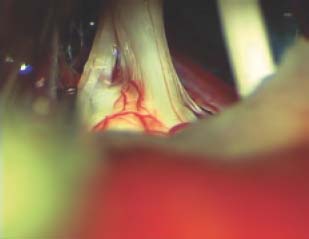
14 The scope of lateral skull base surgery includes surgical treatment of the group of disorders known as microvascular compression syndromes. The most common of these, and the first attributed to the pathology of microvascular compression, is trigeminal neuralgia. Trigeminal neuralgia is marked by unilateral, severe, lancinating facial pain elicited by triggering and (at least partially) relieved by carbamazepine. Each of the other microvascular compression syndromes is defined by a characteristic clinical presentation and is associated with a specific cranial nerve. These are trigeminal neuralgia (trigeminal nerve), hemifacial spasm (facial nerve), geniculate neuralgia (nervus intermedius), severe positional vertigo (vestibular nerve), and glossopharyngeal neuralgia (glossopharyngeal nerve). An understanding of the clinical presentation of each of these disorders is critical to successful treatment. Also critical to the treatment of these patients is an understanding that treatment is always done for quality-of-life reasons. None of these disorders is directly life-threatening, and invasive treatment should be undertaken only after all medical options have been exhausted and only if symptomatology is severe. On the other hand, patients can be assured that, in proper hands, these disorders can safely and effectively be treated with surgery if it becomes necessary. Microvascular decompression (MVD) of each of these nerves is performed via retrosigmoid craniotomy. The goal is isolation of the affected cranial nerve from any compressing vascular structures. Although magnetic resonance imaging (MRI) is useful in confirming the presence of microvascular compression, it should be remembered that each vascular compression syndrome is a clinical diagnosis. We obtain a preoperative MRI for every patient. The main reason to obtain an MRI is to exclude the possibility of tumor or other compressive lesion such as arachnoid cyst and to ensure that there is no intrinsic brain abnormality, such as brainstem infarct or multiple sclerosis plaque, which would contraindicate microvascular decompression. MR sequences such as constructive interference in steady state (CISS) and fast imaging employing steady state acquisition (FIESTA) show the anatomy of the cranial nerves with very good detail. However, the surgeon should not be dissuaded from performing MVD in the absence of MR evidence of microvascular compression if there is a compelling clinical picture. Microvascular decompression is performed via a retrosigmoid craniotomy. We utilize continuous facial nerve and auditory brainstem response monitoring for all cases. The patient is positioned supine on the operating table and is carefully secured to allow rotation of the table. The table may be flexed slightly to create a “lounge” position, which lowers the shoulder in relation to the operative line-of-sight. A Mayfield headholder is used with the head rotated 45 to 60 degrees contralaterally. The neck is flexed slightly, although care is taken with positioning to avoid overrotation and interference with venous return in the neck. For trigeminal nerve decompression, the vertex is dropped to allow for less hindered access below the tentorium. For decompression of the other nerves, a relatively neutral position can be utilized in this access. The surgeon sits behind the patient’s head. As the craniotomy must be oriented to the transverse-sigmoid junction, an appreciation of the courses of these structures is critical. The transverse sinus runs at the level of the zygomatic arch (the Frankfurt line), and the sigmoid sinus is located at the posterior edge of the mastoid. Of course, there is significant individual variation. A curved incision is utilized with its apex approximately three fingerbreadths behind the ear. The scalp is elevated forward toward the mastoid, and an inverted L-shaped incision is made in the periosteum with limbs over the mastoid process and the inferior temporal line. The suboccipital muscles are elevated in a separate flap inferiorly and medially. We prefer this overlapping flap technique rather than a smaller linear incision to prevent postoperative cerebrospinal fluid (CSF) leak. A small craniotomy is performed in the retrosigmoid area using cutting and diamond burs. This craniotomy must include exposure of the posterior edge of the sigmoid sinus and the inferior edge of the transverse sinus. Proper placement is facilitated by entry into the mastoid air cells, via which the inner table over the sigmoid sinus can be directly visualized. It is often necessary to drill away a significant amount of bone to properly orient the bone flap and to expose and control any emissary veins. Being left with a tiny bone flap, or performing a craniectomy, is preferable to lacerating the sinus or to having an improperly positioned bony opening. We aim for a craniotomy approximately 2.5 cm along the sigmoid sinus and 2 cm along the transvers sinus. After removal of bone, any exposed mastoid air cells are occluded with bone wax. Opening of the dura must be performed with great care. The dura is nicked with a scalpel and CSF is drained from over the cerebellar hemisphere. If no CSF is obtained, the patient may be placed in the reverse Trendelenburg position. Also, the attainment of adequate diuresis and hyperventilation should be confirmed with the anesthesiologist. An inferior dural opening is performed, extending to the lower edge of the exposed sigmoid sinus. After a cottonoid is advanced over the cerebellum adjacent to the inferior aspect of the petrous bone, the cerebellum is gently retracted using a microsucker. The working space in this area can be very narrow, and great care is taken to advance to the arachnoid overlying the cisterna magna. The cistern is opened using a sharp micro–nerve hook, and with the egress of CSF, brain relaxation is achieved. The dural opening is completed only after the brain is relaxed to prevent herniation. Care is taken to ensure that the opening is brought to the edge of the sigmoid sinus, as every extra millimeter of exposure paralleling the sigmoid is critical. For microvascular decompression of all nerves, wide arachnoid dissection is critical. It is important not only for direct exposure of the involved nerve, but also for mobilization of the cerebellar hemisphere. With adequate dissection, only minimal cerebellar retraction, if any, is necessary to visualize both nerves and the brainstem surface. For trigeminal nerve decompression, sacrifice of one or more bridging petrosal veins may be required. For other nerves, this is generally unnecessary. Small arteries bridging to the petrous bone may be sacrificed only if they are laterally placed and clearly distant from the neural foramina. These are usually small loops that become adherent to the dura and supply blood to the lateral aspects of the cerebellum. If retraction is needed, a narrow retractor blade with a bluntly hooked end is ideal. Using this type of retractor, cerebellar hemisphere, flocculus, or even choroids plexus can be gently raised instead of being pushed, thus maximizing exposure while minimizing pressure on the cerebellum and traction on the cranial nerves. Decompression is performed using Teflon felt (Bard PV, Inc., Tempe, AZ), which is teased to create small, fluffy pieces. Individual teased pieces are rolled to form pledgets of various sizes, approximating the size and shape of grains of rice. Pledgets are not used without proper hydration. Dissection and pledget insertion is performed with minimal retraction, using preferably only a manually held suction, and, if necessary, self-retaining retractors. If any changes occur with auditory brainstem response (ABR) monitoring, retraction is immediately released. After completion of the MVD, closure is performed. If the patient has been placed in the reverse Trendelenburg position, the head is lowered to a neutral position. The cranial nerves are reinspected after irrigation to ensure that they remain well decompressed. We make no attempt to close the dura in a watertight manner. Instead, the dura is tacked together and overlaid with an onlay collagenous matrix graft. Cranioplasty is always performed, as previously described for the retrosigmoid route, and the wound is closed in layers. We utilize the operating microscope as the primary aid to visualization for MVDs. Endoscopic assistance, however, is very beneficial as an adjunct. The chief advantages of the endoscopic viewpoint are the ability to view at an angle and a wider field of view. These advantages are of particular utility for MVD. The major disadvantage is visual acuity, which is, at this point, clearly inferior to the microscope. In contrast to tumor dissection, however, modern endoscopes and video equipment are in practice generally adequate for these procedures. Before attempting any dissection under endoscopic guidance, the surgeon should become comfortable simply observing the operative field using first a 0-degree endoscope and then a 30-degree angled endoscope. Only after gaining full comfort with these endoscopes should the surgeon begin to utilize a fixed endoscope holder and then carry out any dissection (bimanually). We would not recommend the use of endoscopes angled greater than 30 degrees, as they are generally more awkward to advance in the cerebellopontine angle. For microvascular decompression of the trigeminal nerve, it is useful to tilt the vertex downward to facilitate exposure directly along the tentorium. Although the eventual goal is superior, initial dissection to open the basal cisterns is performed inferiorly along the petrous ridge. An arachnoid opening is performed over the lower cranial nerves and the cranial nerve VII–VIII complex prior to advancing toward the trigeminal nerve. All bridging petrosal veins are coagulated and divided, and the superior portion of the cerebellar hemisphere is gently retracted. It is necessary to visualize the entire length of the trigeminal nerve from the pons to the entrance to Meckel’s cave (Fig. 14.1). The motor branch is seen just medial to the main trunk of the nerve. Microvascular compression can be the result of either arterial or venous structures, and there are often multiple vessels seen. The nerve must be isolated from all vascular structures. When compression is the result of veins only, an arterial loop can typically be seen compressing the vein, which transmits arterial pulsations to the nerve. All arterial loops are carefully dissected away from the nerve using microinstruments. Microspatulas and very fine but blunt micro–nerve hooks are useful. Veins can be coagulated and divided with care. It is critical to be certain that there is no compression from medial or superior to the nerve, as these orientations will be less obvious than the opposing directions. Special attention must be paid to the most proximal portion of the nerve near the brainstem, and the adjacent root entry zone must be isolated from vascular compression as well (Fig. 14.2). The area of the nerve more distal in the cistern, while most easily visualized, is also the most difficult to adequately decompress. Although arterial loops in this region can be dissected free of the nerve, veins must be handled delicately. It is occasionally impossible to safely separate veins from the nerve in this region. These veins can be coagulated and divided well away from the nerve. This will effectively devitalize the veins as they pass the nerve. Care must be taken to ensure not only that the nerve is isolated from any arterial loops but also that any venous fragments are as well. Fig. 14.1 Microsurgical exposure of the trigeminal nerve for MVD (right side). The nerve must be visualized over its entire length, including the root entry zone. The motor branch is seen superoanterior to the main sensory nerve.
Microvascular Cranial Nerve Decompression
 Surgical Technique
Surgical Technique
Trigeminal Nerve
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Neupsy Key
Fastest Neupsy Insight Engine
