FIGURE 58.1 T1 postcontrast axial MRI. Numerous small enhancing lesions are noted bihemispherically in this non–small cell lung cancer patient with brain metastases. These lesions are noted predominantly at the gray–white interface.
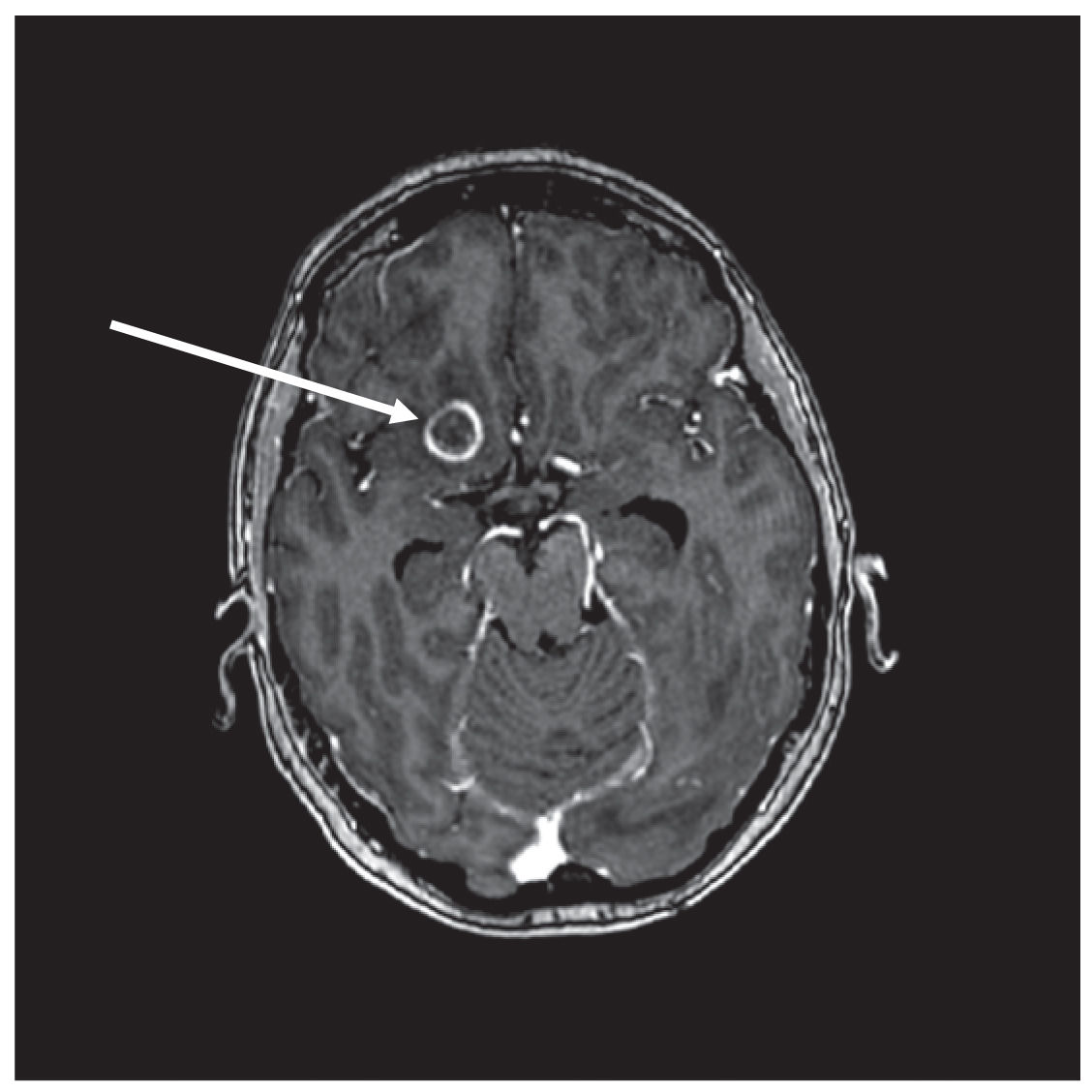
FIGURE 58.2 T1 postcontrast axial MRI. A larger ring-enhancing lesion (arrow) is noted in the inferior right frontal lobe in this patient with non–small cell lung cancer brain metastases. Peritumoral edema can be seen.
b. Disease-specific graded prognostic assessment (DS-GPA).
(1) A histology-specific system that includes lung, breast, melanoma, renal, and gastrointestinal primaries. The factors that influence prognosis vary between histologic groups and can include KPS, age, presence of extracranial metastases, number of brain metastases, and the molecular subtype of the tumor. Patients receive a summary score based on the addition of points for various prognostic factors. Median OS ranges between 2.79 and 25.30 months.
5. Management.
a. Surgery.
(1) Resection of a single brain metastasis followed by postoperative radiation has been shown to improve OS. The optimal surgical candidate has favorable prognostic factors, relatively well-controlled systemic disease, and reasonable systemic treatment options, as well as minimal surgical/anesthesia risk factors.
(2) In larger symptomatic lesions there may be benefit from alleviating symptoms with surgical resection.
(3) The role for surgical resection in the setting of multiple metastases and for recurrent disease is less clearly defined.
(4) In patients with obstructive hydrocephalus an extra-ventricular drain, ventriculoperitoneal shunt, or third ventriculostomy may need to be performed to divert CSF flow.
b. Radiotherapy.
(1) Whole-brain radiation therapy (WBRT)
(a) Conventional WBRT is performed using opposed lateral radiation beams. Although multiple fractionation schemes can be used, the most frequently employed is 30 Gy in 3 Gy fractions.
1) The most frequently encountered acute toxicities are fatigue and alopecia. Some patients experience worsening of their focal neurologic deficits because of progressive peritumoral edema. This can be diminished with the use of steroids. A small subset of patients develop acute encephalopathy.
(b) Methods for limiting potential toxicity
1) Hippocampal Avoidance WBRT (HA-WBRT). The use of an imaging-modulated radiation therapy plan administering a limited dose to the bilateral hippocampi has been evaluated and is undergoing additional investigation.
2) Addition of neuroprotective agents such as memantine has demonstrated decreased rate of decline in performance on neurocognitive tests. This strategy is actively undergoing additional investigation.
(c) Postoperative WBRT
1) The randomized trials of surgical resection for single brain metastases all used postoperative WBRT. There has been a strong growing trend toward limited field radiation in the postoperative setting.
(2) Stereotactic radiosurgery (SRS)
(a) SRS describes the use of a large focal dose of radiation, typically administered in a single fraction. Its role in brain metastases has been extensively studied alone or in combination with surgery, WBRT, or systemic therapies.
(b) Many would agree with a beneficial role for SRS in treating 1 to 4 brain metastases. It is less clear what the role is for SRS in the setting of >4 brain metastases. Other factors such as tumor histology, status of extra-CNS disease, and cumulative tumor volume may be of importance in defining the role for SRS. While the combination of SRS + WBRT provides more optimal local control it is not clear that it provides OS benefit.
c. Systemic therapies.
(1) Systemic therapies for brain metastases currently do not have a role in the upfront setting. They are being investigated in clinical trials and they can be employed in select patients after progression post-standard of care.
(2) As our understanding of the pathophysiology of disease advances the role for systemic therapies in the treatment and prophylaxis of brain metastases will likely evolve.
d. Symptom management.
(1) Seizures.
(a) The risk of seizures in patients with brain metastases is 20% to 80%. Factors such as tumor location (cortical/juxtacortical vs infratentorial, mesial temporal vs occipital) likely play a role in influencing incidence.
(b) There are no formal guidelines recommending prophylactic antiepileptic drugs (AEDs).
(c) When considering AEDs for the treatment of seizures in patients with brain metastases a number of factors in addition to efficacy should be considered. Absence of drug–drug interactions (lack of cytochrome P450 induction or inhibition) is preferred if possible. Cancer patients are often on other medications, including chemotherapies, the concentrations of which could be affected by cytochrome P450 metabolism. Lack of substantial hematologic or hepatic toxicities is also preferable, if possible. Additionally, the hematologic and hepatic toxicity of AEDs should be considered within the context of myelosuppressive/hepatotoxic chemotherapies. Finally, titration schedules should be considered in this patient population, which may be prone to frequent hospitalizations.
(2) Cerebral edema and increased ICP.
(a) Management of cerebral edema and increased ICP because of brain metastases is similar to the management because of other etiologies (see Chapter 63). It typically involves efforts to bring down ICP, often with the use of steroids or hyperosmolar agents such as mannitol.
B. Spinal metastases.
1. Nomenclature. Numerous anatomic locations of metastases are at times lumped together as “spinal metastases.” It is helpful to specify the specific anatomic localization (spinal cord parenchyma, CSF or dural disease affecting the cord, extradural disease affecting or potentially affecting the cord) as this influences clinical decision making. Often, “spinal metastases” should be evaluated and managed emergently. Because of the potential for lesions at multiple levels, imaging of the entire spine is preferred in most cases.
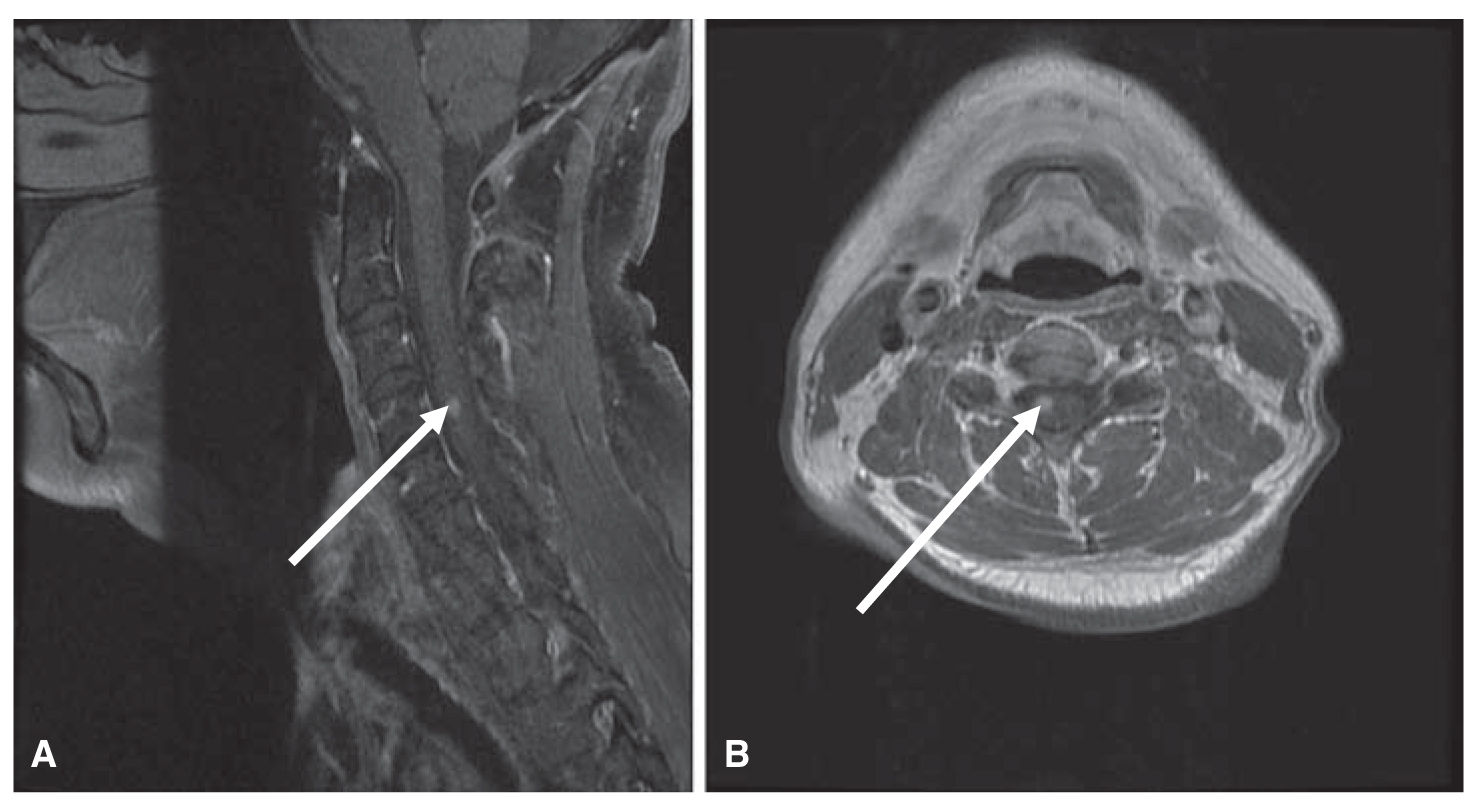
FIGURE 58.3 T1 postcontrast sagittal (A) and axial (B) MRI. These images demonstrate the less commonly encountered parenchymal spinal cord metastasis (arrow) in this patient with non–small cell lung cancer.
2. Spinal cord metastases. The spinal cord is a relatively uncommon location for CNS metastases (Fig. 58.3A and B).
a. Signs/Symptoms. Symptoms would localize to the level of the lesion and would have similarities to other etiologies injuring the cord at the equivalent level. As the metastases may grow without marked invasion of the parenchyma the severity of symptoms may be less pronounced than vascular or traumatic insults to the cord. Spinal cord metastases may also be associated with a syrinx above and/or below the lesion.
b. Management. Data regarding optimal management are limited in comparison to brain metastases. Often concepts better studied in brain metastases are applied to spinal cord metastases. Steroids to diminish edema and the use of focal RT are the modalities that are likely the most frequently employed.
3. CSF and dural metastases (discussed further on) (Fig. 58.4A and B).
a. Signs/Symptoms. Metastases involving the CSF and/or dura can cause direct injury to the spinal cord via compression and/or invasion.
b. Management. Details regarding management of metastases in these anatomic locations will be discussed later. Unique to this location concern for cord compression often leads to the early administration of high-dose steroids to reduce edema within the cord.
4. Extradural metastases.
a. Signs/Symptoms. Extradural metastases encompass metastases involving the vertebral bones and surrounding soft tissue. They can cause symptoms by compression of the cord, again with symptoms being associated with the level of the injury to the cord. There may be associated radicular pain if there is nerve root involvement or focal pain because of bony involvement. This may be exacerbated by percussion of the back.
b. Management. There is randomized data to support the use of high-dose steroids (dexamethasone 10 to 96 mg) in symptomatic cord compression. Patients treated with high-dose steroids had a better ambulatory status at the completion of treatment and 6 months later. There is also data to support surgical decompression followed by RT over RT alone in patients with single symptomatic lesions. The surgical arm was superior on numerous outcome measures. However, the decision for surgical intervention should be made on a case-by-case basis.
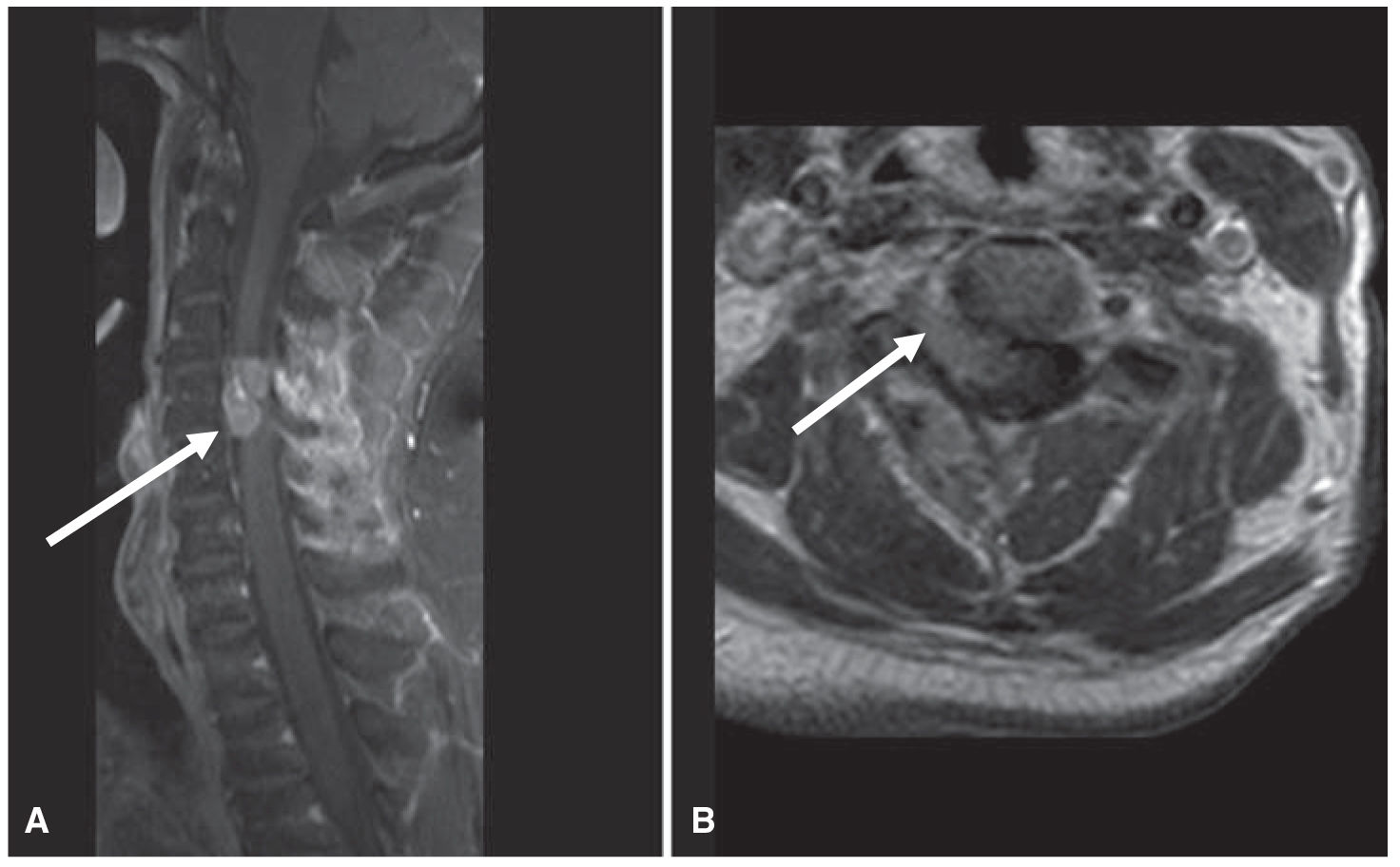
FIGURE 58.4 T1 postcontrast sagittal (A) and axial (B) MRI. An enhancing mass lesion in this patient with metastatic thyroid cancer is seen abutting the cervical spinal cord (arrows). It may be difficult to localize the lesion (cord vs. extraparenchymal) on the sagittal images, whereas the axial images more clearly demonstrate that this is an extradural lesion invading the spinal canal through the neural foramina.
C. CSF metastases.
1. Nomenclature. A number of terms are used interchangeably to describe CSF metastases and include leptomeningeal metastases (LM) and leptomeningeal carcinomatosis (LC).
2. Epidemiology.
a. CSF involvement can be frequently seen with many hematologic malignancies. Breast cancer is the solid tumor with the highest incidence of LM.
3. Clinical presentation.
a. Signs/Symptoms.
(1) Focal/localizable symptoms. Unlike with brain metastases, CSF metastases may manifest more commonly with cranial neuropathies and radiculopathies (including cauda equina syndrome). Often patients may have multifocal signs/symptoms, which are difficult to localize to a single lesion in the neuraxis. Patients may also develop seizures, although this is less frequently seen than in brain metastases.
(2) Nonfocal/localizable symptoms. These are typically secondary to increased ICP and can consist of headaches (worse with lying flat and when first awakening in the morning), nausea, and vomiting. These are analogous to symptoms from increased ICP because of any etiology.
b. Imaging (Figs. 58.5, 58.6A and B). Postcontrast CT or preferably MRI may demonstrate enhancement within the CSF. This can sometimes be seen as enhancement deep in the sulci or between the cerebellar folia. Spine imaging may also reveal enhancement, which may be diffuse or nodular. This may often be present on the surface of the cord or involving the nerve roots. The majority (75% to 90%) of patients with positive cytology will have abnormal MRI findings.
c. CSF studies. Pathologic and cytologic evaluation of CSF is often a key component of the workup for CSF metastases. There are no other definitive biomarkers for diagnosis, prognosis, or prediction of response to therapy.
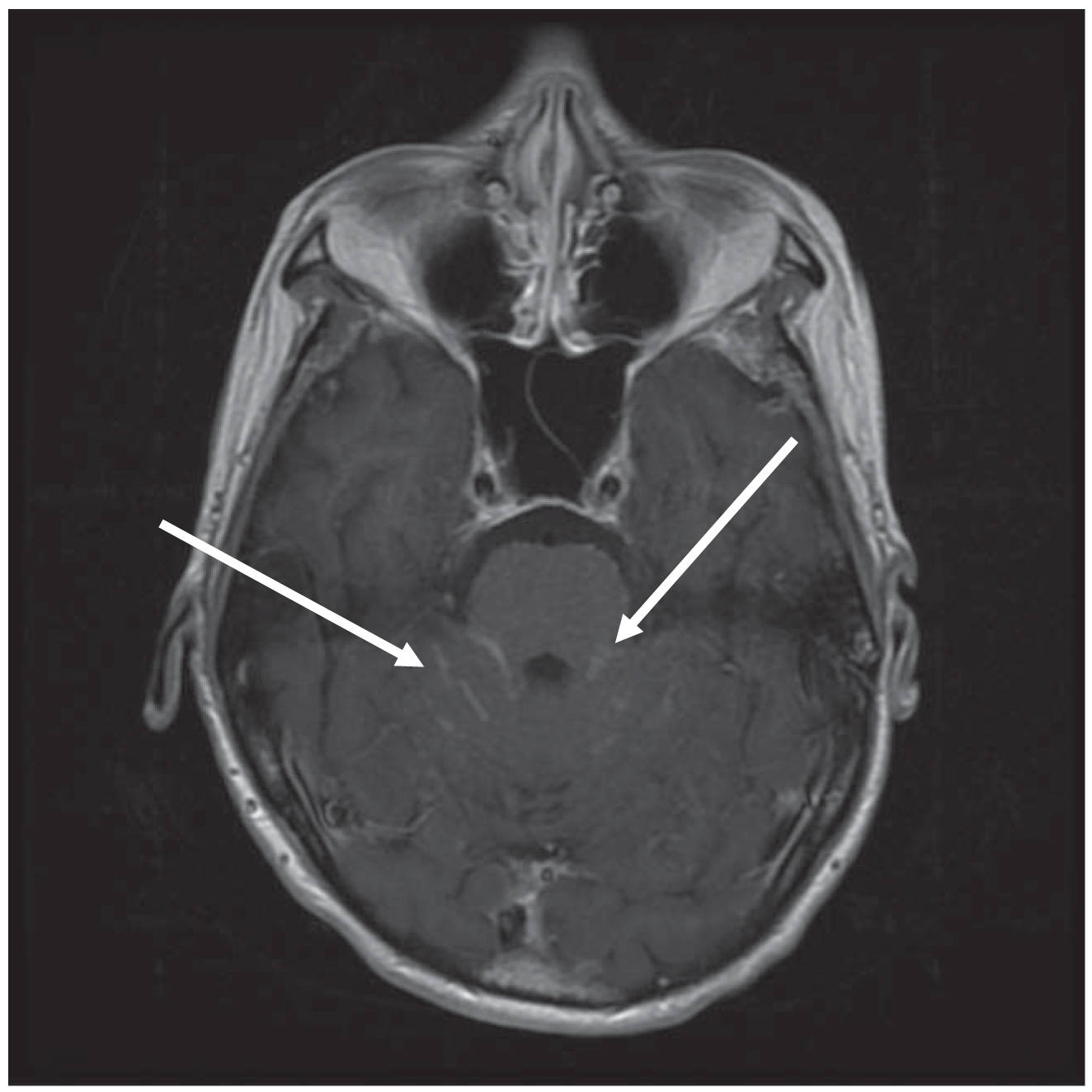
FIGURE 58.5 T1 postcontrast axial MRI. Enhancement between the cerebellar folia and adjacent to the brainstem can be noted bilaterally (arrows) in this patient with LC because of breast cancer. The posterior fossa is a typical location to radiographically detect evidence of LC. LC, leptomeningeal carcinomatosis.
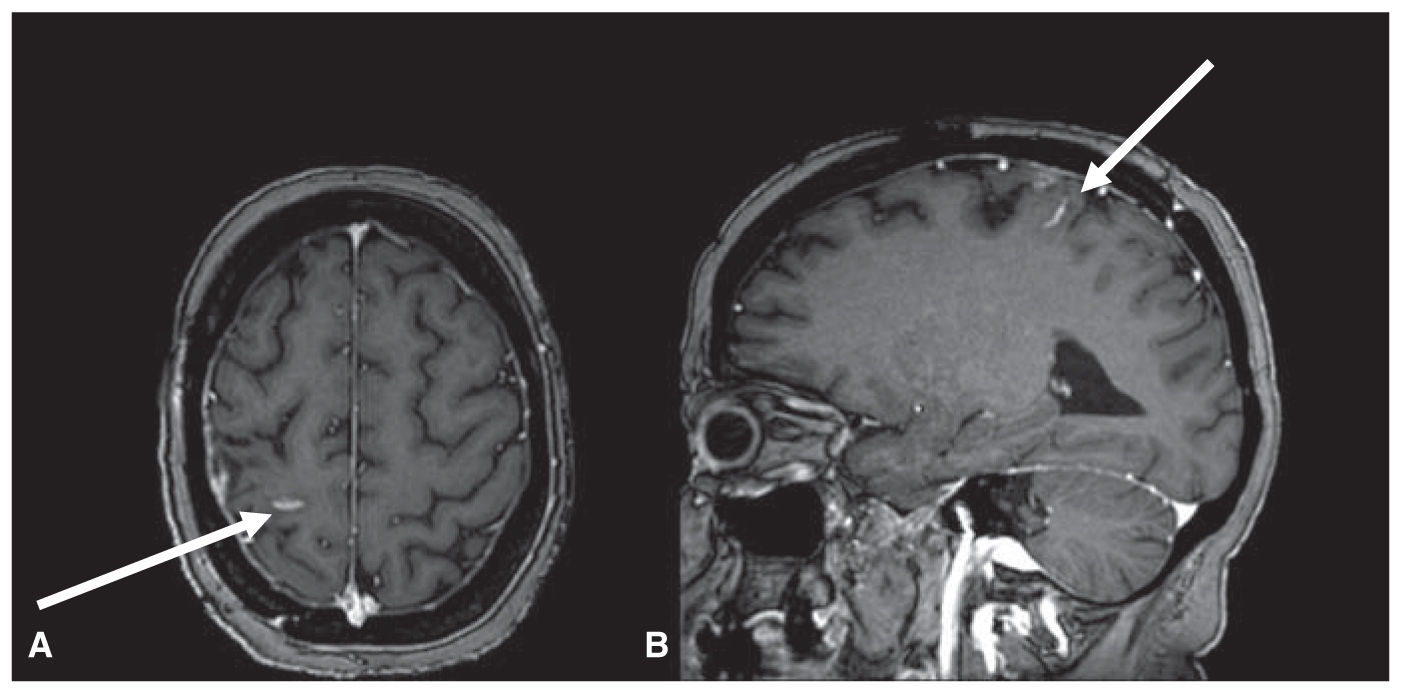
FIGURE 58.6 T1 postcontrast axial (A) and sagittal (B) MRI. Enhancement deep in the sulci is the only radiographic evidence of LC in this patient with breast cancer. Although this is a likely less often encountered radiographic picture in LC than posterior fossa enhancement, it is still relatively frequently seen.
4. Prognostication.
a. The overall prognosis for patients with CSF metastases is poor. Survival is often measured in weeks to months. Clinical progression may be rapid. Patients with breast cancer as the underlying histology may fare best both with respect to the natural history of the disease and response to therapy. Unlike with brain metastases, there are no formal widely employed prognostication systems. Performance status appears to be the most consistent predictor of outcome.
5. Management.
a. Definitive evidence is lacking for an optimal management paradigm for LM. Often the decision making is made on an individualized basis. Do to the overall prognosis hospice is a reasonable management option to offer all patients with LM.
b. Radiation therapy. The most frequently employed radiation regimen is WBRT. This can palliate symptoms and may prolong survival, but it does not address the entire CSF space. Craniospinal radiation, which does treat the entire CSF space, has limitations because of the significant toxicities that can be associated with it, particularly in this often very sick patient population. Additionally, the plan for craniospinal radiation may be further complicated by overlapping radiation fields from prior RT to extra-CNS areas of disease. Focal radiation to bulky disease is often also considered. This may palliate symptoms and has a higher likelihood of decreasing bulky tumor burden compared to systemic or intrathecal (IT) therapies.
c. Systemic therapies. Most systemically administered therapies have not been extensively studied with respect to pharmacokinetics in the CSF. Of those that have, many do not reach adequate concentrations or have adequate therapeutic effects in the CSF. Although there are no data clearly establishing a role for systemic therapies in all patients with LM, responses or stability of disease have been noted in some patients. Often agents with good blood–brain barrier penetration such as lapatinib or temozolomide are considered.
d. IT therapies. Evidence supports more favorable outcomes with administration of IT therapies through an intraventricular catheter such as an Ommaya reservoir as opposed to through lumbar punctures (LPs). The array of drugs available for safe administration into the CSF is limited.
D. Dural metastases.
1. In about half of the patients with dural metastases, they are the only site of intracranial metastases.
2. The most common solid tumors associated with dural metastases are prostate and breast primaries.
3. No definitive management paradigm exists for dural metastases. Surgery, radiation, and systemic therapies may all be considered on a case-by-case basis.
NONMETASTATIC NEUROLOGIC COMPLICATIONS OF CANCER
A. Neurologic complications of cancer secondary to cancer not involving the nervous system.
1. Immune-mediated.
a. Paraneoplastic syndromes. These constitute a heterogeneous group of syndromes affecting the CNS, PNS, or both in the context of cancer. In approximately 2/3 of patients the onset of the paraneoplastic syndrome precedes the diagnosis of the underlying cancer. Establishment of the cancer diagnosis is necessary to appropriately treat the underlying neoplasm and to potentially improve the neurologic symptoms. A wide assortment of malignancies have been associated with paraneoplastic syndromes. The most commonly involved include small cell lung cancer (SCLC), teratomas, and thymomas. Oftentimes serum or CSF paraneoplastic antibodies are present. These antibodies can also be detected in immune-mediated non-neoplasm-related neurologic syndromes. Management of paraneoplastic syndromes includes treatment of the underlying cancer as well as possible immunomodulation. Numerous methods including high-dose steroids, intravenous immunoglobulin, plasma exchange, rituximab, cyclophosphamide, and mycophenolate mofetil have been used with varying degrees of success.
(1) Associated with cell-surface antibodies. Pathogenic antibodies affecting structures on the cell surface include antibodies binding to N-methyl-d-aspartate (NMDA) receptors, α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) receptors, voltage-gated potassium channels, calcium channels, and acetylcholine receptors. Paraneoplastic syndromes associated with cell-surface antibodies are typically more responsive to treatment.
(a) Limbic encephalitis. Patients develop alteration in cognition, memory impairment, personality changes, and may demonstrate seizure activity. Symptoms may wax and wane in severity. This syndrome is most often seen in association with SCLC, germ cell tumors, and gynecologic malignancies. Antibodies to components of the VGKC, NMDA receptor, and AMPA receptor may be detected in the serum and/or CSF. These antibodies and this clinical syndrome can be found independent of an association with an underlying malignancy as well. When these antibodies are detected the patients may prove to be very responsive to treatment. When other antibodies such as anti-Hu (ANNA-1) are present, patients are much less responsive to treatment.
(b) Myasthenia gravis (MG). This is a syndrome of fatigable weakness. Extraocular muscles (EOM) as well as the larger proximal muscle groups are most often involved. This syndrome may or may not be associated with an underlying malignancy. Neurophysiologic studies demonstrate decremental response with repetitive stimulation. The tumor most often associated with this syndrome is thymoma. The majority of patients (in both paraneoplastic and nonparaneoplastic settings) are positive for antibodies to acetylcholine receptors on the postsynaptic neuromuscular junction (NMJ).
(c) Lambert–Eaton mysasthenic syndrome. This is a syndrome of weakness due to dysfunction of the presynaptic NMJ. It is less likely to involve the EOM compared to MG. Neurophysiologic studies demonstrate incremental response to repetitive stimulation. It is most often associated with SCLC. Patients are positive for VGCC antibodies.
(d) Paraneoplastic nerve hyperexcitability syndromes. Patients may develop motor and sensory symptoms including myokymia. They may also develop pain and dysautonomia. These are the result of antibodies targeting the PNS. Isaacs syndrome encompasses this constellation of symptoms. The addition of encephalopathy and sleep disturbance to the syndrome is classified as Morvan’s syndrome. A range of malignancies can be associated with these syndromes. The most commonly associated antibodies are to components of the VGKC ![]() (Video 58.1).
(Video 58.1).
(2) Associated with intracellular antibodies. These antineuronal antibodies (anti-Hu [ANNA-1], anti-Yo [PCA-1], anti-Ri [ANNA-2], amphiphysin, Ma2, CV2/CRMP5, and so on) are almost always associated with neoplasms. Syndromes associated with these intracellular antibodies are less likely to respond to treatment.
(a) Paraneoplastic cerebellar degeneration. Patients develop the subacute onset of ataxia. Cerebellar atrophy is seen on MRI over time. Breast cancer, gynecologic malignancies, SCLC, and Hodgkin’s lymphoma are the most frequently associated cancers. The most frequently detected antibody is anti-Yo (PCA-1).
(b) Opsoclonus-myoclonus. This is a syndrome of irregular involuntary eye conjugate eye movements. Additionally, patients may have myoclonus and ataxia, overlapping somewhat with paraneoplastic cerebellar degeneration. This syndrome is often associated with neuroblastoma, and SCLC. This syndrome is most often associated with anti-Ri (ANNA-2) antibodies. Patients can experience rapid improvement with steroids.
(c) Paraneoplastic brainstem encephalitis. Patients develop subacute onset of signs and symptoms localizable to the brainstem. This can be seen with other nonparaneoplastic etiologies (autoimmune, infectious, and so on). When paraneoplastic in origin, this syndrome is associated with a range of autoantibodies (Hu, Ri, Ma2) and underlying malignancies including SCLC and gynecologic malignancies.
(d) Paraneoplastic parkinsonism (see Chapters 30 and 45). The clinical picture is similar to Parkinson’s disease although the rapidity of symptom onset is quicker. Drug-induced parkinsonism because of antiemetics or antipsychotics should be ruled out before making this diagnosis. This is most often associated with Ma2 antibodies.
(e) Stiff-person syndrome. Patients develop sudden stiffening of the body or parts of the body. They may also develop severe dysautonomia including cardiac arrhythmias. Electroencephalography does not reveal any epileptiform activity. EMG, if performed during the muscle spasms, demonstrates continuous motor unit firing. It is most often associated with breast cancer. This syndrome is most often associated with GAD65 antibody (when not paraneoplastic) and anti-amphiphysin antibody (when paraneoplastic).
(f) Paraneoplastic chorea (see Chapters 28 and 45). Patients develop subacute onset of choreiform movements. They may also develop encephalopathy and ataxia. SCLC, thymoma, lymphoma, and testicular cancer are the most common underlying malignancies. This syndrome is most often associated with antibodies to CV2/CRMP5, less commonly with anti-Hu or anti-Yo.
(g) Paraneoplastic neuronopathy (see Chapter 51). Patients develop subacute sensory deficits and paresthesias. Motor function remains intact. The pattern of sensory symptoms is often patchy and does not correspond to specific peripheral nerves or nerve roots. It is most often associated with SCLC. The majority of patients are positive for anti-Hu (ANNA-1) antibodies.
(h) Paraneoplastic motor neuron disease. The clinical picture has overlap from idiopathic amyotrophic lateral sclerosis. It is most often associated with SCLC and the anti-Hu (ANNA-1) antibodies.
(i) Paraneoplastic myositis (see Chapter 52). Clinically patients can have syndromes consistent with dermatomyositis or polymyositis. These syndromes can be associated with an underlying neoplasm or may be nonparaneoplastic.
2. Vascular.
a. Arterial.
(1) Cancer patients may be on anticoagulation and in turn may be at slightly greater risk of CNS hemorrhagic events. They also may have had prior RT to extracranial blood vessels predisposing them to accelerated atherosclerosis. Depending on the location of said vessels this may increase their risk of stroke.
b. Venous.
(1) Venous sinus thrombosis. Patients with cancer are hypercoagulable and in turn at greater risk of venous clots. Although these most often clinically involve the veins of the lower extremities, they can occur anywhere, including the venous sinuses of the CNS. Venous sinus thrombosis typically requires anticoagulation to decrease the risk of venous infarctions.
B. Neurologic complications secondary to cancer treatment.
1. Secondary to treatment of cancer involving the nervous system.
a. Surgical complications.
(1) The complications of surgery for CNS metastases are similar to complications of surgery for non-CNS metastases indications.
b. Radiotherapy complications.
(1) Involving the CNS
(a) Acutely patients typically develop fatigue. They also develop alopecia in the area radiated.
(b) Patients may develop subacute or delayed (years) areas of increased T2/FLAIR signal abnormality on MRI particularly in the white matter structures. This may be associated with cognitive symptoms including impairment of memory, processing speed, and executive functioning. HA-WBRT and systemic therapies to limit excitotoxicity (as discussed earlier) are methods being investigated to help minimize these symptoms.
(c) Some patients develop radiation necrosis. Onset of this although variable most often occurs 6 to 12 months after RT. Radiographically heterogeneously enhancing lesions are seen. It may be difficult to differentiate radiation necrosis and progressive tumor. Noninvasive modalities such as PET and MRS can be used. Biopsy is sometimes needed to more firmly establish the etiology of radiographic changes. Symptoms from radiation necrosis may be self-limited. However, they may at times require treatment. Steroids are typically the frontline therapy. There is randomized data to support benefit from the anti-VEGF antibody bevacizumab.
(d) RT including the spinal cord in the field can lead to a delayed myelopathy. The risk of myelopathy is dose-dependent.
(2) Involving the PNS
(a) The most frequent RT-related injury to the PNS is likely plexopathy. Often, but not always, RT-related plexopathy is painless. Electromyography may demonstrate myokymic discharges, which are less often seen in plexopathy because of direct invasion of the plexus by tumor.
c. Systemic therapy complications.
(1) As there are no specific systemic therapies definitively employed for the treatment of CNS metastases we will discuss the nervous system toxicities of systemic therapies in the following section (Section B.2 under Nonmetastatic Neurologic Complications of Cancer).
d. IT therapies.
(1) IT administration of drugs can be associated with an increased risk of infection (bacterial meningitis) as well as a chemical arachnoiditis, a pronounced inflammatory response with clinical features similar to a bacterial meningitis but typically lacking a fever. Bacterial meningitis is treated with antibiotics. Chemical arachnoiditis is treated with steroids.
(2) If IT drugs are delivered through LP (as opposed to intraventricular reservoir) there is the potential risk of developing an epidural hematoma, particularly if platelet counts are low.
(3) IT chemotherapy may also predispose patients to the development of leukoencephalopathy, a diffuse injury to the white matter tracts. The risk of this may be higher in specific subsets of patients (elderly, those who have received prior WBRT) as well as with specific agents (methotrexate).
2. Secondary to treatment of cancer not involving the nervous system.
a. Systemic therapy complications.
(1) Seizures
(a) Some chemotherapies may increase the risk of seizures. Prophylactic AEDs are not recommended, however.
(2) Cerebral edema
(a) Some systemically administered chemotherapies such as cisplatin are thought to exacerbate cerebral edema. It is unclear what the exact mechanism of action for this is.
(3) Encephalopathy
(a) Many chemotherapies are associated with both acute and chronic encephalopathies. Agents such as ifosfamide are well known for their potential to cause acute encephalopathy. Chronic encephalopathy (“chemo brain”) has been best studied in breast cancer. It is difficult to parcel out which specific agents contribute to this clinical picture.
(4) Leukoencephalopathy
(a) This describes a radiographic/histologic picture of diffuse white matter injury associated with encephalopathy. It can be a slow insidiously progressive process or it can progress quickly. It has been best described with the use of high-dose methotrexate most often in the setting of primary CNS lymphoma, but can occur with non-CNS tumors as well.
(5) Cerebellar toxicity
(a) The cerebellum is particularly sensitive to toxins. Agents such as high-dose cytarabine can cause acute cerebellar dysfunction. Others such as 5-fluorouracil may be associated with subacute development of symptomatology.
(6) CNS infection (see Chapters 47 and 48)
(a) Patients immune suppressed from systemic therapies are at greater risk for developing infections, including CNS infections.
(7) Cerebrovascular complications
(a) Bevacizumab. This monoclonal antibody targeting VEGF is used in the treatment of high-grade gliomas as well as other cancers. It is associated with an increased risk of arterial and venous clotting events including stroke as well as increased risk of hemorrhage and posterior reversible encephalopathy syndrome.
(8) Neuropathy
(a) Peripheral neuropathy is a common toxicity of a number of systemic therapies utilized to treat cancer.
1) Agents that are associated with a high risk of treatment-related neuropathy include vinca alkaloids (vincristine), taxanes (paclitaxel, docetaxel), platinum agents (cisplatin, carboplatin, oxaliplatin), bortezomib
(b) The most commonly encountered pattern is a symmetric length-dependent neuropathy. Neurophysiologic studies typically demonstrate a pattern of axonal (as opposed to demyelinating) injury. In addition to sensory and motor symptoms patients may have autonomic impairment, which can manifest with a variety of symptoms including constipation.
(c) Patients with preexisting neuropathy or neuropathy risk factors are at greater risk of developing chemotherapy-related neuropathy.
(d) There are no means to prophylax against or treat the underlying neuropathy so management is predominantly symptomatic.
(e) Treatment-related neuropathy often improves slowly after treatment has been discontinued.
Key Points
• Nervous system complications of cancer can be divided into those directly caused by metastases to the nervous system and those which are nonmetastatic neurologic complications of cancer.
• Metastases can often affect the CNS and symptoms are based on the neuroanatomic localization.
• Brain metastases are the best studied of CNS metastases and in turn we have a clearer understanding of prognostication and management.
• Symptoms due to metastases affecting the spinal cord may respond to rapid therapeutic intervention.
• Metastases to the cerebrospinal fluid confer a poor prognosis, and while there exists a range of management options, in general efficacy is suboptimal.
• Nonmetastatic neurologic complication of cancer can either be because of indirect effects of the underlying malignancy or its treatment.
• Extra-CNS malignancy increases the risk of vascular events such as strokes as well as immune dysregulation leading to paraneoplastic syndromes with a myriad of clinical manifestations.
• Focal treatments of cancer such as surgery and radiation as well as systemic treatments of cancer can lead to CNS and PNS symptoms.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








