Chapter 18 Neonatal Brain Injury
Although neonatal stroke and trauma are not uncommon, data about risk factors, causes, and outcomes are still scarce because of a paucity of population-based studies. However, emerging data are changing previous concepts regarding pathophysiology, especially for neonatal stroke. In the past, outcomes for these conditions have been primarily focused on the major neurologic residua of cerebral palsy (see Chapter 69). However, data from the Canadian Pediatric Stroke Registry and the German Childhood Stroke Study Group [deVeber et al., 2000; Kurnik et al., 2003] and from recent longitudinal studies [Westmacott et al., 2009a, b; Ballantyne et al., 2008; Ricci et al., 2008] suggest that cognitive deficits only become apparent during early childhood. This chapter reviews what is known regarding the incidence, causes, risk factors, and outcome of stroke and traumatic brain injury, with attention to the clinical presentations and methods of diagnosis and management after these conditions occur. Pathophysiology is discussed in more detail in Chapter 17 because hypoxic-ischemic injury and focal ischemia-reperfusion injury in the setting of stroke are thought to have common pathogenetic mechanisms. However, pathophysiology of trauma in the newborn period is relatively unstudied, and only broad assumptions can be made for these conditions.
Perinatal Arterial Ischemic Stroke
Definitions and Epidemiology
A recent National Institute of Neurological Disorders and Stroke workshop on perinatal stroke provided consensus recommendations on the definition and classification of ischemic perinatal stroke (Raju et al., 2007). Agreement about the following working definition was obtained during the workshop: “a group of heterogeneous conditions in which there is a focal disruption of cerebral blood flow secondary to arterial or cerebral venous thrombosis or embolization, occurring between 20 weeks of fetal life through the 28th postnatal day and confirmed by neuroimaging or neuropathology studies.” The two major subtypes, ischemic (including cerebral venous thrombosis) and hemorrhagic perinatal stroke, will be discussed separately. Time of onset can be considered as “late fetal” (28 weeks to birth), “perinatal” (28 weeks to 7 days after birth), “neonatal” (0–28 days after birth), and “presumed perinatal” (symptoms presenting beyond day 28 of life). This part of the chapter will focus on perinatal arterial ischemic stroke (PAIS).
Brain infarction due to occlusion of a major artery or one of its territorial branches is now recognized in an increasing number of full-term infants, with an incidence of approximately 1 per 2300–4000 live births, not dissimilar to that of stroke in the elderly [Estan and Hope, 1997; Nelson and Lynch, 2004; Schulzke et al., 2005; Schneider et al., 2004; Kirton and deVeber, 2009]. Data in preterm infants are scarce, but have recently been reported with an estimated incidence of 0.7 percent in a hospital-based case-control study [Benders et al., 2007].
Stroke in newborns is reported to have a male predominance [Chabrier et al., 2009], but this was not seen in a recent study from the International Pediatric Stroke Study (IPSS) registry data, of 249 infants aged less than 29 days with arterial ischemic stroke. Just over half of the patients (57 percent) were male, with a 1.3:1 male:female ratio, which is not significant [Golomb et al., 2009]. There is a tendency toward involvement of left-sided lesions within territories of the middle cerebral artery [Golomb et al., 2009; Trauner et al., 1993]. This is thought to be due to hemodynamic differences in cerebral blood flow between the right and left carotid arteries due to the hemodynamic effects of a patent ductus arteriosus or possibly due to preferential flow of placental emboli into the left-sided cerebral vessels.
Despite relatively low mortality (3–10 percent in the Canadian Stroke Register) [deVeber et al., 2000] and low recurrence rates (1.1–5 percent) [deVeber et al., 2000; Fullerton et al., 2007; Kurnik et al., 2003], there is a significant risk of developing adverse neurologic sequelae. Unilateral spastic cerebral palsy develops in about one-third of infants, and impairments in vision, cognition, language, and behavior are common [Lee et al., 2005; Westmacott et al., 2009].
The infant mortality rate in the United States for 1995–1998 due to stroke (ICD-9 CM 430–437) is reported to be 5.33 deaths per 100,000 per year, the perinatal mortality rate is 2.21 per 100,000, and the neonatal mortality rate is 3.49 per 100,000 live births per year [Lynch and Nelson, 2001]. The National Hospital Discharge Survey (1980–1998) determined that, for infants younger than 30 days of age, the hospital mortality rate for neonatal stroke is 10.1 percent, or 2.67 deaths per 100,000 live births [Lynch and Nelson, 2001; Lynch et al., 2002], which is similar to a more recent review, in which the infant death rate due to cerebrovascular disorders was 3 per 100,000 in the United States in 2005 [Kung et al., 2008].
Pathophysiology and Risk Factors
Many maternal, fetal, and neonatal risk factors are associated with neonatal arterial ischemic strokes, but evidence for true causation is lacking in most cases. Often, more than one potential risk factor can be identified (Figure 18-1).
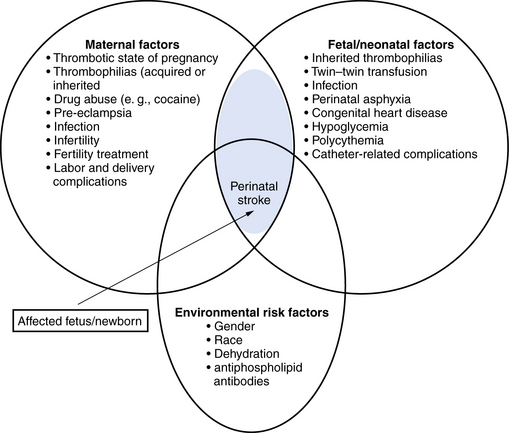
Fig. 18-1 Risk factors for perinatal arterial stroke.
(Adjusted from Raju TN, Nelson KB, Ferriero D et al. Ischemic perinatal stroke: summary of a workshop sponsored by the National Institute of Child Health and Human Development and the National Institute of Neurological Disorders and Stroke. Pediatrics 2007;120:609.)
Maternal Risk Factors
A history of infertility was identified as a risk factor in two recent studies (7–11 percent) [Lee et al., 2005; Curry et al., 2007]. An assumption is made that ovarian stimulation drugs would result in a hypercoagulable state, leading to placental thrombosis, but this still has to be confirmed with placental examination. Other maternal conditions, such as thyroid disease, diabetes mellitus, and gestational diabetes, were noted, but have not been identified as independent risk factors.
Antepartum Risk Factors
Pre-eclampsia was identified as a consistent independent risk factor in several studies (15–25 percent) [Lee et al., 2005; Curry et al., 2007]. This condition is known to affect uteroplacental blood flow. Oligohydramnios and decreased fetal movements have also been identified as risk factors, but both have no longer found to be significant in multiple regression analyses [Lee et al., 2005; Curry et al., 2007].
Intrapartum Risk Factors
While Ramaswamy et al. [2004] found only six cases with neonatal arterial ischemic stroke among 124 infants admitted with neonatal encephalopathy, the birth process tends to be reported as being ‘complicated’ and an intervention to deliver the infant is often required. Infants were not often severely depressed at birth, however, and were often well enough to stay with their mothers rather than in a neonatal intensive care unit. Factors that emerged after multivariate analysis were a prolonged second stage of labor (OR = 8.8; P = 0.001) and prolonged rupture of membranes (OR = 3.4; P = 0.05) [Lee et al., 2005]. More general signs of fetal distress (fetal heart-rate abnormalities, meconium-stained amniotic fluid, an Apgar score <7 at 5 minutes), cord abnormalities (tight nuchal cord), and need for intervention during delivery (instrumental delivery or need for emergent cesarean section) were more common in cases compared to controls, but were not identified as independent risk factors. A combination of three or more risk factors led to an odds ratio of 25.3. This implied a risk of 1 in 200 when three or more of the following risk factors were present: primiparity, infertility, oligohydramnios, pre-eclampsia, chorioamnionitis, prolonged rupture of membranes, decreased fetal movements, prolonged second stage of labour, and fetal heart-rate abnormalities [Lee et al., 2005].
Although the placenta is the most likely source of emboli to the fetal brain, data about the placenta are scarce, mainly because the placenta is often unavailable by the time the infant develops symptoms. Several placental abnormalities have been reported in infants with perinatal stroke, such as placental thrombosis, infarction, chorioamnionitis, funisitis, and placental chorioangiomas [Benders et al., 2007; Burke et al., 1997; Kraus et al., 1999; Elbers et al., 2011].
Postnatal Risk Factors
Prothrombotic risk factors
The peripartum period is a prothrombotic state in both the mother and fetus. Prothrombotic factors have been reported to play a possible role in the child, and more recently, it also has been shown that a prothrombotic factor is more often present in both mother and child [Simchen et al., 2009]. Gunther et al. [2000] performed a case-control study in 91 cases and showed that the percentage of prothrombotic risk factors was significantly higher in affected infants (68 percent compared to 24 percent; OR = 6,7, CI = 3,8–11,7). The most commonly found prothrombotic factors were elevated lipoprotein(a) and factor V Leiden, but hyperhomocysteinemia and mutations in the methyltetrahydrofolate reductase (MTHFR) gene also have been identified as associated factors. In two recent studies, high percentages of prothrombotic factors were found in both the neonate and the mother, as well as in mother–infant pairs, demonstrating the importance of also performing a thrombophilia screen in the mother [Curry et al., 2007; Simchen et al., 2009]. Simchen et al. [2009] noted a 8.5-fold increased risk for arterial ischemic stroke in a mother carrying factor V G1691A mutation (FVL), and a 2.1- and 3.8-fold increased risk in mothers with factor II G20210A mutation, or acquired antiphospholipid antibodies (APA) as compared to controls. In the study by Curry et al. [2007], a heterozygous or homozygous MTHFR C677T mutation was most common, seen in 41 of 60 neonates (68 percent). However, no significant differences between frequencies of prothrombotic coagulation defects were found in another recent cohort study [Miller et al., 2006].
Other neonatal risk factors
Arterial ischemic stroke is found in almost 40 percent of a series of infants preceding repair of congenital heart disease, and this is especially common in those infants who require atrial septostomy [McQuilllen et al., 2006]. In a more recent study, 10 percent of 122 patients with congenital heart disease were diagnosed with stroke, and in 6 patients the stroke occurred preoperatively. Stroke was clinically silent in 11 of the 12 infants [Chen et al., 2009]. Newborn infants undergoing extracorporeal membrane oxygenation are also at increased risk for developing arterial ischemic stroke (Wu et al., 2005).
Pais in the preterm infant
Independent risk factors identified in a case-control study of 31 preterm infants with stroke, using multivariate risk factor analysis, were different from risk factors identified in full-term infants. Twin-to-twin transfusion syndrome (TTTS) (OR = 31,2; 95 percent CI = 2.9–340.00), abnormal heart rate pattern (5,2; 1,5–17,6), and hypoglycaemia (<2 mmol/L) (3.9, 1,2–12,6) were independently associated with PAIS [Benders et al., 2007].
Perinatal Hemorrhagic Stroke
In a population-based cohort over a period of 10 years, a prevalence of 6.2 in 100,000 live births was observed for perinatal hemorrhagic stroke (PHS) [Armstrong-Wells et al., 2009]. A case-control study was performed with three controls per case. In contrast to what is known in infants with PAIS, all newborn infants with PHS presented with encephalopathy and more than half with seizures (65 percent). PHS was typically unifocal (74 percent) and unilateral (83 percent). Etiologies included thrombocytopenia (n = 4) and cavernous malformation (n = 1); 15 (75 percent) were idiopathic. Fetal distress and postmaturity were identified as independent predictors in a multivariate analysis.
Clinical Presentation and Diagnosis
PAIS in the newborn often manifests with seizures. The infants are initially well and have stayed with their mother on the ward, or can even be admitted from home. Whereas seizures associated with other forms of neonatal encephalopathy tend to be multifocal or myoclonic, seizures associated with arterial ischemic stroke tend to be persistently focal motor, usually involving only one upper extremity. Seizures tend to occur later than in neonates with hypoxic-ischemic encephalopathy (HIE) [Rafay et al., 2009]. Using multivariate analysis in 27 with PAIS and 35 with HIE, delayed seizure onset (≥12 hrs after birth) (p <0.0001; OR = 39.7; CI = 7.3, 217) and focal motor seizures (p = 0.007; OR = 13.4; 95 percent CI = 2.1,87.9) predicted stroke. Other more subtle symptoms observed in a large cohort of 215 newborn infants included poor feeding, hypotonia, and apnea [Kurnik et al., 2003; Redline et al., 2008].
In a population-based study of PAIS in children with motor impairment, most of the children presented after 3 months of life with hemiparesis (first recognized as early hand preference) or seizures [Wu et al., 2004]. These children are considered to have so-called “presumed perinatal ischemic stroke” (PPIS) [Kirton et al., 2008], which is diagnosed as a term (>36 weeks) infant older than 28 days with a normal neonatal neurologic history presenting with a neurological deficit or seizure referable to focal, chronic infarction on neuroimaging. Focal infarction specifies stroke (without limiting lesions to arterial territories) and includes unilateral/multiple/bilateral infarcts while excluding global injuries (HIE, watershed infarction, and periventricular leukomalacia [PVL]). Lesion chronicity is implied by imaging (restricted diffusion absent, encephalomalacia, gliosis, and atrophy of connected structures or Wallerian degeneration).
Infants with PPIS are less likely to have cortical involvement and more often show involvement of one or more of the lenticulostriate arteries. In a recent study, only 40 percent of late-diagnosed cases showed cortical involvement [Laugesaar et al., 2007]. In a large series of 59 cases with PPIS, venous periventricular venous infarction (PVI) was included in the spectrum of presumed perinatal stroke. Arterial proximal middle cerebral artery M1 segment infarction was most common (n = 19; 35 percent), and venous PVI was second (n = 12; 22 percent) and accounted for 75 percent of subcortical injuries [Kirton et al., 2008]. When risk factors are studied and outcome is reported, it is better to report children who are diagnosed in the neonatal period separately from those diagnosed later, referred to as PPIS.
Imaging
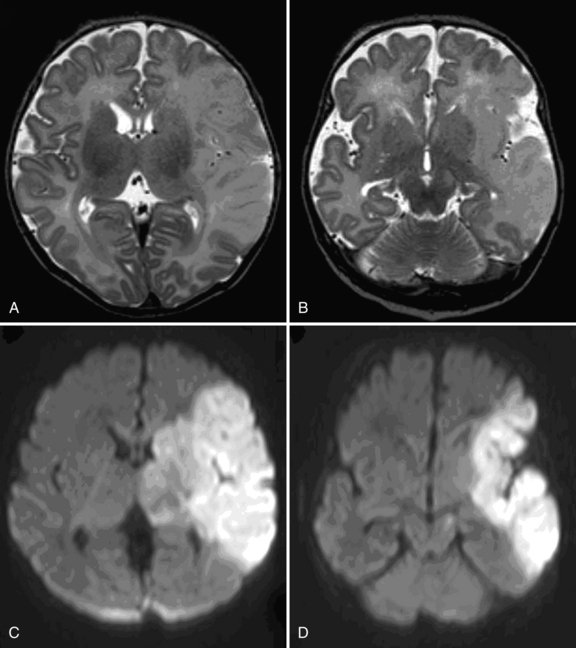
Cranial ultrasound studies (Box 18-1) may be normal if the stroke is superficial and ischemic, or they may reveal a wedge-shaped area of increased echogenicity with a linear demarcation line, usually within the territory of the middle cerebral artery. The echogenicity tends to become visible during the second half of the first week. Magnetic resonance imaging (MRI), including diffusion-weighted imaging (DWI), and MR angiography, is superior to head ultrasound, and should be performed in any newborn presenting with focal neonatal seizures.
Box 18-1 Evaluation of Neonates with Arterial Ischemic Stroke or Cerebral Sinovenous Thrombosis*
* There are few studies in this area, and there are no universally accepted guidelines.
† Normal levels (determined by antigen measurement and activity) for neonates are lower than for older children and adults. Levels may be temporarily low in the setting of acute illness; the significance of this is unclear.
(Data from Debus OM et al. The factor V G1691A mutation is a risk for porencephaly: A case-control study. Ann Neurol 2004;56:287; Manco-Johnson MJ, Grabowski EF, Hellgreen M et al. Laboratory testing for thrombophilia in pediatric patients. On behalf of the Subcommittee for Perinatal and Pediatric Thrombosis of the Scientific and Standardization Committee of the International Society of Thrombosis and Haemostasis [ISTH]. Thromb Haemost 2002;88:155–156.)
The role of DWI in the prediction of motor outcome was first shown by Mazumdar et al. [2003] and subsequently by others [de Vries et al., 2005; Kirton et al., 2007], showing restricted diffusion within the descending corticospinal tracts. Restricted diffusion at the level of the internal capsule and especially the middle part of the cerebral peduncle is now referred to as “pre-Wallerian degeneration,” as it is followed by Wallerian degeneration at 6–12 weeks and beyond (Figure 18.2).
A simple classification has been used, based on the primary artery involved [de Vries et al., 1997; Mercuri et al., 2004]. Infarcts in the territory of the middle cerebral artery were further subdivided into main branch, cortical branch, and lenticulostriate branch infarctions. Others have used a different classification, making a distinction between proximal M1 (PM1) of the middle cerebral artery that includes the lateral lenticulostriate (LLS) arteries, associated with infarction of the basal ganglia or distal M1 (DM1), in which compromised flow occurs distal to LLS and spares the basal ganglia while infarcting distal (cortical) middle cerebral artery territory. A third type of occlusion involves the anterior trunk of the middle cerebral artery, which is a superior middle cerebral artery division infarction that includes the frontal lobe (anterior-to-central sulcus) and the anterior temporal lobe (distal anterior trunk branch occlusions included). A fourth type involves the posterior trunk of the middle cerebral artery and is an inferior middle cerebral artery division infarction that includes parietal (posterior-to-central sulcus) and posterior temporal lobes (distal posterior trunk branch occlusions included). Finally, a fifth type, LLS infarcts, affects the basal ganglia (putamen and caudate body) and posterior limb of internal capsule (PLIC), with sparing of the subcortical white matter and cortex [Kirton et al., 2008]. Comparison of full-term and preterm infants showed that main branch involvement is rare in preterm infants, who tend to have LLS infarcts and less often show cortical involvement [Benders et al., 2009].
Newly developed techniques, such as diffusion tensor imaging (DTI), which allows quantification and visualization of white matter pathways in vivo, are more frequently being used in the evaluation of neonatal stroke [Lequin et al., 2009]. DTI characterizes the three-dimensional spatial distribution of water diffusion in each MRI voxel (see Chapter 11). Water diffuses preferentially along the direction of the axons and is restricted perpendicular to axons by myelin. This directional dependency is referred to as anisotropy. Directionality-encoded color maps (red–green–blue, RGB) or fiber tracking are commonly used. A fractional anisotropy (FA) map can show asymmetry of the PLIC as early as the neonatal period. In a study of 15 patients with congenital hemiparesis of different etiologies, studied at a median age of 2 years and compared with 17 age-matched controls, the clinical severity of hemiparesis was noted to correlate with asymmetry in FA (p <0.0001), transverse diffusivity (p <0.0001), and mean diffusivity (p <0.03) [Glenn et al., 2007]. Another promising technique is volumetric determination of stroke volumes, which was noted to predict motor outcome in animal studies [Ashwal et al., 2009]. Functional MRI (fMRI) tends to be used in childhood or adolescence to study reorganization of the sensorimotor cortex [Staudt et al., 2002; Seghier et al., 2005], but it was recently shown that passive unilateral sensorimotor stimulation is feasible, even in the preterm infant, resulting in bilateral activation of the sensorimotor cortex [Arichi et al., 2009; Heep et al., 2009].
Management/Treatment
Acute Period
For all types of perinatal stroke, supportive care with particular attention to monitoring of intracranial pressure is essential [Roach et al., 2008]. The use of continuous EEG monitoring or amplitude integrated EEG (aEEG) may be helpful for recognizing and treating neonatal seizures. Unilateral ictal discharges can be identified using two-channel aEEG, often suggesting a unilateral parenchymal lesion, usually PAIS or PHS before an MRI can be acquired [van Rooij et al., 2009]. Guidelines on the management and treatment of PAIS were published in 2008 [Roach et al., 2008; Monagle et al., 2008]. The American College of Chest Physicians (ACCP) only recommends administration of unfractionated heparin (UFH) or low molecular weight heparin (LMWH) in neonates with their first PAIS and an on-going documented cardioembolic source. Thrombolytic agents are not recommended.
Outcomes
Motor Effects
Arterial ischemic stroke may lead to permanent motor impairment or cerebral palsy. Estimates of motor impairment after neonatal arterial ischemic stroke range from 9 percent to 91 percent [Clancy et al., 1985; deVeber et al., 2000; de Vries et al., 1997; Golomb et al., 2008, Koelfen et al., 1995; Lee et al., 2005; Mercuri et al., 2004; Sran and Baumann, 1988; Trauner et al., 1993]. In a study by Lee et al. [2005], 58 percent of 40 children developed cerebral palsy and a delayed presentation was associated with increased risk for cerebral palsy (relative risk [RR], 2.2; 95 percent CI = 1.2–4.2). Cerebral palsy occurred in 68 percent of the largest cohort studied so far, and 87 percent of these children developed hemiplegia. This large cohort also consisted of infants with PAIS, with 47 percent developing cerebral palsy, as well as infants with PPIS, with 91 percent developing cerebral palsy. Most children with ischemic stroke diagnosed in the neonatal period attain independent walking, with the median time to taking first steps for the whole cohort, including infants with cerebral sinovenous thrombosis, being 13 months [Golomb et al., 2003]. In a study looking specifically at children with PPIS, motor outcomes (mean follow-up, 5.3 years) that were predicted by basal ganglia involvement included leg hemiparesis, spasticity, and need for assistive devices (p <0.01) [Kirton et al., 2008].
Sensory Deficits
Children with hemiplegic cerebral palsy are known to have sensory impairments but there are few detailed studies, in part because it is difficult to assess sensory function in young children [Cooper et al., 1995]. Thalamic atrophy has been seen in children with neonatal middle cerebral artery infarction, but whether this atrophy has long-term implications for sensory perception or memory is unclear [Giroud et al., 1995]. When children are carefully studied for visual field defects, hemianopia or quadrantanopia is not uncommon. Six (28 percent) of 16 school-age children with perinatal stroke had impaired visual function [Mercuri et al., 2003]. Children with unilateral pre- or perinatal stroke have more difficulty with facial recognition [Ballantyne and Trauner, 1999] and other visuospatial tasks [Schatz et al., 2000; Stiles et al., 1996, 1997].
Cognitive Effects
In some studies, most children with PAIS were noted to have intelligence within the normal range [Ricci et al., 2008; Trauner et al., 1993]. Cognitive impairment tends to be more common in the presence of hemiplegia and/or epilepsy. In a larger study of 46 infants, studied at a mean age of 42 months, cognitive impairment was present in 41 percent [Sreenan et al., 2000]. Cognitive outcome has only more recently been studied into childhood, showing more problems than intitially reported in small groups of infants studied early in life. Westmacott et al. [2009] studied 26 children with a history of acutely diagnosed unilateral PAIS as preschoolers (3 years 6 months to 5 years 11 months) and again as grade-school students (6 years 1 month to 12 years 5 months). While patients’ performance did not differ from the normative sample for full-scale IQ, verbal IQ, or performance IQ, and there were no significant differences associated with infarct laterality as preschoolers, performance was significantly lower than the normative sample for full-scale IQ working memory and processing speed, but not for verbal IQ or performance IQ at school age. Contrasts between preschool evaluation and grade-school evaluation revealed a significant decline in full-scale IQ, which reflected emerging deficits in nonverbal reasoning, working memory, and processing speed. Individual subject analyses revealed that 69 percent of the children showed significant declines in one or more IQ index measures. There were no significant differences in cognitive performance associated with lesion laterality, although males performed more poorly than females on several cognitive measures at grade school. These data are not in agreement with a study by Ballantyne et al. [2008], who found no evidence of a decline in cognitive function over time in children with perinatal unilateral brain damage, suggesting that there was sufficient on-going plasticity in the developing brain following early focal damage to result in the stability of cognitive functions over time. The time span between the two tests was shorter than in the study by Westmacott et al. [2009], and the time at first test was later, which could explain some of the differences. In another study by the same group, comparing effects of age at stroke and lesion location, it was found that the perinatal group performed more poorly than the childhood group, regardless of lesion location [Westmacott et al., 2009].
Language delay is not uncommon after perinatal stroke and was found in 25 percent of 36 survivors of the Kaiser Permanente study [Lee et al., 2005]; lesion laterality does not appear to predict degree of language impairment, but development of postneonatal epilepsy was a major contributor to language outcome [Ballantyne et al., 2008]. Above-average academic performance is, however, also possible in the setting of large middle cerebral artery strokes and hypsarrhythmia [Golomb et al., 2005]. In children with presumed perinatal stroke, nonmotor outcomes were associated with cortical involvement, including cognitive/behavioral outcomes, visual deficits, and epilepsy (p <0.01) [Kirton et al., 2008].
Epilepsy
Data about rates of childhood epilepsy after PAIS are still scarce. The longer the follow-up period, the greater the risk that there has been a recurrence of epilepsy following seizures seen in the neonatal period. During the first year there is a subset of children who will develop hypsarrhythmia, usually associated with cerebral palsy and cognitive impairment [Golomb et al., 2006]. Infants with hypsarrhythmia following “presumed perinatal stroke” appeared to have a better cognitive outcome. The highest rate of epilepsy after the neonatal period for neonates with arterial ischemic stroke reported is 67 percent, established in a group of 64 children, with resolution of their epilepsy in 32 percent [Golomb et al., 2007]. In a series of 46 children with a mean follow-up duration of 42.1 months (range 18–164 months), 46 percent developed epilepsy [Sreenan et al., 2000]. In a series of 67 preterm and full-term infants with PAIS, epilepsy occurred in 16 percent and was seen most often in children following main branch middle cerebral artery infarction (7/15, 47 percent) [Benders et al., 2009].
Recurrence
The recurrence rate after PAIS is low. The largest prospective study, which followed 215 neonates with arterial ischemic stroke for a median of 3.5 years, found that only 1.8 percent of children developed a recurrent stroke and 3.3 percent a recurrent symptomatic thromboembolism. Prothrombotic risk factors and the presence of additional morbidities, such as complex congenital heart disease or dehydration, were associated with an increased recurrence risk [Kurnik et al., 2003]. More recently a similar 5-year cumulative recurrence rate of 1.2 percent was reported (1 of 84 infants with PAIS) [Fullerton et al., 2007]. Also observed was the fact that children whose stroke occurred later in childhood had a 16-fold increased recurrence risk compared with the neonates (HR = 16; 95 percent CI = 2.1–120; p = 0.008).
Predictors of Outcomes
Vascular territory
The vascular territory that is infarcted aids in predicting outcome but is not highly accurate. One study of 24 children with perinatal stroke reported that concomitant involvement of cortex, basal ganglia, and internal capsule on MRI predicted hemiplegia more strongly than involvement of just one of these territories [Mercuri et al., 1999]. Two studies of initially 23 and subsequently 54 preterm and term neonates with middle cerebral artery ischemic stroke found that involvement of the main branch territory was predictive of hemiplegia [de Vries et al., 1997; Benders et al., 2009]. In a study of 62 children with neonatal arterial ischemic stroke, the presence of bilateral infarctions predicted a lower probability of walking [Golomb et al., 2003]. Another study found that large stroke size (RR = 2.0; 95 percent CI = 1.2–3.2) and injury to Broca’s area (RR = 2.5; 95 percent CI = 1.3–5.0), internal capsule (RR = 2.2; 95 percent CI = 1.1–4.4), Wernicke’s area (RR = 2.0; 95 percent CI = 1.1–3.8), or basal ganglia (RR = 1.9; 95 percent CI = 1.1–3.3) were predictors of hemiplegia [Lee et al., 2005]. Using DWI, “pre-Wallerian degeneration” can be recognized as restricted diffusion involving the descending corticospinal tracts. Restricted diffusion of the internal capsule and especially the cerebral peduncles is now considered to be the best and earliest predictor of hemiplegia [de Vries et al., 2005; Kirton et al., 2007]. In the small series of 14 infants studied by Kirton et al. [2007], percentage of peduncle being affected (p = 0.002), length of descending corticospinal tracts (p <0.001), and volume of descending corticospinal tracts (p = 0.002) were significantly associated with subsequent development of hemiplegia.
Neonatal seizures and early electroencephalogram
In one study of 46 neonates with arterial ischemic stroke, the presence of seizures in the neonatal period was predictive of one or more disabilities in the first years of life [Sreenan et al., 2000]. The presence of an abnormal background EEG pattern was a predictor of hemiplegia in another study [Mercuri et al., 1999].
Prothrombotic coagulation factors
Two studies showed an association between the presence of prothrombotic coagulation factors and neurological outcome. Among the 24 children studied by Mercuri et al. [2001], 9 developed hemiplegia and 8 had at least one prothrombotic coagulation factor (FVL or increased factor VIIIc), compared to only 2 (factor VIIIc) of the 13 with a normal outcome. In the study by Suppiej et al. [2008], inherited thrombophilia was significantly more common in patients with a poor neurologic outcome (Fisher’s exact test, p = 0.002), but this only applied to the children with PPIS.
Rehabilitation
As PAIS usually involves the middle cerebral artery, the upper extremity tends to be more severely affected than the lower extremity. Most children will be able to walk and first steps were taken at a mean age of 13 months, but this cohort consisted of children with PAIS (n = 62) or cerebral sinovenous thrombosis (n = 25) [Golomb et al., 2003]. Performing the assisting hand assessment (AHA) at 18 months can, to a certain degree, predict function of the assisting hand [Holmefur et al., 2009]. Until recently, treatment has been aimed at reducing spasticity and improving range of motion and function, using serial casting or botulinum toxin injections to reduce increased tone [Russo et al., 2007]. More recently, novel strategies have been used, such as constraint-induced movement therapy (CIMT), which involves restraining the unaffected arm to force use of the plegic arm, and this treatment strategy has been shown to be promising for improving function in hemiplegic cerebral palsy [Taub et al., 2004; Deluca et al., 2006]. In 4 out of 10 patients (age range 10–30 years) with hemiplegia following PAIS and preserved corticospinal projections from the affected hemisphere, increases in fMRI activation at the level of the primary sensorimotor cortex, as well as in the supplementary motor areas, were seen [Juenger et al., 2007]. In subsequent studies by the same group it was noted that improvement only occurred in patients with preserved crossed projections, with increased amplitude shown with transcranial magnetic stimulation (TMS) when stimulating the ipsilesional hemisphere [Kuhnke et al., 2008; Walther et al., 2009]. These studies, involving only small groups of children, do suggest that CIMT in PAIS with preserved crossed corticospinal projections induces neuroplastic changes in the primary motor cortex of the lesioned hemisphere, detected as increased cortical excitability (TMS) and increased task-related cortical activation (fMRI).
Neonatal Cerebral Sinovenous Thrombosis
Epidemiology
Population-based studies of neonatal cerebral sinovenous thrombosis (CSVT) are rare. Based on data from the Canadian Pediatric Stroke Registry, the incidence of CSVT in children is 0.67 per 100,000, and in neonates is 0.41 per 100,000 [deVeber et al., 2001]. This study included 69 neonates and is one of the larger studies in this population. A study from the Netherlands included 52 infants, admitted to five neonatal intensive care units [Berfelo et al., 2010]. The largest study to date, which was not population-based, included 84 neonates with symptomatic CSVT [Jordan et al., 2010]; 61 percent presented during the first week of life.
Pathophysiology and Risk Factors
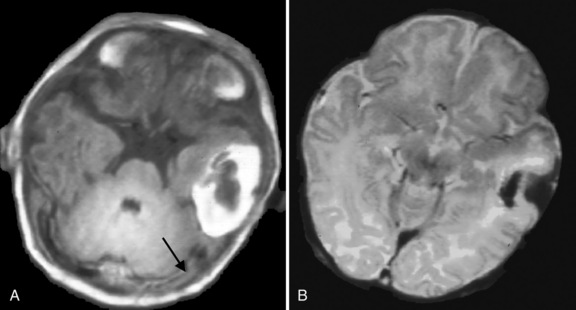
The cerebral veins drain into the dural venous sinuses and this slowly flowing blood is presumably more susceptible to thrombosis. The dural venous sinuses are near suture lines and this location may result in trauma at the time of birth [Shroff et al., 2003]. Occlusion of venous sinuses results in cerebral venous congestion and persumably is the cause of brain injury [Adams, 2007]. Increased venous pressure may cause cerebral edema, venous infarction with or without hemorrhagic conversion, and intraventricular hemorrhage [Wu et al., 2002]. Wu et al. first pointed out that CSVT should always be considered in the presence of an intraventricular hemorrhage (IVH) occurring at term, especially when this is associated with a unilateral thalamic hemorrhage. Thirty-one percent of 29 infants born > 36 weeks’ gestation, who were diagnosed with CSVT, presented with an IVH. Thalamic hemorrhage was diagnosed in 16 percent of infants with CSVT [Wu et al., 2002]. Intracranial hypertension is often present due to venous congestion and to impaired cerebrospinal fluid reabsorption through the arachnoid granulations that are within the venous sinuses [Adams, 2007].
Risk factors for CSVT during the first 28 days of life include dehydration, infection, maternal fever/chorioamnionitis, hypoxic-ischemic injury, and thrombophilia [Fitzgerald et al., 2006; Kenet et al., 2007]. Often, neonates have more than one risk factor [Wu et al., 2002].
Clinical Presentation and Diagnosis
Seizures are the most common presentation in a neonate, followed by nonspecific signs such as lethargy, hypotonia, feeding difficulties, respiratory distress, and apnea [deVeber et al., 2001; Fitzgerald et al., 2006; Wasay et al., 2008]. Focal deficits are present in very few neonates: 6 percent in two different studies [deVeber et al., 2001; Jordan et al., 2010]. With an extensive CSVT, neonates will present with tense or bulging fontanels, splaying of the cranial sutures, and prominent scalp veins. These are signs of poor cerebral venous drainage and increased intracranial pressure.
Cranial ultrasound may detect CSVT, particularly in the presence of a midline thrombus in the superior sagittal sinus, or a unilateral thalamic hemorrhage. Power Doppler may be superior to color Doppler when available [Govaert et al., 1992a; Tsao et al., 1999]. Additional imaging is required to exclude CSVT in more peripheral locations and to confirm the extent of the thrombus. Unenhanced CT may detect a thrombus, and contrast-enhanced CT may show the “empty delta” sign which is a filling defect in the posterior portion of the superior sagittal sinus due to thrombus. Radiologically based false-positive or missed diagnoses have been reported in up to 40 percent of children with CSVT [Davies and Slavotinek, 1994]. MRI of the brain and MR or computed tomography (CT) venography are needed to confirm the diagnosis [Eichler et al., 2007] (Figure 18-3 and Figure 18-4). Susceptibility-weighted imaging (SWI) has recently been reported as another useful sequence in confirming the presence of CSVT and for following to establish progression or resolution [Takekawa et al., 2008; Kawabori et al., 2009].
Management/Treatment
Published consensus-based guidelines for antithrombotic treatment of neonatal CSVT are not in agreement. The American College of Chest Physicians guidelines, published in 2004 [Monagle et al., 2004] and updated in 2008 [Monagle et al., 2008], suggest anticoagulation for neonates without significant intracranial hemorrhage, while the American Heart Association (AHA) guidelines, published in 2008 [Roach et al., 2008], recommend anticoagulation only when there is evidence of thrombus propagation, multiple cerebral or systemic emboli, or a severe prothrombotic state. From the Canadian Pediatric Ischemic Stroke Registry, only 25 of 69 neonates (36 percent) with CSVT, diagnosed between 1992 and 1998, were anticoagulated [deVeber et al., 2001]. Most treated neonates received low molecular weight heparin (LMWH; 20 of 25). None had hemorrhagic complications associated with death or neurologic deterioration. Hemorrhagic infarction was reported in 24 of 69 neonates (35 percent), while ischemic infarction was reported in 5 of 69 (7 percent). Similar data were obtained in the Netherlands, where 23 of 52 infants received anticoagulation without any hemorrhagic complications [Berfelo et al., 2010]. In a European collaborative study of childhood CSVT, 75 neonates were included [Kenet et al., 2007]. Specific data for neonates were not reported in detail, but there was no significant difference in treatment for children younger versus those older than age 2 years. If anticoagulation is deferred in a neonate with a CSVT, then follow-up neuroimaging should be performed after 5–7 days of supportive care to assess for thrombus propagation. If anticoagulation is used, the duration of therapy is typically 6 weeks to 3 months [Shroff et al., 2003; Moharir et al., 2010].
A recent small series (n = 10) of neonates with CSVT and intracerebral hemorrhage (unilateral thalamic hemorrhage) showed that 7 out of 10 neonates who were treated with anticoagulation did not show an increase of the thalamic hemorrhage [Kersbergen et al., 2009]. There are insufficient data to recommend thrombolysis; however, there are case reports of thrombolysis in the setting of progressive CSVT despite anticoagulation [Wasay et al., 2006; Wong et al., 1987].
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree