that we consider most important.2–7 In summary, patients with cerebral AVMs bleed at a rate of approximately 2% to 4% per year. For decision making and timing of potential procedures, it is crucial to understand that this rate of bleeding is similar in most studies regardless of whether the patient has ever bled, although patients initially seen with hemorrhage have about twice the risk for rebleeding during the first year. It appears clear, then, that unruptured cerebral AVMs have a much greater annual rate of bleeding than unruptured aneurysms do, although of course the consequences of hemorrhage from an aneurysm are worse than those of hemorrhage from an AVM; AVM hemorrhages result in significant morbidity in about 25% to 30% of patients and death in approximately 10%.8
isa compilation of the natural history studies that we consider most important.2–7 In summary, patients with cerebral AVMs bleed at a rate of approximately 2% to 4% per year. For decision making and timing of potential procedures, it is crucial to understand that this rate of bleeding is similar in most studies regardless of whether the patient has ever bled, although patients initially seen with hemorrhage have about twice the risk for rebleeding during the first year. It appears clear, then, that unruptured cerebral AVMs have a much greater annual rate of bleeding than unruptured aneurysms do, although of course the consequences of hemorrhage from an aneurysm are worse than those of hemorrhage from an AVM; AVM hemorrhages result in significant morbidity in about 25% to 30% of patients and death in approximately 10%.8
Spontaneous obliteration of an AVM is rare, but does occur, and has been reported in single cases.9–13 Several factors appear to be associated with spontaneous occlusion: a single draining vein, a solitary arterial feeder, and nidus size smaller than 3 cm.14,15
In the process of decision making, it is important to try to understand what factors may influence the rate of bleeding, which undoubtedly is not identical for every patient with an AVM. Several anatomic and hemodynamic factors seem to increase the risk for bleeding. Apart from associated aneurysms on the feeding artery (see later), the significance of nidus size has engendered controversy, with some suggesting that small AVMs bleed more frequently.16,17 Deep location has been suggested to increase the risk for hemorrhage (basal ganglia, periventricular or intraventricular space, posterior fossa),18,19 as has deep venous drainage,20 feeding by perforators, and location in the vertebrobasilar system.21 We believe that an intraventricular or periventricular location has a higher risk for hemorrhage, although we cannot substantiate this opinion with numbers. The role of feeding artery and draining vein pressure in this regard has been investigated. Norbash and coworkers saw no correlation in a group of 32 patients.22 In a large series of 340 reviewed patients, Duong and colleagues identified “high arterial input pressure” and “venous outflow restriction (exclusively deep venous drainage)” as the most powerful risk predictors for a hemorrhagic AVM manifestation.23 We also believe that venous outlet obstruction potentiates the risk for hemorrhage. Spetzler and colleagues associated AVM size smaller than 3 cm with higher feeding artery pressure and larger hemorrhage.16 Graf and collaborators calculated a 5-year risk for hemorrhage of 10% for AVMs larger than 3 cm and 52% for AVMs smaller than 3 cm.2 Whether smaller size really results in a higher risk for bleeding is still subject to controversy.
Pregnancy does not seem to increase the likelihood of hemorrhage.24,25 Horton and colleagues reviewed the cases of 451 women with AVMs who had 540 pregnancies, 17 of which were complicated by hemorrhage. None of the hemorrhages occurred during labor, vaginal delivery, or cesarean section. The authors concluded that the method of delivery should be based on obstetric consideration alone and the decision to operate after intracranial hemorrhage “should be based on neurosurgical principles.”25
Microsurgical Treatment
Indications for Microsurgical Treatment
Patient-Related Factors
When evaluating a patient for treatment of an AVM, we must consider factors such as age, general health, neurological status, history of recent or past hemorrhage, symptoms from the AVM, occupation and hobbies, and psychological makeup. The patient’s age is most important in determining the cumulative risk for AVM rupture during the remainder of the patient’s life expectancy. Assuming an annual hemorrhage rate of 2% to 4% and an average life expectancy of 70 years, the cumulative risk (in percentage) for AVM rupture may be estimated by the following formula: 105 minus the patient’s age in years.26,27 Younger age may thus justify a more aggressive approach because the cumulative risk is so high. At the same time, of course, it is easier for the young to tolerate a prolonged operation, and the chances for satisfactory recovery from any neurological deficit that might occur are better. Likewise, the general health of the patient needs to be taken into account. The current overall and neurological condition of a patient may dictate the timing, and severe comorbid conditions may preclude surgery. The occupation and lifestyle of a patient are also important aspects to consider. For a schoolteacher or lawyer it may be intolerable to have a speech deficit, but a mild visual field deficit might not hinder them from practicing their profession, whereas the latter would be intolerable for a pilot or truck driver. Some patients simply cannot live with the thought of harboring a brain lesion that has a relatively high potential to bleed at some point, whereas others live perfectly well with this knowledge and might prefer to decline treatment.
Arteriovenous Malformation–Related Factors
Location, size, configuration (compact versus diffuse), location and pattern of arterial supply, presence of deep perforators, location and pattern of venous drainage, evidence of outflow obstruction, associated aneurysms, blood flow to the AVM, evidence of “steal,” and fresh or old hematoma from the AVM are all factors that must be considered in determining the feasibility of surgery and the involved surgical risk.28 To help in this process, several classifications have been developed,29 including the one that is used most frequently today—the Spetzler-Martin grading scheme.30 Although such a classification is very helpful, especially in terms of reporting and comparing results, no classification could possibly take all the necessary variables into account that an experienced surgeon should consider when estimating surgical risk. Such factors as the presence of deep perforator arterial supply,31 location of the venous drainage, nidus configuration, and others would be difficult to account for in a classification that is still of practical use.32 Other classifications that have attempted to specifically address the risk for hemorrhage from an AVM (in contrast to surgical risk) never gained popularity in daily use (e.g., that of Nataf and coworkers20).
Embolization
Embolization is used with curative intention, as a palliative maneuver, or before surgical excision33,34 or radiosurgery. Partial embolization has its indications for palliation and presurgical treatment, but even in experienced hands it carries substantial risk. As can be seen from the review in![]() Table 388-E2,35–53 embolization in all its forms has morbidity and mortality rates that are not negligible, with morbidity ranging from 2% to 27% and mortality from 0% to 8%. Preoperative embolization has made it possible to operate on cerebral AVMs that before this technique became available could not undergo surgery without risking substantial morbidity. Frequent use has also been made of pre-radiosurgical embolization. The rationale to “downsize” large inoperable AVMs in an attempt to reach a so-called critical size for effective Gamma Knife surgery is very weak in our opinion. In many of these cases radiosurgery fails to completely obliterate the nidus, and in fact it has been shown to reduce the obliteration rate of linear accelerator radiosurgical treatment.54 Previous embolization had been identified as a negative predictor of successful AVM radiosurgery.55 After it became evident that particle embolization before radiosurgery resulted in a 15% to 20% recanalization rate, it was generally abandoned and liquid embolic agents are now preferred.55–57 However, with these agents the problem is inadvertent embolization of the feeding artery, thus resulting in proximal occlusion with only minimal or no nidal penetration.33 Inadvertent, premature closure of draining veins is a specific significant risk with all liquid agents. With a partially “open” nidus, creation of new arteriovenous shunts can be anticipated. In addition, there is no evidence that pre-radiosurgical embolization reduces the risk for hemorrhage during the critical period after radiosurgery until the induced devascularization and fibrosis lead to complete obliteration of the nidus.33 This is why in general we see only rare indications for preradiosurgical embolization.
Table 388-E2,35–53 embolization in all its forms has morbidity and mortality rates that are not negligible, with morbidity ranging from 2% to 27% and mortality from 0% to 8%. Preoperative embolization has made it possible to operate on cerebral AVMs that before this technique became available could not undergo surgery without risking substantial morbidity. Frequent use has also been made of pre-radiosurgical embolization. The rationale to “downsize” large inoperable AVMs in an attempt to reach a so-called critical size for effective Gamma Knife surgery is very weak in our opinion. In many of these cases radiosurgery fails to completely obliterate the nidus, and in fact it has been shown to reduce the obliteration rate of linear accelerator radiosurgical treatment.54 Previous embolization had been identified as a negative predictor of successful AVM radiosurgery.55 After it became evident that particle embolization before radiosurgery resulted in a 15% to 20% recanalization rate, it was generally abandoned and liquid embolic agents are now preferred.55–57 However, with these agents the problem is inadvertent embolization of the feeding artery, thus resulting in proximal occlusion with only minimal or no nidal penetration.33 Inadvertent, premature closure of draining veins is a specific significant risk with all liquid agents. With a partially “open” nidus, creation of new arteriovenous shunts can be anticipated. In addition, there is no evidence that pre-radiosurgical embolization reduces the risk for hemorrhage during the critical period after radiosurgery until the induced devascularization and fibrosis lead to complete obliteration of the nidus.33 This is why in general we see only rare indications for preradiosurgical embolization.
Complete cure of AVMs with embolization alone is usually very difficult to achieve, although in very experienced hands success rates have reached 40% ![]() (Table388-E2). Complete cure in this context should be defined as disappearance of the nidus and early venous drainage. Generally, only relatively “simple” AVMs with readily accessible feeders are potential candidates for a curative endovascular approach. However, in many instances these AVMs are also the ones that are easy to remove surgically with minimal morbidity but with a higher certainty of complete resection. Therefore, we would recommend attempts at endovascular curative obliteration only for lesions that in addition to accessible feeders have a deep, critical location, where the risk associated with surgery would exceed the risk related to embolization, or in patients with advanced age or significant comorbid conditions.
(Table388-E2). Complete cure in this context should be defined as disappearance of the nidus and early venous drainage. Generally, only relatively “simple” AVMs with readily accessible feeders are potential candidates for a curative endovascular approach. However, in many instances these AVMs are also the ones that are easy to remove surgically with minimal morbidity but with a higher certainty of complete resection. Therefore, we would recommend attempts at endovascular curative obliteration only for lesions that in addition to accessible feeders have a deep, critical location, where the risk associated with surgery would exceed the risk related to embolization, or in patients with advanced age or significant comorbid conditions.
In general, we believe that there are very limited indications for palliative embolization because the procedure changes flow dynamics and the actual bleeding risk can be increased with this form of partial treatment. The best indications for palliative embolization are “steal” symptoms (progressive neurological deficits) and chronic venous hypertension as a result of high-flow lesions with limited drainage capacity.42,58,59 Other indications can be associated aneurysms and large inoperable AVMs with inherent angiographic “risk factors” that could be addressed endovascularly. Patients with “inoperable” AVMs sometimes undergo palliative embolization after they have already experienced multiple hemorrhages to reduce the flow in an attempt to lower the risk for further rupture. Thus far, however, there is no evidence to support this empirical method. Other indications are intractable headaches and seizures.
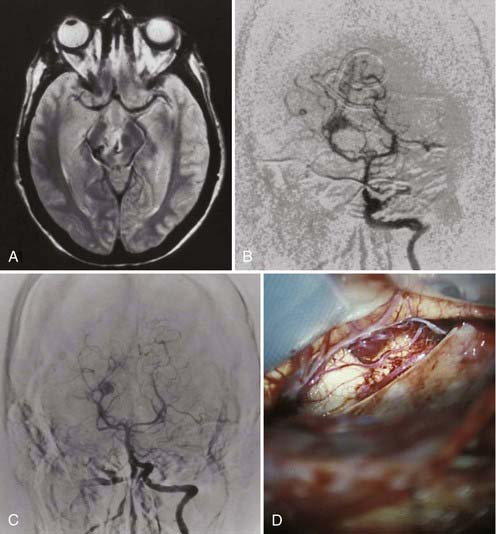
Probably the most important role for embolization is preoperative reduction of arterial inflow, occlusion of deep feeders (Fig. 388-1), and management of prenidal aneurysms. The goal is to enable or facilitate surgery. In our opinion, preoperative embolization should only be performed when in the estimate of an experienced neurosurgeon and endovascular surgeon it will reduce the overall risk—in other words, when it can be estimated that the risk of preoperative embolization added to the risk of surgical excision after embolization would be less than the risk of surgical excision without embolization. Additionally, preoperative embolization should be guided by the surgeon who intends to excise the AVM and knows which feeding pedicles are easy to access at surgery and therefore need not be embolized and which are the deep inaccessible feeders that would be helpful to have occluded by embolization before surgery.
Radiosurgery
Radiosurgery was developed and to date is mostly applied by neurosurgeons trained in this treatment modality. Its impact on the treatment of AVMs has been huge. In all its different techniques it is still most effective for smaller AVMs that are 3 cm or less in diameter. Obviously, the great advantages of radiosurgery are its minimal invasiveness and its potential to treat lesions that would otherwise not be amenable to surgical excision because of deep or critical location, or both.60–64 Other indications are patients who as a result of age and general condition are not considered good candidates for surgery. Achievable obliteration rates are related to size and configuration (diffuse versus compact) and range between 60% and 80% over a 2- to 3-year period (Table 388-E3)![]() .44,55,65–73 Obliteration rates are clearly higher for smaller AVMs.70
.44,55,65–73 Obliteration rates are clearly higher for smaller AVMs.70
TABLE 388-E3 Outcomes of Arteriovenous Malformations after Radiosurgery
| SERIES | OBLITERATION RATES | MORBIDITY/MORTALITY RATES |
|---|---|---|
| Steinberg et al.,65 1991 | 100% (<4 cc); 70% (>3.7 cm in diameter) by angiography | 0.9% permanent morbidity; 11% hemorrhage |
| Friedman,66 1997* | 79% (<10 cc); 47% (>10 cc) | N/A |
| Pollock et al.,55 1998 | 61% overall; 83% (<4 cc) by angiography | N/A |
| Nozaki et al.,44 2000 | N/A | 36% morbidity from rehemorrhage; 0% mortality |
| Stieg et al.,67 2000* | 76% at 3 yr | N/A |
| Inoue and Ohye,68 2002 | 81.3% by angiography | N/A |
| Pollock et al.,69 2003 | 78.0% by angiography | 11.9% morbidity; 4.2% mortality |
| Shin et al.,70 2004 | 87.1% by angiography | 1.9% annual hemorrhage rate; 1.5% permanent morbidity |
| Pollock et al.,71 2004† | 75.4% by angiography; 22.8% by MRI | 12% morbidity from hemorrhage; 17.6% morbidity from radiation; 9% mortality |
| Maruyama et al.,72 2004 | 66% by angiography | 1.7% latency-interval hemorrhage rate per year for 1st 3 yr, then 0% |
| Fractionated Radiation Therapy for Large Arteriovenous Malformations | ||
| Karlsson et al.,73 2005 | 8% obliterated | 6% annual hemorrhage rate after radiation; 18% died of hemorrhage; overall 25% mortality (7 deaths, 2 of unknown cause) |
N/A, not available.
* It is not clear that the obliteration rates in these two articles were confirmed by angiography or magnetic resonance imaging and angiography.
† A series of grade IIIB and IV arteriovenous malformations (AVMs) in the basal ganglia, thalamus, and brainstem. Grade IIIB is defined as small Spetzler-Martin grade III AVMs located in areas where surgical resection is either too difficult or prohibitive.20
The main disadvantage of radiosurgery is its uncertainty of cure in terms of complete AVM obliteration and the fact that this process takes between 1 and 3 and sometimes 4 years to occur.61,74–76 It has been shown that during this “latent period” of incomplete obliteration the risk for hemorrhage is similar to the risk with untreated AVMs (3% to 4% per year),77–81 although there are also reports claiming lower rates72,80,82 and some series suggest that it may be higher (5.3% to 10%).83,84 In addition, there is a small but significant risk for neurological injury and other types of complications from radiation damage (3% to 10%, depending on location).84–87 There are a few reports of late hemorrhage after angiographically proven complete obliteration, and some reports describe histologically proven patency of some of the components of the AVM after angiographic obliteration. However, in view of the large number of AVMs treated by radiosurgery within the past 20 years, it can be concluded that the risk for future hemorrhage is extremely low and practically negligible once angiographic obliteration has been demonstrated. Clinically significant complications that can be directly attributed to radiosurgery, such as symptomatic radiation necrosis, appear in 3% to 6% of treated patients ![]() (Table388-E3). The morbidity of treating AVMs in very critical regions such as the brainstem and internal capsule is most likely substantially higher.71
(Table388-E3). The morbidity of treating AVMs in very critical regions such as the brainstem and internal capsule is most likely substantially higher.71
Coexisting Intracranial Aneurysms
Thompson, Deruty, and their associates found coexisting intracranial aneurysms in 45 of 600 patients with AVMs (7.5%),88,89 and Cunha e Sa and colleagues’ analysis of 400 AVMs identified aneurysms in 39 patients (10%).90 They can be proximal to the lesion (prenidal on a feeding artery), within the lesion (intranidal), or in a remote location. The risk of these patients having an intracranial hemorrhage is higher than the risk in patients without associated aneurysms and is estimated at 7% per year91; another study found a higher rebleeding rate in AVMs associated with intranidal aneurysms.92 Patients with coexisting aneurysms tend to be older and are more frequently initially evaluated for epilepsy and neurological events.93 Half of these patients have multiple aneurysms. The majority (85%) of aneurysms are prenidal on a feeding artery or on a major arterial trunk that is participating in the supply. If the aneurysms are postnidal and remote from the AVM, their risk for rupture seems to be lower. In consideration of these numbers, we think that aneurysms need to be addressed during the initial evaluation and early in care if the location of the hemorrhage suggests aneurysmal rupture. Some authors also recommend initial treatment of the aneurysm, even when the AVM has bled, or in cases in which the source is not clear.
It has been proposed that hemodynamic stress from high flow or high arteriovenous shunting in feeding arteries is an important factor for the development of coexisting aneurysms; however, they can also develop in low-flow shunt situations. In high-flow situations, coexisting prenidal aneurysms may diminish in size and disappear when the shunt is obliterated.93
General Principles of Microsurgical Treatment
Circumferential Dissection of the Nidus
In AVMs with a surface representation, the nidus is “developed” by circumferential corticectomy around the AVM after all superficial feeders have been identified and disconnected by opening the adjacent sulci as a first step. It is very helpful to carry this perpendicular dissection and corticectomy around the nidus to a depth of 2.5 to 3 cm before proceeding with deeper dissection. We have found empirically that a perpendicular corticectomy to such a depth will have disconnected all the superficial arterial supply. Once this has been accomplished, the dissection is continued in a more vertical plane around the AVM, usually in a “spiral” fashion until its deepest aspect is reached. During this deeper, spiraling dissection phase it is very important to not mistake loops and eccentric lobules of the AVM that project into normal brain for feeding arteries or draining veins. Coagulation and interruption of such loops can lead to significant and hard-to-control bleeding from the AVM. Great care should be taken to avoid coagulation of the AVM itself during the early phase, when internal AVM pressure and flow are still very high, because any decrease in nidus volume without a decrease in inflow will increase internal pressure and thus the risk for rupture. After the major arterial feeding pedicles have been taken, the turgor of the nidus will substantially diminish. Only then can coagulation be used to stroke some of the AVM loops in an effort to shrink them away from eloquent brain areas. It is not advisable at all to use such a maneuver in the early portion of the procedure when the AVM is still under high inflow pressure and thus more prone to rupture with manipulation.94 As the dissection approaches deeper areas, it usually becomes more tedious because more persistent bleeding will frequently be encountered as a result of the smaller and more fragile arterial supply from deep perforators or subependymal choroidal branches as a ventricle is approached. Once inadvertently lacerated or cut, bleeding from these deep vessels is extremely difficult to control because the vessel diameter is small and there is an element of retraction into parenchyma once cut. The late Dr. Thoralf Sundt designed microclips specifically for placement on these tiny fragile vessels. Sundt microclips are a far better option than creating an ever-deeper tunnel into normal brain in an attempt to reach the retracted and still patent bleeder with bipolar coagulation. Bleeding from the AVM itself can usually be stopped by placing a cottonoid pad, gently retracting on the bleeding point, and moving to a different plane of dissection. If one uses such a maneuver, it is important to remember to never pack when bleeding in a direction away from the AVM because this will risk significant parenchymal or intraventricular hemorrhage. In the case of AVMs that reach the ventricle, oftentimes the bleeding will not stop until the ependyma of the ventricle has been reached and small ependymal feeders to the AVM are controlled.
Transection of Deep Venous Drainage and Removal of the Lesion
If an important draining vein prematurely bleeds, it is usually advisable to attempt hemostasis with hemostatic agents and gentle pressure instead of coagulation and sacrifice. When the major arterial pedicles have been disconnected, the color of the large draining veins should become darker and finally change from red to blue because the drained blood will be progressively less arterialized. After inflow to the nidus has been completely eliminated, the nidus should deflate. The draining veins can be taken at that point unless they still have an arterialized element, which is frequently the case with the last large draining vein. Quite often there is one hidden arterial feeder left, in close proximity or underneath the vein, that needs to be identified and disconnected. After this final step the nidus can be removed. Complete resection of the malformation should be confirmed by intraoperative angiography.95,96 Promising new techniques are intraoperative ICG angiography (near-infrared indocyanine green fluorescence angiography),97–101 intraoperative computed tomographic (CT) angiography,102 and three-dimensional intraoperative ultrasound.103,104 It can be foreseen that the intraoperative application of magnetic resonance imaging (MRI) will not remain limited to tumor surgery.105,106