CHAPTER 137 Neoplastic Meningitis
The survival time of patients with NM is short, an average of 3.5 to 6 months. Longer survival rates are occasionally observed in patients with breast cancer (13% at 1 year and 6% at 2 years).1 In retrospective series, prognostic factors that correlate with longer survival times include young age, a Karnofsky Performance Scale (KPS) score higher than 70, a long duration of related symptoms, controlled systemic disease, lack of encephalopathy or cranial nerve deficits, low cerebrospinal fluid (CSF) protein levels, the presence of breast cancer, and in neuroimaging studies, the lack of bulky leptomeningeal deposits.1–7
Incidence
NM occurs in approximately 4% to 8% of patients with solid tumors, in 5% to 15% of patients with hematologic malignancies, and in less than 2% of those with primary brain tumors.1 The frequency of NM is also dependent on the histology and grade of the tumor and the duration of the disease. In approximately 1% to 7% of patients with NM, the primary tumor has not been located at the patient’s initial evaluation.8 NM is more likely to develop in patients with high-risk lymphomas (diffuse large B-cell lymphoma and Burkitt’s lymphoma), ALL, melanoma, breast adenocarcinoma, and small cell lung carcinoma.9–11
Pathogenesis
Metastasis to the leptomeninges is thought to result from hematogenous dissemination, but direct extension to the meninges can occur from parameningeal or contiguous bony structures (e.g., the base of the skull or vertebrae, regional lymph nodes, soft tissues) or by retrograde growth along the adventitia of blood vessels or perineurium of the spinal and cranial nerve roots.10,12 After entering the meninges, the CSF circulation provides an effective conduit for dissemination of tumor cells to the distant surfaces of the brain, spinal cord, nerve roots, and the ventricular ependymal surfaces. Late in the disease, invasion of the brain and spinal cord parenchyma by tumor from the meninges produces edema, microinfarction, and striking neurological dysfunction. Symptoms may also result from secondary complications such as hydrocephalus and increased intracranial pressure. Although this paradigm is accepted, it does not completely explain why NM does not develop in some patients with extensive systemic metastases, and little is known regarding the initial molecular and cellular mechanisms involved in the pathogenesis of NM.
Clinical Features
The typical clinical manifestations of NM are dependent on the level of nervous system involvement. Most commonly, patients have altered mental status, headache, diplopia, seizures, back pain, lower extremity weakness or numbness, or other cranial neuropathies. Positional headache, intractable nausea with vomiting, and visual obscurations may reflect increased intracranial pressure from CSF obstruction. The classic meningeal sign of nuchal rigidity has been reported in less than 20% of patients.8 Occasional patients, in particular those with breast cancer, may have more indolent courses. However, most patients rapidly progress at an alarming rate and experience neurological and systemic decline despite treatment efforts.
Diagnosis
Examination of Cerebrospinal Fluid
Definitive diagnosis of NM is made by identification of malignant cells in CSF. The relative sensitivity of this test depends on the number and volume of CSF specimens examined, the state of preservation of the CSF cells, and the source of the specimen (lumbar or cisternal being more reliable than intraventricular fluid). In one study, the sensitivity of the initial CSF examination was approximately 50% to 60% and improved to about 80% with a second specimen, and then each subsequent sample increased sensitivity by 2% to 5%.13 In another study, the yield of a single CSF analysis improved if the CSF volume studied exceeded 10.5 mL and was removed from the location (lumbar, cisternal, ventricular) nearest the symptomatic site or the area of greatest involvement, as seen on neuroimaging studies.14 Most cytopathologists prefer to assess serial samples obtained on different dates rather than a single large-volume specimen.
Although nonspecific, CSF protein determination has historically been the most sensitive indicator of NM because this protein level is abnormal (>45 mg/dL) in approximately 80% to 90% of patients.13 Conversely, finding a normal CSF protein reading is relatively strong (but not absolute) evidence against this diagnosis. Lumbar CSF is more likely than ventricular fluid to have elevated protein and malignant cells. Cisternal fluid has also been proposed to be a more reliable indicator, but its acquisition may carry greater risk.15 If high elevations (>500 mg/dL) of CSF protein are found in lumbar fluid, either NM is advanced or there is a partial or complete blockage of CSF flow from cephalad locations. Approximately 30% to 57% of patients have lumbar CSF pressure higher than 150 mm H2O, and approximately 31% to 55% have a reduction in CSF glucose levels. In patients with subsequently verified NM, less than 5% have completely normal CSF profiles.8
Neuroimaging
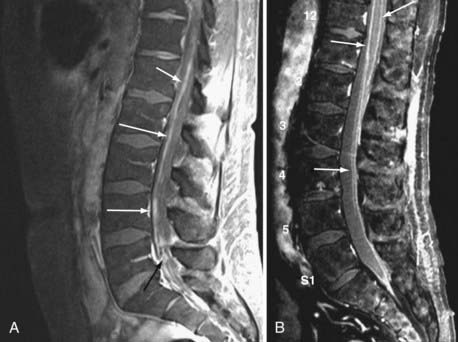
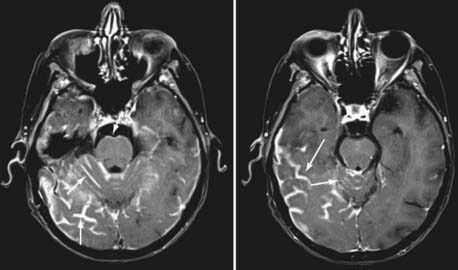
Currently, magnetic resonance imaging (MRI) of the brain and spine with gadolinium contrast enhancement is the procedure of choice. Findings on contrast-enhanced MRI will appear abnormal during the course of the disease in approximately 70% of patients with NM.16 High-resolution MRI is necessary for proper evaluation of the meninges of the spine. In general, lower resolution “open” MRI is less reliable for identification of NM unless florid abnormalities are present. Patchy nerve root enhancement, matting of nerve roots of the cauda equina, nodular deposits, and linear, continuous enhancement of the pia-arachnoid of the conus medullaris, brainstem, and cerebrum are hallmarks of the diagnosis (Fig. 137-1). Patchy, asymmetric enhancement of the leptomeninges over the cerebral convexities and in the sulci, basilar cisterns, insulae, pituitary stalk, cranial nerve roots, or superior cerebellar folia (or any combination of these sites) may be observed (Fig. 137-2). Indirect signs include communicating hydrocephalus, bilateral transependymal edema, and effacement of convexity sulci. Contrast enhancement of the meninges can be associated with many non-neoplastic conditions, including prior or current infections, previous subarachnoid hemorrhage, previous lumbar puncture or intrathecal chemotherapy, prior neurosurgical procedures (e.g., placement of an intraventricular reservoir), chronic CSF leak syndrome, and other neurological conditions.16 Nevertheless, in a patient with a high KPS score and active systemic cancer in whom symptoms or signs and neuroimaging findings strongly suggest NM, some physicians believe that treatment is justifiable even if the CSF cytologic study is negative.17,18
Cerebrospinal Fluid Flow Studies
Some investigators have proposed that CSF flow dynamics should be evaluated before initiating treatment of NM. CSF flow is usually assessed by nuclear medicine techniques with either 111In-diethylenetriamine pentaacetic acid or, less commonly, 99Tc-labeled albumin. Partial or total blockage of CSF flow has been identified in up to 30% to 70% of patients with NM.19,20 The stated rationale for performing flow studies includes identifying CSF flow blockage before treatment, preventing accumulation of high concentrations of administered drug in areas of CSF loculation, and conversely, identifying areas that would not receive adequate drug concentration beyond the areas of blockage. Retrospective studies have suggested that outcome is poorer in patients with CSF blocks or those in whom blocks cannot be opened with focal radiation therapy (RT).19,20 To date, routine use of flow studies has not gained widespread acceptance among practitioners.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree