11
Movement Disorders
Movement disorders (sometimes called extrapyramidal disorders) impair the regulation of voluntary motor activity without directly affecting strength, sensation, or cerebellar function. They include hyperkinetic disorders associated with abnormal, involuntary movements and hypokinetic disorders characterized by poverty of movement. Movement disorders result from dysfunction of deep subcortical gray matter structures termed the basal ganglia. Although there is no universally accepted anatomic definition of the basal ganglia, for clinical purposes they may be considered to comprise the caudate nucleus, putamen, globus pallidus (GPi), subthalamic nucleus, and substantia nigra. The putamen and the globus pallidus are collectively termed the lentiform nucleus; the combination of lentiform nucleus and caudate nucleus is designated the corpus striatum.
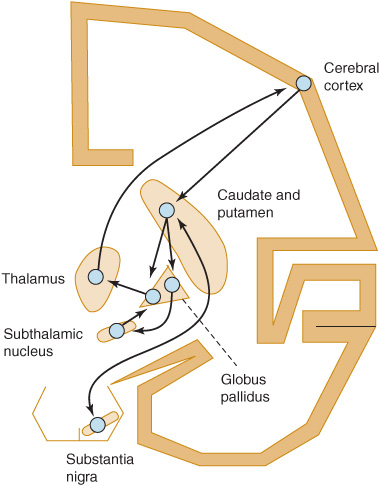
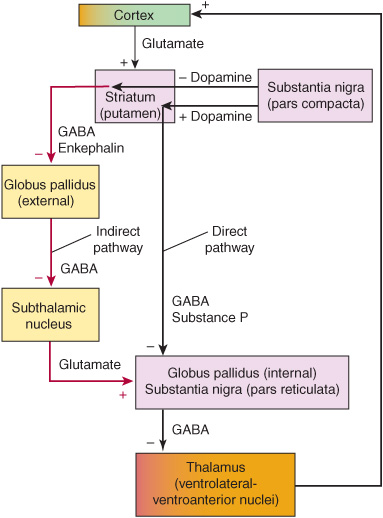
The basic circuitry of the basal ganglia consists of three interacting neuronal loops (Figure 11-1). The first is a corticocortical loop that passes from the cerebral cortex, through the caudate and putamen, the internal segment of the globus pallidus, and the thalamus and then back to the cerebral cortex. The second is a nigrostriatal loop connecting the substantia nigra with the caudate and putamen. The third, a striatopallidal loop, projects from the caudate and putamen to the external segment of the globus pallidus, then to the subthalamic nucleus, and finally to the internal segment of the globus pallidus. In some movement disorders (eg, Parkinson disease), a discrete site of pathology within these pathways can be identified; in other cases (eg, essential tremor), the precise anatomic abnormality is unknown.
Figure 11-1. Basic neuronal circuitry of the basal ganglia.
TYPES OF ABNORMAL MOVEMENTS
Categorizing an abnormal movement is generally the first step toward arriving at the neurologic diagnosis. Abnormal movements can be classified as tremor, chorea, athetosis or dystonia, ballismus, myoclonus, or tics. Such movements can arise in a variety of contexts, such as in degenerative disorders or with structural lesions. In many disorders, abnormal movements are the sole clinical features.
TREMOR
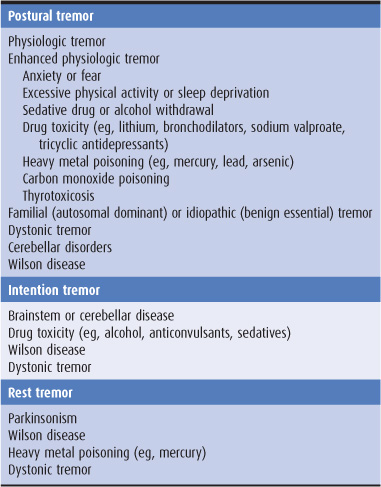
A tremor is a rhythmic oscillatory movement best characterized by its relationship to voluntary motor activity, that is, according to whether it occurs at rest, during maintenance of a particular posture, or during movement. The major causes of tremor are listed in Table 11-1. Tremor is enhanced by emotional stress and disappears during sleep. Tremor that occurs when the limb is at rest is generally referred to as static tremor or rest tremor. If present during sustained posture, it is called a postural tremor; although this tremor may continue during movement, movement does not increase its severity. When present during movement but not at rest, it is generally called an intention or kinetic tremor. Both postural and intention tremors are also called action tremors.
Table 11-1. Causes of tremor.
POSTURAL TREMOR
Physiologic Tremor
An 8- to 12-Hz tremor of the outstretched hands is a normal finding. Its physiologic basis is uncertain.
Enhanced Physiologic Tremor
Physiologic tremor may be enhanced by fear or anxiety. A more conspicuous postural tremor may also be found after excessive physical activity or sleep deprivation. It can complicate treatment with certain drugs (notably lithium, tricyclic antidepressants, sodium valproate, and bronchodilators) and is often conspicuous in patients with alcoholism or in alcohol or drug withdrawal states. It is common in thyrotoxicosis, and it can also result from poisoning with a number of substances, including mercury, lead, arsenic, and carbon monoxide. There is no specific medical therapy.
Other Causes
The most common type of abnormal postural tremor is benign essential tremor, which often has a familial basis. Postural tremor may also be conspicuous in patients with Wilson disease or cerebellar disorders. A postural tremor of the hands that is indistinguishable from essential tremor may occur in patients with dystonia. The designation dystonic tremor refers to a postural or intention tremor that occurs in a part of the body already affected by dystonia. It is a part of the dystonia and is most prominent when an attempt is made to oppose the dystonic posturing.
ASTERIXIS
Asterixis may be associated with postural tremor, but is itself more properly considered a form of myoclonus (see later discussion) than of tremor. It is seen most commonly in patients with metabolic encephalopathy such as occurs with hepatic or renal failure.
To detect asterixis, the examiner asks the patient to hold the arms outstretched with fingers and wrists extended. Episodic cessation of muscular activity causes sudden flexion at the wrists followed by a return to extension, so that the hands flap in a regular or, more often, an irregular rhythm. A similar phenomenon may be demonstrable at the ankles. The asterixis resolves with clearing of the metabolic encephalopathy.
INTENTION (KINETIC) TREMOR
Intention or kinetic tremor occurs during activity. If the patient is asked to touch his or her nose with a finger, for example, the arm exhibits tremor during movement, often more marked as the target is reached. This form of tremor is sometimes mistaken for limb ataxia, but the latter has no rhythmic oscillatory component.
Intention tremor results from a lesion affecting the superior cerebellar peduncle. Because it is often very coarse, it can lead to severe functional disability. No satisfactory medical treatment exists, but stereotactic surgery of the contralateral ventrolateral nucleus of the thalamus or high-frequency thalamic stimulation through an implanted device is sometimes helpful when patients are severely incapacitated.
Intention tremor can also occur—together with other signs of cerebellar involvement—as a manifestation of toxicity of certain sedative or anticonvulsant drugs (eg, phenytoin) or alcohol; it is seen in patients with Wilson disease.
REST TREMOR
Parkinsonism
Rest tremor usually has a frequency of 4 to 6 Hz and is characteristic of parkinsonism whether the disorder is idiopathic or secondary (ie, postencephalitic, toxic, or drug-induced in origin). The rate of the tremor, its relationship to activity, and the presence of rigidity or hypokinesia usually distinguish the tremor of parkinsonism from other forms of tremor. Tremor in the hands may appear as a “pill-rolling” maneuver—rhythmic, opposing circular movements of the thumb and index finger. There may be alternating flexion and extension of the fingers or hand or alternating pronation and supination of the forearm; in the feet, rhythmic alternating flexion and extension are common. Parkinsonism is discussed in more detail later.
Other Causes
Less common causes of rest tremor include Wilson disease and poisoning with heavy metals such as mercury.
CHOREA
The word chorea denotes rapid irregular muscle jerks that occur involuntarily and unpredictably in different parts of the body. In florid cases, the often forceful involuntary movements of the limbs and head and the accompanying facial grimacing and tongue movements are unmistakable. Voluntary movements may be distorted by the superimposed involuntary ones. In mild cases, however, patients may exhibit no more than a persistent restlessness and clumsiness. Power is generally full, but there may be difficulty in maintaining muscular contraction such that, for example, handgrip is relaxed intermittently (milkmaid grasp). The gait becomes irregular and unsteady, with the patient suddenly dipping or lurching to one side or the other (dancing gait). Speech often becomes irregular in volume and tempo and may be explosive in character. In some patients, athetotic movements or dystonic posturing (see later) may also be prominent. Chorea disappears during sleep.
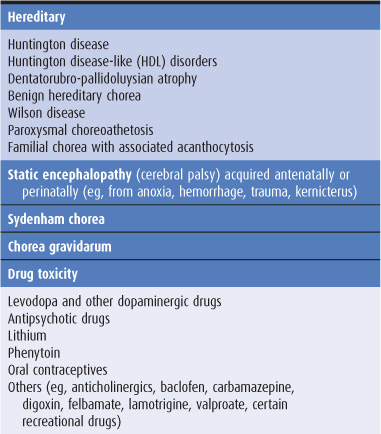
The pathologic basis of chorea is unclear, but in some cases it is associated with cell loss in the caudate nucleus and putamen, and it can be provoked by dopaminergic agonist drugs. Causes of chorea are shown in Table 11-2 and are discussed later in this chapter. When chorea is due to a treatable medical disorder, such as polycythemia vera or thyrotoxicosis, adequate treatment of the primary disorder abolishes the dyskinesia.
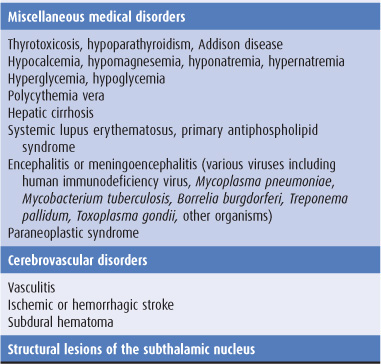
Table 11-2. Causes of chorea.
HEMIBALLISMUS
Hemiballismus is unilateral chorea that is especially violent because the proximal muscles of the limbs are involved. It is due most often to vascular disease in the contralateral subthalamic nucleus and commonly resolves spontaneously in the weeks after its onset. It is sometimes due to other types of structural disease; in the past, it was an occasional complication of thalamotomy. Pharmacologic treatment is similar to that for chorea (discussed later).
DYSTONIA & ATHETOSIS
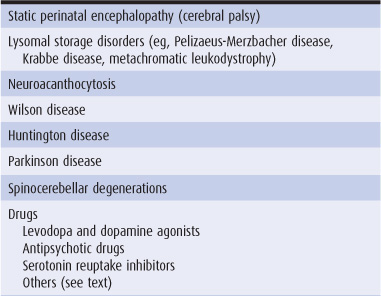
The term athetosis generally denotes abnormal movements that are slow, sinuous, and writhing in character. When the movements are so sustained that they are better regarded as abnormal postures, the term dystonia is used, and many now use the terms interchangeably. In dystonia, excessive or inappropriate contraction of muscles (often agonists and antagonists) leads to sustained abnormal postures of the affected region of the body. The abnormal movements and postures may be generalized or restricted in distribution, such as to the neck (torticollis), hand and forearm (writer’s cramp), or mouth (oromandibular dystonia). With restricted dystonias, one or more of the limbs may be affected (segmental dystonia), or the disturbance may be restricted to localized muscle groups (focal dystonia). Generalized dystonia may be idiopathic or secondary (Table 11-3).
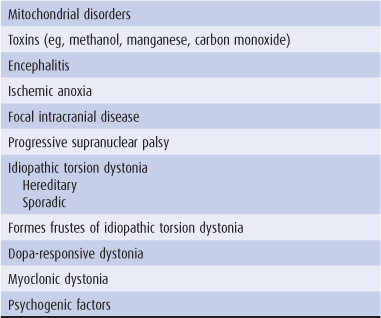
Table 11-3. Causes of dystonia and athetosis.
FACTORS INFLUENCING DYSTONIA
The abnormal movements are not present during sleep. They are generally enhanced by emotional stress and by voluntary activity. In some cases, abnormal movements or postures occur only during voluntary activity and sometimes only during specific activities such as writing, speaking, or chewing.
ETIOLOGY
Table 11-3 lists some of the conditions in which these movement disorders are encountered. Perinatal anoxia, birth trauma, and kernicterus from hyperbilirubinemia are the most common causes. In these circumstances, abnormal movements usually develop before the age of 5 years. Careful questioning usually discloses a history of abnormal early development and often of seizures. Examination may reveal signs of cognitive dysfunction or a pyramidal deficit in addition to the movement disorder.
Dystonic movements and postures are the cardinal features of idiopathic torsion dystonia (discussed later). Torsion dystonia may also occur as a manifestation of Wilson disease or Huntington disease or as a sequela of encephalitis.
Acute dystonic posturing may result from treatment with dopamine receptor antagonist drugs (discussed later).
Lateralized dystonia may occasionally relate to focal intracranial disease, but the clinical context in which it occurs usually identifies the underlying cause.
MYOCLONUS
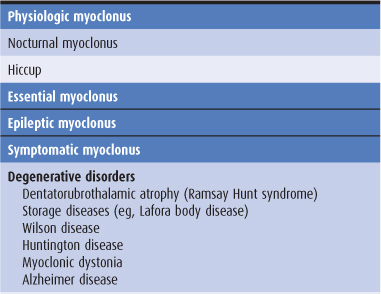
Myoclonic jerks are sudden, rapid, twitchlike muscle contractions. They can be classified according to their distribution, relationship to precipitating stimuli, or etiology. Generalized myoclonus has a widespread distribution, whereas focal or segmental myoclonus is restricted to a particular part of the body. Myoclonus can be spontaneous, or it can be brought on by sensory stimulation, arousal, or the initiation of movement (action myoclonus). Myoclonus may occur as a normal phenomenon (physiologic myoclonus) in healthy persons, as an isolated abnormality (essential myoclonus), or as a manifestation of epilepsy (epileptic myoclonus). It can also occur as a feature of a variety of degenerative, infectious, and metabolic disorders (symptomatic myoclonus) affecting the cerebral cortex, brainstem, or spinal cord. Myoclonus is sometimes manifest not by a sudden twitchlike muscle contraction but by a sudden loss of muscle activity (negative myoclonus). This is best seen in asterixis: The patient tries to maintain the wrists hyperextended in the outstretched arms, but the sudden intermittent transient relaxation of the extensor muscles followed by return of the contraction leads to a “flapping tremor.”
GENERALIZED MYOCLONUS
The causes of generalized myoclonus are summarized in Table 11-4. Physiologic myoclonus includes the myoclonus that occurs upon falling asleep or awakening (nocturnal myoclonus), as well as hiccup. Essential myoclonus is a benign condition that occurs in the absence of other neurologic abnormalities and is sometimes inherited. Epileptic myoclonus may be impossible to differentiate clinically from nonepileptic forms. It may be possible to distinguish the two types electrophysiologically, however, by the duration of the electromyographic burst associated with the jerking, by demonstrating an electroencephalographic (EEG) correlate with a consistent temporal relationship to the jerks, or by determining whether muscles involved in the same jerk are activated synchronously.

Table 11-4. Causes of generalized myoclonus.
SEGMENTAL MYOCLONUS
Segmental myoclonus can arise from lesions affecting the cerebral cortex, brainstem, or spinal cord. For example, involvement of the dentatorubroolivary pathway by stroke, multiple sclerosis, tumors, or other disorders can produce palatal myoclonus, which may be associated with an audible click or synchronous movements of ocular, facial, or other bulbar muscles. Rhythmic vertical oscillation of the soft palate occurs that is best regarded as a tremor. Segmental myoclonus can result from many of the same disturbances that produce symptomatic generalized myoclonus (Table 11-4). Metabolic disorders such as hyperosmolar nonketotic hyperglycemia can cause epilepsia partialis continua, in which a repetitive focal epileptic discharge occurs from the contralateral sensorimotor cortex and leads to segmental myoclonus. Segmental myoclonus is usually unaffected by external stimuli and persists during sleep.
PROPRIOSPINAL MYOCLONUS
Propriospinal myoclonus arises in the spinal cord and then spreads up and down the cord, leading to a brief bodily contraction. Its myoclonic basis is often not recognized. Electromyographic surface recordings may be necessary to show the spread of muscle activity in an orderly sequence and may help to localize the site of origin of the myoclonus. The underlying spinal lesion may be revealed by imaging in some cases.
TREATMENT
Although myoclonus can be difficult to treat, it sometimes responds to anticonvulsant drugs such as valproic acid 250 to 500 mg orally three times daily or to benzodiazepines such as clonazepam 0.5 mg orally three times daily, gradually increased to as much as 12 mg/d. Postanoxic action myoclonus has been found to be remarkably responsive to 5-hydroxytryptophan, the metabolic precursor of the neurotransmitter 5-hydroxytryptamine (serotonin). The dosage of 5-hydroxytryptophan is increased gradually to a maximum of 1 to 1.5 mg/d orally and may be combined with carbidopa (maximum, 400 mg/d orally) to inhibit metabolism in peripheral tissues.
TICS
Tics are sudden, recurrent, quick, coordinated abnormal movements that can usually be imitated without difficulty. The same movement occurs again and again and can be suppressed voluntarily for short periods, although doing so may cause anxiety. Tics tend to worsen with stress, diminish during voluntary activity or mental concentration, and disappear during sleep.
CLASSIFICATION
Tics can be classified into four groups depending on whether they are simple or multiple and transient or chronic.
1. Transient simple tics are very common in children, usually terminate spontaneously within 1 year (often within a few weeks), and generally require no treatment.
3. Persistent simple or multiple tics of childhood or adolescence generally begin before the age of 15 years. There may be single or multiple motor tics, and often vocal tics, but complete remission occurs by the end of adolescence.
4. The syndrome of chronic multiple motor and vocal tics is generally referred to as Gilles de la Tourette syndrome, after the French physician who was one of the first to describe its clinical features. It is discussed in detail later. Tics also may occur with levodopa or amphetamine use and after chronic neuroleptic use (tardive tic), after head trauma or viral encephalitis, and in autistic children. They can occur in association with degenerative disorders of the basal ganglia, such as Huntington disease, and are well described in neuroacanthocytosis, when they may have a self-mutilating character.
BRADYKINESIA & HYPOKINESIA
Bradykinesia (slowed movement) and hypokinesia or akinesia (poverty or lack of movement) are major features of parkinsonism and may be more disabling than the presence of abnormal movements. Manifestations include a fixity of facial expression (the so-called masked facies, with reduced blinking, widened palpebral fissures, and an apparently impassive appearance) and a paucity of spontaneous movement of the limbs (eg, a reduced arm swing on walking). Some patients have “freezing,” by which is meant a difficulty in starting or maintaining a rhythmic repetitive activity such as walking. Such symptoms are difficult for patients to describe; they are often erroneously attributed to weakness.
These phenomena are tested clinically by, for example, asking the patient to make repetitive alternating movements of each extremity in turn. This can involve the repetitive tapping of the thumb on the terminal interphalangeal joint of the index or third finger or on the pad of these digits, repetitive pronation and supination of the raised arm (as if screwing a light bulb into the ceiling), repetitive opening and closing of the fist of each hand, and repetitive tapping the foot on the floor while the heel is maintained on the ground. A progressive reduction in amplitude or speed of the movements, irregularity in rhythm, or arrests in movement are indicative of abnormality. Activity should be continued until at least 15 repetitions have occurred, and sometimes for longer. It is important to distinguish any abnormalities from the slowness of movement without fatiguing and decrement that may occur in patients with pyramidal or cerebellar dysfunction (often with an irregular rhythm in the latter context). The inexpressive face of depressed patients may simulate the masked facies of parkinsonism and should be distinguished by the lack of other extrapyramidal findings and the abnormal affect.
CLINICAL EVALUATION OF PATIENTS
HISTORY
AGE AT ONSET
The age at onset of a movement disorder may suggest the underlying cause. For example, onset in infancy or early childhood suggests birth trauma, kernicterus, cerebral anoxia, or an inherited disorder; abnormal facial movements developing in childhood are more likely to represent tics than involuntary movements of another sort; and tremor presenting in early adult life is more likely to be of the benign essential variety than due to Parkinson disease.
The age at onset can also influence the prognosis. In idiopathic torsion dystonia, for example, progression to severe disability is much more common when symptoms develop in childhood than when they develop in later life. Conversely, tardive dyskinesia is more likely to be permanent and irreversible when it develops in the elderly than when it develops in the adolescent years.
MODE OF ONSET
Abrupt onset of dystonic posturing in a child or young adult should raise the possibility of a drug-induced reaction; a more gradual onset of dystonic movements and postures in an adolescent suggests the possibility of a chronic disorder such as idiopathic torsion dystonia or Wilson disease. Similarly, the abrupt onset of severe chorea or ballismus suggests a vascular cause, and abrupt onset of severe parkinsonism suggests a neurotoxic cause; more gradual, insidious onset suggests a degenerative process.
COURSE
The manner in which the disorder progresses from its onset may also be helpful diagnostically. For example, Sydenham chorea usually resolves within about 6 months after onset and therefore should not be confused with other varieties of chorea that occur in childhood.
MEDICAL HISTORY
Drug History
It is important to obtain an accurate account of all drugs that have been taken by the patient over the years, because many of the movement disorders are iatrogenic. The phenothiazine and butyrophenone drugs may lead to the development of abnormal movements either while patients are taking them or after their use has been discontinued, and the dyskinesia may be irreversible. These drugs and the dyskinesias associated with their use are discussed later in this chapter.
General Medical History
1. Chorea may be symptomatic of the disease in patients with a history of rheumatic fever, thyroid disease, systemic lupus erythematosus, polycythemia, hypoparathyroidism, or cirrhosis of the liver.
2. Movement disorders, including tremor, chorea, hemiballismus, dystonia, and myoclonus, have been described in patients with acquired immunodeficiency syndrome (AIDS). Opportunistic infections such as cerebral toxoplasmosis appear to be the cause in some cases, but infection with human immunodeficiency virus type 1 (HIV-1) may also have a direct pathogenetic role.
3. A history of birth trauma or perinatal distress may suggest the cause of a movement disorder that develops during childhood.
4. Encephalitis lethargica is no longer encountered clinically; it was epidemic in the 1920s, however, and was often followed by a wide variety of movement disorders, including parkinsonism. It is therefore important to inquire about this disease when elderly patients are being evaluated.
Family History
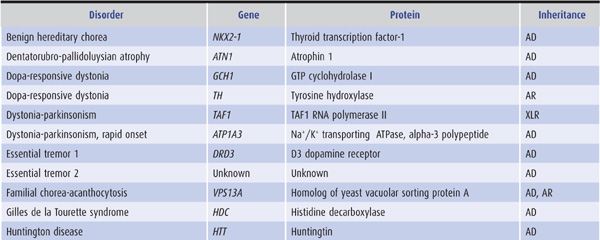
Some movement disorders have an inherited basis (Tables 11-5 and 11-6), and it is essential that a complete family history is obtained, supplemented if possible by personal scrutiny of close relatives. Any possibility of consanguinity should be noted.
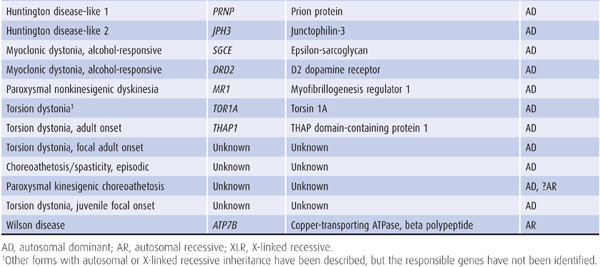
Table 11-5. Selected hereditary nonparkinsonian movement disorders.
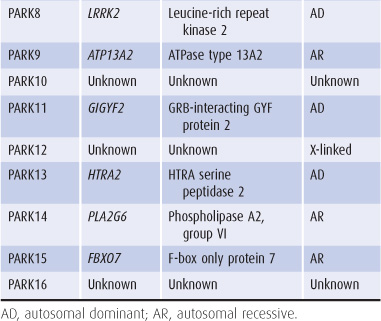
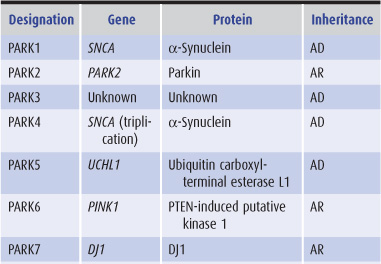
Table 11-6. Genes implicated in hereditary parkinsonism.
EXAMINATION
Clinical examination will indicate the nature of the abnormal movements and motor deficit, the extent of neurologic involvement, and the presence of coexisting disease; these in turn may suggest the diagnosis.
The mental status examination may suggest psychiatric disease, raising the possibility that the abnormal movements are related to the psychiatric disorder or to its treatment with psychoactive drugs—or that the patient has a disorder characterized by both abnormal movements and behavioral disturbances, such as Huntington disease or Wilson disease.
Focal motor or sensory deficits raise the possibility of a structural space-occupying lesion, as does papilledema. Kayser-Fleischer rings suggest Wilson disease. Signs of vascular, hepatic, or metabolic disease may suggest other causes for a movement disorder, such as acquired hepato-cerebral degeneration or vasculitis.
INVESTIGATIVE STUDIES
Several investigations may be of diagnostic help.
BLOOD & URINE TESTS
1. Serum and urine copper and serum ceruloplasmin levels are important in diagnosing Wilson disease.
2. Complete blood count and sedimentation rate are helpful in excluding polycythemia, vasculitis, or systemic lupus erythematosus, any of which can occasionally lead to a movement disorder. A wet film of the blood may reveal circulating acanthocytes.
3. Blood chemistries may reveal hepatic dysfunction related to Wilson disease or acquired hepatocerebral degeneration; hyperthyroidism or hypocalcemia as a cause of chorea; or a variety of metabolic disorders associated with myoclonus.
4. Serologic tests are helpful for diagnosing movement disorders caused by systemic lupus erythematosus or lupus anticoagulant syndrome. Neurosyphilis and HIV-1 infection can be manifested clinically in a variety of ways and should always be excluded by appropriate serologic tests in patients with neurologic disease of uncertain etiology.
ELECTROPHYSIOLOGIC TESTS
An EEG is sometimes helpful in diagnosing patients with myoclonus and in distinguishing paroxysmal dyskinesias from seizures; otherwise, it is of limited usefulness. Electromyography and somatosensory evoked potentials may help to determine the level of neural involvement in myoclonus.
IMAGING
Radiologic studies are occasionally helpful in evaluating patients with movement disorders. In some patients, intracranial calcification may be found by skull x-rays or computed tomography (CT) scans; the significance of this finding, however, is not clear. CT scans or magnetic resonance imaging (MRI) may also reveal a tumor or other lesion associated with focal dyskinesia or dystonia or with symptomatic myoclonus, caudate atrophy due to Huntington disease, or basal ganglia abnormalities associated with Wilson disease. Positron emission tomography (PET) using 18F-dopa can monitor the loss of nigrostriatal projections in Parkinson disease and may be helpful diagnostically in patients with incomplete parkinsonian syndromes, but is not widely available. Dopamine transporter imaging using beta-CIT single-photon emission computed tomography (SPECT) can also be used for this purpose.
GENETIC STUDIES
Recombinant DNA technology has been used to generate probes for genes that determine certain inheritable movement disorders, such as Huntington disease and Wilson disease. Genetic markers are therefore of diagnostic value in such disorders (Tables 11-5 and 11-6). Their use may be limited, however, by the genetic heterogeneity of some diseases, imprecise gene localization by certain probes, ethical concerns about adverse psychologic reactions to the presymptomatic diagnosis of fatal disorders, and the potential for misuse of such information by prospective employers, insurance companies, and government agencies.
PSYCHOLOGIC EVALUATION
Cognitive and affective disturbances can be documented and characterized by neuropsychologic evaluation. This may be helpful in diagnosing certain disorders such as Huntington disease or diffuse Lewy body dementia. Some movement disorders, such as Gilles de la Tourette syndrome, are associated with behavioral abnormalities such as attention deficit disorder and obsessive-compulsive disorder. The findings also may be important in guiding decisions regarding invasive interventions such as deep brain stimulation. Such a therapeutic intervention is contraindicated in patients with atypical parkinsonian syndromes or in classic Parkinson disease when significant dementia or major depression is also present.
SELECTED MOVEMENT DISORDERS
The more common and well-defined diseases or syndromes characterized by abnormal movements are discussed here with the principles of their treatment.
FAMILIAL OR BENIGN ESSENTIAL TREMOR
PATHOGENESIS
A postural tremor may be prominent in otherwise normal subjects. Although the pathophysiologic basis of this disorder is uncertain, it often has a familial basis with an auto-somal dominant mode of inheritance. At least three gene loci have been implicated; in some cases (ETM1), the disorder is related to a polymorphism in the D3 dopamine receptor gene (DRD3).
CLINICAL FINDINGS
Symptoms may develop in the teenage or early adult years but often do not appear until later. The tremor typically involves one or both hands or the head and voice, whereas the legs tend to be spared. Examination usually reveals no other gross abnormalities, but some patients may have ataxia or personality disturbances. Although the tremor may become more conspicuous with time, it generally leads to little disability other than cosmetic and social embarrassment. In occasional cases, tremor interferes with the ability to perform fine or delicate tasks with the hands; handwriting is sometimes severely impaired. Speech is affected when the laryngeal muscles are involved. Patients commonly report that a small quantity of alcohol provides remarkable but transient relief; the mechanism is not known.
TREATMENT
If treatment is warranted, propranolol, 40 to 120 mg orally twice daily, can be prescribed, but it will need to be taken for an indefinite period. Other beta-blockers, such as atenolol and sotalol, have also been used. If tremor is particularly disabling under certain predictable circumstances, it can be treated with a single oral dose of 40 to 120 mg of propranolol taken in anticipation of the precipitating circumstances.
Primidone has also been effective, but patients with essential tremor are often very sensitive to this drug, so that it must be introduced more gradually than when it is used to treat epilepsy. Patients are therefore started on 50 mg/d, and the daily dose is increased by 50 mg every 2 weeks until benefit occurs or side effects limit further increments. A dose of 100 or 150 mg three times a day is often effective. There is no evidence that high doses (exceeding 750 mg daily) provide any added benefit.
Occasional patients respond to alprazolam, up to 3 mg/d in divided doses. Some patients reportedly benefit from gabapentin (1,200 mg/d), topiramate (400 mg/d), zonis-amide (up to 200 mg daily), or intramuscular injections of botulinum toxin. Anecdotal reports of benefit from mirtazapine were not confirmed in a double-blind study, which found no effect on the tremor in most patients.
Some patients have disabling tremor that is unresponsive to pharmacologic measures. Thalamotomy may be helpful, but a significant morbidity is associated with bilateral procedures. High-frequency thalamic stimulation by an implanted electrode is an effective alternative to thalamotomy and has a lower morbidity. It may be particularly useful for treatment of the unoperated side in patients who have already undergone unilateral thalamotomy. Benefit is maintained over the years in most patients with severe disability.
PARKINSONISM
Parkinsonism occurs in all ethnic groups; in the United States and western Europe it has a prevalence of 1 to 2 per 1,000 population, with an approximately equal sex distribution. The disorder becomes increasingly common with advancing age. It is characterized by tremor, hypokinesia, rigidity, and abnormal gait and posture.
ETIOLOGY
Idiopathic
The most common variety of parkinsonism occurs without obvious cause; this idiopathic form is called Parkinson disease or paralysis agitans when there are no atypical features, it is not secondary to some known cause, and there is a sustained response to treatment with dopaminergic medication. A preclinical phase extending back for several years before the development of the motor deficit is now recognized, during which hyposmia, constipation, anxiety, depression, and rapid-eye-movement (REM) sleep behavior disorder may be present. This is important to recognize in studies seeking predictive biomarkers and in attempts to identify protective factors.
Encephalitis Lethargica
In the first half of the 20th century, parkinsonism often developed in patients with a history of von Economo encephalitis. Because this type of infection is not now encountered, cases of postencephalitic parkinsonism are becoming increasingly rare.
Drug- or Toxin-Induced Parkinsonism
1. Therapeutic drugs—Many drugs, such as phenothiazines, butyrophenones, metoclopramide, reserpine, and tetrabenazine, can cause a reversible parkinsonian syndrome (see later). This is important to recognize because it is usually reversible by withdrawing the offending medication, although symptoms and signs may take many months to resolve.
2. Toxic substances—Environmental toxins such as manganese dust or carbon disulfide can lead to parkinsonism; manganese used in the home manufacture of methcathinone appears to have been responsible for parkinsonism in intravenous users of this illegal stimulant. The disorder may also appear as a sequela of severe carbon monoxide poisoning or exposure to fumes during welding. Experimental studies suggest that pesticide exposure is associated with the development of parkinsonism as well.
3. MPTP (1-methyl-4-phenyl-1,2,5,6-tetrahydropyri-dine)—A drug-induced form of parkinsonism has been described in individuals who synthesized and self-administered a meperidine analogue, MPTP. This compound is metabolized to a toxin that selectively destroys dopaminergic neurons in the substantia nigra and adrenergic neurons in the locus ceruleus and induces a severe form of parkinsonism in humans and in subhuman primates. The ability of this drug to reproduce neurochemical, pathologic, and clinical features of Parkinson disease suggests that an environmental toxin could be responsible for the idiopathic disorder. MPTP-induced parkinsonism has been used as a model to assist in the development of new drugs for treatment of this disease.
Vascular Parkinsonism
Multiple subcortical white-matter infarcts may lead to symptoms and signs suggestive of parkinsonism, usually accompanied by brisk tendon reflexes and extensor plantar responses. Tremor is often relatively inconspicuous and, in some patients, abnormalities of gait are especially evident (“lower-body parkinsonism”). The MRI findings help to suggest or support the diagnosis, and management is focused on preventing stroke. The response to antiparkinsonian medication is usually disappointing.
Post-traumatic Parkinsonism
Boxers and those engaged in certain other sports, such as football, may develop a syndrome of dementia (dementia pugilistica), behavioral and psychiatric disturbances, parkinsonism, and pyramidal and cerebellar deficits from recurrent head trauma leading to a chronic traumatic encephalopathy. There is no satisfactory treatment.
Familial & Genetic Parkinsonism
Rarely, parkinsonism occurs on a familial basis. Autosomal dominant parkinsonism may result from mutations of one of several genes, including α-synuclein (SNCA), leucine-rich repeat kinase 2 (LRRK2), and ubiquitin carboxyl-terminal esterase L1 (UCHL1) (Table 11-6). Mutations in parkin (PARK2) and DJ1 cause early-onset, autosomal recessive, and sporadic juvenile-onset parkinsonism. Several other genes or chromosomal regions have been implicated in familial forms of the disease or as susceptibility factors, including the gene for beta glucosidase (GBA), the enzyme deficient in the lysosomal storage disorder Gaucher disease.
Parkinsonism Associated with Other Neurologic Diseases
Parkinsonism that occurs in association with symptoms and signs of other neurologic disorders is considered briefly in the later section on differential diagnosis.
PATHOLOGY
Idiopathic parkinsonism (Parkinson disease) is a proteinopathy characterized by the misfolding and aggregation of α-synuclein. It is thus also referred to as a synucleinopathy. Histopathologic examination at advanced stages shows loss of pigmentation and cells in the substantia nigra and other brainstem centers, cell loss in the globus pallidus and putamen, and filamentous eosinophilic intraneural inclusion granules (Lewy bodies) containing α-synuclein in the basal ganglia, brainstem, spinal cord, and sympathetic ganglia. The distribution of Lewy bodies is more widespread than originally appreciated, with early involvement of the lower brainstem (eg, dorsal motor nucleus of the vagus [X] nerve), olfactory bulb, and enteric nervous system, and subsequent spread to the locus ceruleus, substantia nigra, transentorhinal cortex, hippocampus, and neocortex. Lewy bodies are not seen in postencephalitic parkinsonism; instead there may be nonspecific neurofibrillary degeneration in a number of diencephalic structures as well as changes in the substantia nigra.
PATHOGENESIS
As in other neurodegenerative proteinopathies (discussed at greater length in Chapter 5), the disease is thought to be triggered by protein misfolding and aggregation—in Parkinson disease, the protein involved is α-synuclein. Abnormal protein may subsequently spread from cell to cell and thereby propagate the disease to contiguous parts of the nervous system.
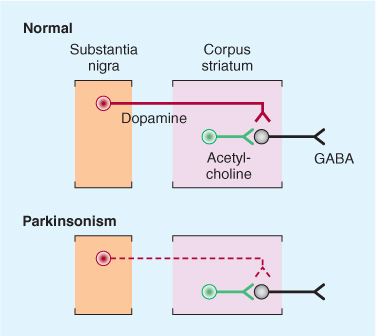
The motor manifestations of Parkinson disease appear to result from altered patterns of inhibition and excitation within the basal ganglia and its connections via direct and indirect pathways (Figure 11-2). Dopamine and acetylcho-line act as neurotransmitters in this region. In idiopathic parkinsonism, the normal balance between these two antagonistic neurotransmitters is disturbed because of dopamine depletion in the dopaminergic nigrostriatal system (Figure 11-3). Other neurotransmitters, such as norepinephrine, are also depleted in the brains of patients with parkinsonism, but the clinical relevance of this deficiency is less clear.
Figure 11-2. Functional circuitry between the cerebral cortex, basal ganglia, and thalamus. The major neurotransmitters and their excitatory (+) or inhibitory (–) effects are indicated. In Parkinson disease, there is degeneration of the pars compacta of the substantia nigra, leading to overactivity in the indirect pathway (red) and increased glutamatergic output from the subthalamic nucleus. (From Aminoff MJ. Pharmacologic management of parkinsonism and other movement disorders. In Katzung BG, Masters SB, Trevor AJ, eds. Basic and Clinical Pharmacology. 11th ed. New York, NY: McGraw-Hill; 2009.)
Figure 11-3. Schematic representation of the sequence of neurons involved in parkinsonism. Top: Dopaminergic neurons (red) originating in the substantia nigra normally inhibit the GABAergic output from the striatum (caudate and putamen), whereas cholinergic neurons (green) exert an excitatory effect. Bottom: In parkinsonism, there is a selective loss of dopaminergic neurons (dashed, red). This leads to increased inhibitory output from the striatum. (From Aminoff MJ. Pharmacologic management of parkinsonism and other movement disorders. In Katzung BG, Masters SB, Trevor AJ (eds). Basic and Clinical Pharmacology. 12th ed. New York, NY: McGraw-Hill; 2012.)
CLINICAL FINDINGS
Tremor
The 4- to 6-Hz tremor of parkinsonism is characteristically most conspicuous at rest; it increases at times of emotional stress and often improves during voluntary activity. It commonly begins in the hand or foot, where it takes the form of rhythmic flexion–extension of the fingers or of the hand or foot—or of rhythmic pronation–supination of the forearm. It frequently involves the face in the area of the mouth as well. Although it may ultimately be present in all of the limbs, it is not uncommon for the tremor to be confined to one limb—or to the two limbs on one side—for months or years before it becomes more generalized. In some patients, tremor never becomes prominent.
Rigidity
Rigidity or increased tone, that is, increased resistance to passive movement, is a characteristic clinical feature of parkinsonism. The disturbance in tone is responsible for the flexed posture of many patients with parkinsonism. The resistance is typically uniform throughout the range of movement at a particular joint and affects agonist and antagonist muscles alike—in contrast to the findings in spasticity, where the increase in tone is often greatest at the beginning of the passive movement (clasp-knife phenomenon) and more marked in some muscles than in others. In some instances, the rigidity in parkinsonism is described as cogwheel rigidity because of ratchet-like interruptions of passive movement that may be due, in part, to the presence of tremor.
Hypokinesia
The most disabling feature of this disorder is hypokinesia (sometimes called bradykinesia or akinesia)—a slowness of voluntary movement and a reduction in automatic movement, such as swinging the arms while walking. The patient’s face is relatively immobile (hypomimia or masklike facies), with widened palpebral fissures, infrequent blinking, a certain fixity of facial expression, and a smile that develops and fades slowly. The voice is of low volume (hypophonia) and tends to be poorly modulated. Fine or rapidly alternating movements are impaired, but power is not diminished if time is allowed for it to develop. The handwriting is small (micrographia), tremulous, and hard to read.
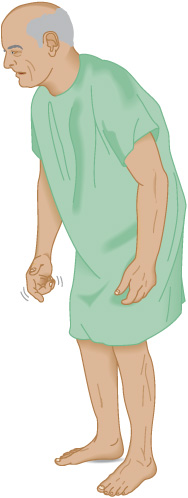
Abnormal Gait & Posture
The patient generally finds it difficult to get up from bed or an easy chair and tends to adopt a flexed posture on standing (Figure 11-4). It is often difficult to start walking, so the patient may lean farther and farther forward while walking in place before being able to advance. The gait itself is characterized by small, shuffling steps and absence of the arm swing that normally accompanies locomotion; there is generally some unsteadiness on turning, and there may be difficulty in stopping. Retained arm swing, wide-based gait, or marked imbalance at an early stage suggests a nonparkinsonian disorder. In advanced cases, the patient tends to walk with increasing speed to prevent a fall (festinating gait) because of the altered center of gravity that results from the abnormal posture.
Figure 11-4. Typical flexed posture of a patient with parkinsonism.
Other Motor Abnormalities
There is often mild blepharoclonus (fluttering of the closed eyelids) and occasionally blepharospasm (involuntary closure of the eyelids). The patient may drool, perhaps because of impairment of swallowing. There is typically no alteration in the tendon reflexes (although a mild hyperreflexia may occur on the affected side in asymmetric parkinsonism), and the plantar responses are flexor. Repetitive tapping (approximately twice per second) over the bridge of the nose produces a sustained blink response (Myerson sign); the response is not sustained in normal subjects.
Nonmotor Manifestations
Anosmia is an early symptom (but may arise from many other causes, and is therefore not a specific indicator of Parkinson disease). Cognitive decline, executive dysfunction, and personality changes are common, as also are depression and anxiety. A sense of fatigue may be prominent, and some patients complain of pain or sensory disturbances. Dysautonomic symptoms are often troublesome, especially urinary urgency and urge incontinence, and constipation; postural hypotension relates most commonly to dopaminergic therapy or inactivity but may also reflect baroreflex failure or denervation of cardiac muscle. Sleep disorders including REM behavior disorder are common. Seborrheic dermatitis may occur.
DIFFERENTIAL DIAGNOSIS
The diagnosis may be difficult to make in mild cases. Some degree of slowing is normal in the elderly, and certain otherwise normal people have a deliberate slowness about them.
Depression
Depression may be accompanied by a somewhat expressionless face, poorly modulated voice, and reduction in voluntary activity; it can thus simulate parkinsonism. Moreover, the two diseases often coexist in the same patient. A trial of antidepressant drug treatment may be helpful if diagnostic uncertainty cannot be resolved by the presence of more widespread neurologic signs indicative of parkinsonism.
Essential (Benign Familial) Tremor
This has been considered separately (see earlier discussion). An early age at onset, a family history of tremor, the relationship of the tremor to activity, a beneficial effect of alcohol on the tremor, and a lack of other neurologic signs distinguish this disorder from parkinsonism. Furthermore, essential tremor commonly affects the head (causing a nod or head shake); parkinsonism typically affects the lower jaw rather than the head.
Dystonia
A dystonic tremor may also be mistaken for parkinsonism, particularly when the dystonia is mild or unrecognized.
Diffuse Lewy Body Disease
This disorder occurs especially in patients aged between 60 and 80 years and is marked clinically by the combination of a rapidly progressing neurobehavioral syndrome of dementia and hallucinations and extrapyramidal motor features characteristic of Parkinson disease. Myoclonus may also be seen. There is only an incomplete response to levodopa, but patients are extremely sensitive to parkinsonian complications of neuroleptics as well as to the side effects of antiparkinsonian drugs. It is discussed further in Chapter 5 and later in this chapter.
Wilson Disease
Wilson disease (discussed in more detail later) can also lead to a parkinsonian syndrome, but other varieties of abnormal movements are usually present as well. Moreover, the early age at onset and the presence of Kayser-Fleischer rings should distinguish Wilson disease from Parkinson disease, as should the abnormalities in serum and urinary copper and serum ceruloplasmin that occur in Wilson disease.
Huntington Disease
Huntington disease may occasionally be mistaken for parkinsonism when it presents with rigidity and akinesia, but a family history of Huntington disease or an accompanying dementia, if present, should suggest the correct diagnosis, which can be confirmed by genetic studies.
Striatonigral Degeneration
This rare disorder is one subtype of multisystem atrophy (MSA) and is now referred to as MSA-P. It is associated with neuronal loss in the putamen, globus pallidus, and caudate nucleus and presents with bradykinesia and rigidity. Antiparkinsonian drugs are typically ineffective. Striatonigral degeneration may be associated with cerebellar degeneration (Chapter 8), in which case the term MSA-C is applied. When autonomic insufficiency is a conspicuous accompaniment, the eponymous designation of Shy-Drager syndrome
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree