12/100,000 (adults)
10-70% (adults)
cryptococcus
toxoplasmosis
TB
5-700,000
50%
10-20%
>50%
Meningitis
Meningitis is defined as inflammation of the pia and arachnoid meninges and the cerebrospinal fluid (CSF) that surrounds the brain and spinal cord. The main infectious causes are viral, bacterial and fungal. Meningitis is classified clinically as either acute or chronic. Acute meningitis occurs within hours or days, whereas chronic meningitis evolves over weeks. Acute meningitis is classified as aseptic which is mostly viral in origin or septic or pyogenic which is caused by bacteria.
The term acute bacterial meningitis (ABM) refers to acute infections caused by pyogenic bacteria. The main causes of pyogenic meningitis in Africa are Streptococcus pneumoniae (pneumococcus), Neisseria meningitidis (meningococcus) and Haemophilus influenzae type b (Hib). Chronic meningitis by definition persists for weeks (four or more). The main causes in Africa are cryptococcal infection and tuberculosis. The overall pattern of meningitis in adults has changed in Africa, whereas ABM used to be the leading cause of meningitis, cryptococcus is now the most common cause followed by tuberculous meningitis (TBM) and ABM. Their exact order depends on the geographic location, the extent of HIV epidemic and the age group affected.
ACUTE BACTERIAL MENINGITIS (ABM)
EPIDEMIOLOGY
ABM causes over a quarter of a million deaths globally each year with a large proportion of these occurring in Africa. ABM occurs mostly in young children, particularly in those <2 years but affects all age groups including adults. Africa has some of the highest rates of ABM in the world affecting as many as 1/250 of children <5 yrs, in mainly urban parts of West Africa. However as many as 1-2% of whole populations in the “meningitic belt” may be affected during cyclical meningococcal epidemics which occur every 5-10 years. It is estimated that ABM in Africa is 5-10 times more common in children (24/100,000/yr) as compared to high income countries. Risk factors for ABM in Africa are crowded living conditions, extremes of age, organism virulence and antibiotic resistance, and host predisposition. Individual host factors include HIV infection, malnutrition, sickle cell disease, splenectomy, a non functioning spleen, recent head injury with fracture or, post neurosurgery with CSF leak, middle ear infection and pneumonia. The overall frequency of ABM in adults (10-12/100,000/yr) in Africa appears to have remained relatively constant despite the current HIV epidemic there.
Aetiology
In children in Africa the main causative organisms of ABM are Hib, pneumococcus and meningococcus. In those countries where Hib vaccination has been instituted, Hib has now been replaced as the main cause by pneumococcus and meningococcus. Recently a new pneumococcal conjugate vaccine is being used in children in some countries, including South Africa, Gambia and Kenya. In adults the main causative organisms are pneumococcus and meningococcus. Other less common causes include group B streptococcus, nontyphoidal salmonella, (NTS), staphylococci, and Escherichia coli in neonates. Listeria monocytogenes may cause ABM in pregnancy and in HIV. Gram negative bacilli and salmonella may cause ABM in HIV and in the elderly.
Streptococcus pneumoniae
Pneumococcus is a gram positive coccus which exists in pairs (Fig 6.1) with many subtypes. It affects mostly infants aged <12 months, young children and adults, but all age groups may be affected. It may occasionally occur as epidemics. The main source of meningeal infection is haematogenous spread arising from the respiratory tract (pneumonia) and from otitis media, although individual host factors are also important. Sporadic invasive pneumococcal disease has increased significantly since the arrival of the HIV epidemic and is a significant cause of bacteraemia, pneumonia and death in HIV disease. It accounts for >90% of cases of ABM in adults in the main HIV affected areas in Africa. The main risk factors for ABM have already been outlined above. The case fatality rate (CFR) in Africa in treated pneumococcal meningitis is high, ranging from 30-40% in children to 50-70% in adults.
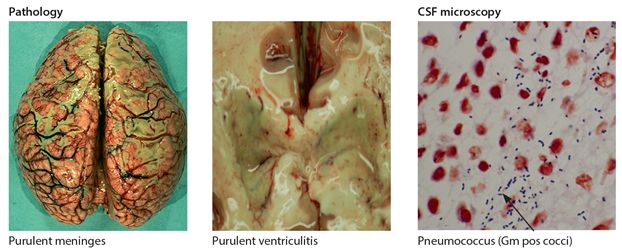
Figure 6.1 Brain/csf in acute bacterial meningitis
Neisseria meningitidis
Meningococcus is a gram-negative diplococcus and infection results in meningococcal disease (fig. 6.2). It is classified into serogroups with A, B, C, Y, W-135 and X predominating. The most common serogroup in Africa is A but there have been recent outbreaks there with serogroups W-135 and X. Epidemic strains are sometimes introduced by Hajj pilgrims returning from Mecca, where similar epidemics have occurred. Protective vaccines exist for serogroups A and C and more recently a quadrivalent vaccine for A, C, Y, and W-135 (meningococcal A-conjugate vaccine) but not for group B or X. The usual incubation period for meningococcal disease is 2-7 days. The peak incidence is in children with a second peak in teenagers and young adults. The main risk factor for infection is close household contact with an infected person, when the risk of contracting the disease is increased a thousand fold. While most cases are sporadic, meningococcal disease also occurs as epidemics in Africa.
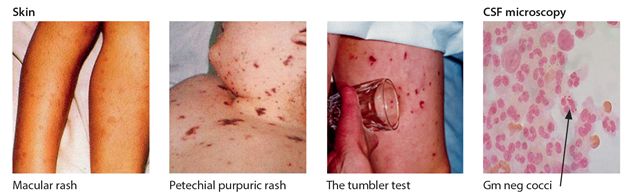
Figure 6.2 Meningococcal meningitis
Large scale epidemics occur in sub-Saharan Africa during the dry season in approximately 10 year cycles. These epidemics occur in a large “meningitis belt” which stretches from the Gambia and Senegal in the West to Sudan and Ethiopia in the north and as far south as Kenya and Tanzania in the east and Nigeria and Ghana in the west. The reason for epidemics is unclear but has been attributed to the loss of accumulated herd immunity and the presence of suitably dry conditions for transmission, usually from March to May. During meningococcal epidemics, outbreaks occur typically in areas of overcrowding such as towns, schools, barracks, and prisons.
The overall case fatality ratio (CFR) in adults is of around 10% but this can vary (5-20%). A lower overall CFR (5%) generally reported across parts of Africa is attributed to infection with the most common serogroup A and to meningococcal disease presenting with mostly meningitis without associated septicaemia. However a higher CFR of around 20% has been reported recently in patients infected with serogroups W-135 and X, and also with HIV infection in South Africa.
Haemophilus influenzae type b (Hib)
Hib is a small gram-negative coccobacillus. It primarily affects young children under the age of six years and is a major cause of respiratory tract infection and ABM. Hib related ABM primarily affects infants 1-24 months and rarely occurs in adults. The CFR in Africa in children is 20-30% and is higher in adults 30-40%.
Key points
- ABM is a major cause of mortality & morbidity in Africa
- occurs in any age group but mostly in infants & young children
- main causes are pneumococcus, meningococcus & Hib
- host risk factors are HIV, sickle cell disease, asplenia & head injury
- meningococcal infection occurs in both sporadic & epidemic forms
- CFR varies with the organism & the age group affected
Pathogenesis
All the three main bacterial causes of meningitis colonise the nasopharynx in asymptomatic carriers. Colonisation rates of around 10-20% are commonplace in schools, universities etc with higher seasonal rates in children, young adults and in case contacts. Spread is by droplets from close physical contact with asymptomatic carriers or occasionally direct from cases. The presence of a lipopolysaccharide capsule helps bacteria survive and they reach the meninges via the bloodstream or by direct invasion. Clinical disease is rare and only occurs when there is penetration across the blood-brain barrier with infection of the meninges and subarachnoid space (Fig. 6.1). This may occur in association with bacteraemia and septicaemia. The multiplication of bacteria in the sterile CSF triggers a massive host immune response with release of inflammatory cytokines, which result in activated macrophages and invasion with neutrophils, immunoglobulins and other markers of inflammation. This leads to a further breakdown in the blood brain barrier and can result in vasculitis, thrombosis, infarction, raised intracranial pressure, brain damage and death.
Clinical diagnosis
The main clinical features of ABM are headache, fever and meningism. When this triad is accompanied by alteration in consciousness or seizures, the diagnosis is usually not in doubt. Other symptoms include photophobia, nausea, vomiting, backache and lethargy. The finding of a haemorrhagic rash on the skin is strongly suggestive of meningococcal infection. Progression occurs rapidly over 1-3 days but a smaller number may have an acute fulminant course lasting hours. However patients with HIV infection may present with only one or two of these main features.
Seizures occur in about one third of patients, typically in children and may be the presenting complaint. Focal neurological abnormalities, status epilepticus and coma occur mainly as complications. There may also be evidence of infection outside the CNS or an underlying condition predisposing to meningitis e.g. pneumonia, HIV, middle ear infection and head injury. The differential diagnosis for ABM in adults in Africa includes the other main causes of meningitis (cryptococcus, TBM and viral), opportunistic infections in HIV, cerebral malaria, viral encephalitis, typhoid fever and other CNS infections.
Signs of meningitis
The cardinal signs of meningitis are neck stiffness and Kernig’s sign (Chapter 1). Neck stiffness is the most important sign and is present when the neck resists passive flexion to bring the chin on to the chest. It is found in most adults and over three quarters of children with ABM. Kernig’s sign is elicited by passively attempting to straighten the leg with the hip and knee flexed to >90 degrees. In cases of meningitis this is met with resistance and pain, caused by spasm in the hamstrings as a result of stretching inflamed nerve roots. A forward flexing of the neck elicits involuntary hip and knee flexion or Brudzinski’s sign. Brudzinski’s sign is found mainly in infants and young children but not in adults.
These signs of meningitis are present in most cases of established meningitis but are less likely to be present early on in the disease and in the young and the elderly. In older children and adults, in addition to the classic features, there may be back pain and myalgia and seizures in around 20%. In infants, the combination of fever, respiratory distress, irritability, crying, vomiting, drowsiness and failure to feed may be the only findings. In babies, the association of bulging fontanel, neck retraction and seizures should prompt the correct diagnosis. In the elderly, alteration in the level of consciousness and fever may be the only clinical findings. It is important to remember that whenever in doubt about the diagnosis of meningitis, to return to re-examine the patient for signs of meningitis, in particular for neck stiffness.
Pneumococcal meningitis
Patients with pneumococcal meningitis present with marked meningism. Signs of an underlying pneumonia and septicaemia may be present particularly in children. Patients tend to progress rapidly in 24-48 hours to drowsiness, confusion, seizures and coma.
Meningococcal disease
The main clinical features of meningococcal disease are those of either septicaemia with or without meningitis or meningitis alone. The proportion of patients presenting with meningitis alone appears to be greater in tropical countries. Meningococcal meningitis without septicaemia has a favourable recovery rate (95%). The clinical features of meningococcal septicaemia may vary from mildly symptomatic patients to acute fulminant infection. The onset is typically abrupt over 24-48 hours. However, symptoms can progress rapidly from drowsiness and rash to circulatory failure, coma and death within hours of onset.
The diagnostic feature of meningococcal disease is the typical haemorrhagic rash, which is non-blanching and present in the majority of patients (Fig 6.2). However it may be absent, particularly in uncomplicated meningitis in children. The rash may begin as a maculopapular rash and develops in a matter of hours into a petechial and purpuric rash all over. The conjunctiva, palate, soles of the feet and palms of the hands should be carefully examined as the rash may be easily missed on the limbs and trunk in dark skin. The lesions do not blanch under pressure and this can be confirmed by pressure with a glass when the rash can be seen to persist. This is called the “tumbler test” (Fig 6.2). Petechiae may later progress to larger confluent purpuric areas called purpura fulminans. Complications of meningococcal disease include skin necrosis, arthritis, gangrene and Waterhouse-Friderichsen syndrome of adrenal failure.
Hib meningitis
This has a characteristic slow onset over several days often starting with fever or respiratory tract infection. The onset of drowsiness, vomiting and convulsions in an infant in this setting may suggest the diagnosis.
Key points
- headache, fever & meningism are the cardinal clinical features of ABM
- neck stiffness is the most sensitive clinical sign
- signs are less sensitive in the young, old & in HIV infection
- meningism & bleeding into skin suggests meningococcal disease
- When in doubt about the diagnosis return & re-examine the patient
Diagnosis
The diagnosis of ABM is based on clinical and laboratory findings (Table 6.2). Laboratory tests include a full blood count, blood glucose, malaria slide, blood culture and an HIV test. A lumbar puncture (LP) is the key investigation and is an overall simple and very safe test (see appendix). It is always indicated in suspected ABM, unless there is a clear contraindication. A LP is contraindicated in the presence of suspected raised intracranial pressure (ICP). The clinical features suggestive of raised ICP in ABM are altered level of consciousness, coma, focal neurological deficit and papilloedema. These are all indications to avoid a LP and also for doing a CT scan of the head if it is available. The CT with contrast may show meningeal enhancement in ABM. If the CT of the head shows no mass lesion and there is no other evidence of raised intracranial pressure e.g. papilloedema then it is reasonable to proceed with the lumbar puncture. However it is important to note that even a normal CT may not necessarily rule out raised ICP, particularly if carried out early on in ABM.
Table 6.2 Summary CSF findings in meningitis*
| acute bacterial meningitis | tuberculous | cryptococcal | viral | |
| opening pressure (n = <20 cm in adults) | increased | increased | increased | normal/increased |
| appearance (n = clear) | cloudy, purulent | yellow/cloudy | clear/cloudy | clear (cloudy) |
| cells/mm3* n = <5/mm3 main type | high >2,000/mm3 neutrophils | increased 50-500 lymphocytes | normal/increased 0-100 lymphocytes | normal/increased 0-500 lymphocytes |
| glucose (n = >50% plasma) | very low/absent <1 mmol/L | low | normal/low | normal |
| protein (n=<0.5gm/L) | elevated 1-2 | high/very high 1-5 | normal/elevated 0.5-2 | normal/elevated 0.5-1.0 |
| diagnosis confirmed | Gm stain & culture | ZN stain & culture | India ink stain, CRAg/culture | PCR/culture |
* see appendix for exceptions
CSF in ABM
The opening CSF pressure is typically elevated >20 cm and the colour is cloudy (Table 6.2). On analysis there is a characteristic high white cell count (>60% neutrophils), a very low glucose and an elevated protein. In HIV patients who are unable to mount a full inflammatory response, a much lower cell count is used as a cut off (>10 cells/mm3) for diagnosis of ABM and any protein elevation is also less. A similar pattern may be seen with the other causes of meningitis in HIV disease. A gram stain should always be performed on the CSF and a specimen sent for bacterial culture. Suspected cases of chronic meningitis patients should have their CSF screened for cryptococcus by India ink and cryptococcal antigen (CRAg) if available and also for tuberculosis by Ziehl-Neelsen (ZN) stain and culture.
Management
The mainstay of management of ABM is prompt diagnosis and early treatment with antimicrobials (Table 6.3). It is important that antimicrobials should be given straight away (within 20-30 mins of first seeing the patient) and not to delay treatment because of ongoing investigations including a LP or CT. The early treatment is based on a presumed diagnosis of ABM and the patient is usually covered with antibiotics for the main possible bacterial causes (Table 6.3). In adults ceftriaxone or another extended-spectrum cephalosporin, cefotaxime are now the drugs of first choice. If unavailable then it is recommended to give soluble penicillin in combination with chloramphenicol. A history of anaphylaxis is a contraindication for penicillin but a history of a rash is not.
Table 6.3 Antimicrobial treatment of adult ABM
| Drug | Dose/ route | Frequency | Duration* |
| ceftriaxone or cefotaxime | 2 gm/iv 2 gm/iv | 12 hourly 4 hourly | 10-14 days 10-14 days |
| or | |||
| penicillin & chloramphenicol | 2.4 gm or 4 million units/iv 1 gm/iv | 4 hourly 6 hourly | 10-14 days 10-14 days |
* children with ABM & meningococcal disease may have shorter courses of antibiotics (5-7 days)
Patients at the extremes of life or with a particular risk factor may need additional antibiotic cover e.g. flucloxacillin for staphylococcal infection in neonates or gentamycin for some gram negatives in neonates and in old age. Additions or changes in antimicrobials are guided by laboratory based bacteriology stains and cultures. The use of steroids in the treatment of ABM in adults is currently not recommended in Africa as evidenced by recent prospective ABM studies in Malawi, showing no additional benefit. Supportive measures include oxygen, careful rehydration at less than 1-2 litres in the first 24 hours, maintenance of normal blood pressure, urinary output, electrolyte balance and control of pain and fever.
Outcome
Death is inevitable in untreated ABM. Mortality in treated ABM varies with the age group affected, the organism causing it and in particular how early on the appropriate antimicrobial was given. The case fatality ratio (CFR) in ABM is highest in neonates and adults (50-70%). In adults in Africa, CFR is highest in pneumococcus (70%) and lowest in meningococcus (10-20%). The lowest CFR in ABM is in children with uncomplicated meningococcal meningitis (5%). The presence of underlying HIV infection significantly increases the CFR in ABM. Resistance to penicillin (20%), chloramphenicol (20%) or both (10%) and a decreased susceptibility to cephalosporins (5%) is an increasing problem particularly in Africa because of their widespread usage. Permanent neurologic deficits persist in over a quarter of all surviving ABM patients. This also varies by age group and organism. Over 50% of neonates and 40% of those who survive pneumococcal meningitis have permanent neurological deficits, in contrast to about 5-7% of those with meningococcus. The main neurological deficits after ABM are hearing loss (>25%), motor loss (12%), cognitive impairment (9%) visual disturbance (6%) and seizures.
Key points
- death is inevitable in untreated ABM
- early antibiotics is the most important treatment affecting outcome
- cephalosporins are the drugs of first choice
- over a quarter of all surviving patients have permanent neurological disabilities
- disabilities includes deafness, motor loss, cognitive impairment, mental retardation, visual disturbance & seizures
Prevention
In sporadic meningococcal infection, chemoprophylaxis should be provided for all household and close contacts of the patient within the previous 24 hours. The risk of developing meningitis in close contacts is estimated to be about 1 in 300. Adults and children over 12 yrs should receive rifampicin 600 mg orally twice daily for 2 days or ciprofloxacin or azithromycin 500 mg orally as a single dose. Rifampicin should not be given in pregnancy. For children up to the age of 12 years, use rifampicin10 mg/kg twice daily for two days or ceftriaxone 125 mg im as a single dose. Chemoprophylaxis is not indicated for close contacts of pneumococcal or Hib meningitis. Early recognition is the key to management when epidemic meningococcal meningitis is suspected. If the number of cases exceeds 15/100,000 per week or 5-10 cases per week if the population <30,000, then emergency preventative measures include alerting the appropriate authorities, identifying the organism and use of mass chemoprophylaxis and vaccination. All three main causes of ABM are now largely preventable by vaccination.
Key points
- prevention of individual cases meningococcal infection is based on prophylaxis of close contacts
- epidemic prevention is based on early recognition, mass chemoprophylaxis & vaccination
- vaccines are available to prevent Hib & for some strains of meningococcus & pneumococcus
- ABM in SSA is largely preventable by vaccination
TUBERCULOUS MENINGITIS (TBM)
Each year there are around 10 million new cases of tuberculosis worldwide, approximately one third of which occur in Africa. TBM now accounts for 8-44% of all cases of meningitis in SSA depending on the local HIV and TB prevalence. TB of the CNS is estimated to account for <1% of all new cases of TB, but this figure is significantly higher, when there is coexisting HIV infection. Tuberculous meningitis (TBM) is the most common CNS presentation. Other CNS presentations of TB infection include focal neurological disorders in intracranial tuberculoma and paraplegia in spinal cord involvement. In Africa, non HIV associated TBM affects mostly children, in particular the age group <5 years but can affect all age groups. In contrast TBM in HIV disease mostly affects adults. TB is clinically classified as pulmonary type (85%) and extra pulmonary type (15%). Only the pulmonary type is infectious to others. TBM can arise in two main ways: either as a complication of pulmonary e.g. disseminated or miliary, or less commonly as a result of reactivation of extra pulmonary TB.
Pathogenesis
Mycobacterium tuberculosis is the main cause of TB but other members of the M. tuberculosis complex such as M. bovis and M. africanum may also cause human disease. TB of the CNS arises indirectly from primary infection in the lungs, from where it spreads via the blood stream to other organs including the brain and spinal cord. In the brain or spinal cord, it has a predilection for the subpial sites, where it may present either acutely as TBM or lie dormant for years and later reactivate. Under different conditions, notably immunosuppression in HIV or sometimes pregnancy these tubercles which are known as Rich foci reactivate and rupture. If they rupture into the subarachnoid space they result in TBM, into the brain a tuberculoma, or into the spinal cord a myeloarachnoiditis. The immune reaction generated is mainly inflammatory with exudates particularly around the base of the brain and in the ventricles (Fig. 6.3). This may lead to multiple cranial nerve palsies, arteritis with strokes and obstruction to CSF flow and absorption resulting in hydrocephalus.
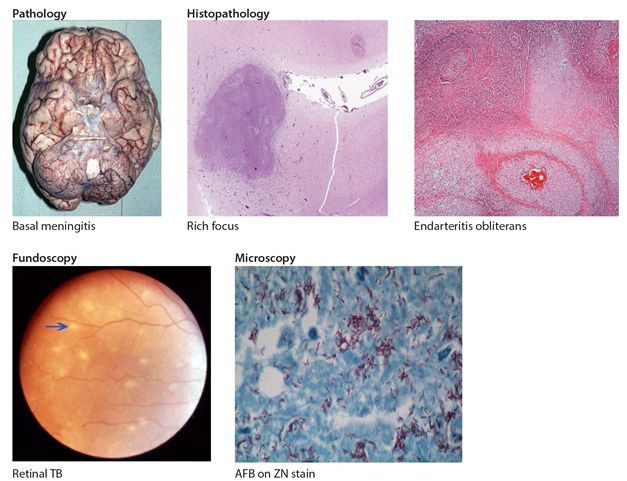
Figure 6.3 Brain/csf in tuberculous meningitis
Clinical features
TBM is a difficult condition to diagnose and confirm clinically. The clinical features are those of slowly progressive chronic meningitis frequently with associated encephalopathy. Symptoms develop gradually, usually over 1-3 weeks but can be more acute in children. Constitutional TB symptoms including fever, night sweats, weight loss and malaise may be present for a week or more early on but these may also be absent or are not specific for TB. The main neurological symptoms suggestive of TBM are headache, nausea, vomiting, irritability, behaviour change and meningism of gradual onset usually for a period of one week or usually longer in adults.
However, headaches may be less prominent in children, fever may be absent in 10-20% of adults and the signs of meningitis are generally less prominent as compared to ABM. The main neurological signs are those of meningism including neck stiffness coupled with combinations of cranial nerve palsies (3rd, 4th, 6 th, 7th & 8th). The presence of focal neurological deficits, visual loss, papilloedema, altered level of consciousness, seizures and coma all suggest either parenchymal brain involvement or hydrocephalus. Fundoscopy may occasionally reveal typical TB retinopathy (characteristic white spots e.g. Fig 6.3). Hydrocephalus may be present early on or develop later during the course of the illness.
Other neurological presentations of TB involving the CNS are tuberculoma in the brain and spinal cord TB. Tuberculoma may be solitary or multiple presenting mainly as focal neurological deficits, seizures and occasionally raised intracranial pressure. Tuberculoma may sometimes complicate TBM and the most common site in adults is above the tentorium (supratentorium), whereas in children it is below the tentorium (infratentorium). Spinal cord TB presents as paraplegia and the site may sometimes be the source of TBM (Chapter 10). A WHO staging based on the main neurological features of TBM is outlined below (Table 6.4).
Table 6.4 WHO Staging of TBM
| Disease Stage | Neurological features |
| Stage I | no disturbance of consciousness no *FND |
| Stage II | alteration in consciousness but not in coma or delirium, no FNDs, cranial nerve palsies may be present |
| Stage III | coma &/or FNDs |
* FND focal neurological disorder
Key points
- TBM occurs either as an acute complication of pulmonary TB or as a reactivation of extra pulmonary TB
- TBM affects mostly young children whereas TBM in HIV affects mostly adults
- HIV significantly increases the risk of TBM
- symptoms of TBM include headache, fever, vomiting, & meningism for >1 week
- signs include neck stiffness, seizures & multiple cranial nerve palsies
Differential diagnosis
The differential diagnosis includes cryptococcal meningitis, partially treated ABM, cerebral malaria, brain abscess and other infectious causes of meningoencephalopathies.
Diagnosis
The diagnosis of TBM is based on clinical suspicion and characteristic CSF findings (Table 6.2). Routine laboratory investigations are of limited value in the diagnosis of TBM. The tuberculin skin test is of little diagnostic benefit in adult populations with high levels of TB infection or previous BCG exposure and may also be negative in disseminated TB and HIV disease. The diagnosis of TBM is supported if there is evidence of concomitant TB elsewhere, most frequently pulmonary as evidenced by chest radiograph. Lumbar puncture is safe if there are no contraindications e.g., alteration in consciousness, lateralising clinical signs or signs of raised intracranial pressure (see appendix).
In TBM the opening pressure is often raised and the CSF clear in colour but may be slightly yellow in established disease. If a sample is left standing overnight in a test tube, the development of an appearance of a cobweb or lattice is supportive of TBM. The CSF white cell count in TBM is usually elevated, 50-500 cell/mm3 mostly lymphocytes, but notably these may be absent in HIV disease or are polymorphs in early infection, particularly in young children. The CSF protein level is usually quite elevated and the glucose is low (<50% plasma glucose) but these can be normal in early disease and also in HIV infection. The organism is identified by acid-fast staining and culture. In TBM, the sensitivity of routine unconcentrated CSF staining with Ziehl-Neelsen stain is very low (<5%), but this yield can be improved markedly with increased quantity of CSF (10-20 ml in adult), by concentrating the CSF by centrifugation and by careful examination or the residue (for at least 20 mins) and by repeated CSF examinations. Polymerase chain reaction (PCR) has better sensitivity depending on bacillary load and good specificity (90-95%) but the test is not widely available in Africa and the result is no better than culture. A new automated PCR test on sputum is now available which gives a result in 4 hours but running cost is approx 20 US dollars per test which makes it relatively prohibitive in most parts of Africa. Also it has yet to be validated on CSF. Culture is the gold standard but limitations include the fact that the result takes 4-6 weeks which is too slow to be of value clinically and this facility is again not widely available. The CRAg test in TBM is negative.
Imaging with CT/MRI (Fig. 6.4) can be very helpful. In TBM it may show evidence of hydrocephalus and after contrast generalised meningeal enhancement with irregular basilar/ cisternal involvement. It may also reveal infarction or tuberculoma. A tuberculoma shows as a rounded lesion with ring enhancement with irregular walls, nodular enhancement, oedema and mass effect. They are most commonly situated near the cortex, may be multiple and accompany TBM.
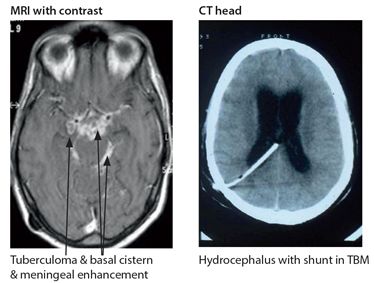
Figure 6.4 Neuroimaging in TB CNS
Key points
- diagnosis of TBM requires a high index of clinical suspicion
- laboratory confirmation is by finding evidence of TB in CSF by ZN stain or culture
- routine CSF screening sensitivity for AFB is very low
- typical CSF findings in TBM are increased lymphocytes, elevated protein & low glucose
- CT findings in TBM include basilar meningeal enhancement & hydrocephalus
Management of TB
Treatment for CNS TB should start as early as possible with 4 drugs as any delay in treatment greatly increases mortality. These include isoniazid, rifampicin, pyrazinamide and a fourth drug ethambutol (Table 6.5). Streptomycin is also available but is a second line drug used when there is drug resistance or toxicity. The four drugs are continued for the first 2 months after which isoniazid and rifampicin are continued usually for another 10 months. In practice the standard total period of treatment is 12 months for TBM and longer for tuberculoma (18 months). A shorter period of treatment for TBM (total 9-10 months) has been proposed but is not common practice in Africa. The main side effects are hepatitis with isoniazid and rifampicin, neuropathy with isoniazid and deafness with streptomycin, and rarely optic neuritis with ethambutol (Table 6.5). Pyridoxine 20-50 mg daily should be prescribed with isoniazid to prevent neuropathy. All HIV uninfected patients with CNS TB in WHO Stages II and III of the disease should be given steroids for the first 6 weeks of chemotherapy. The dose can be decreased gradually after the first two weeks. Hydrocephalus is a major complication of TBM occurring in >50% cases and may require ventricular peritoneal shunting or drainage as early as is clinically indicated.
Table 6.5 Treatment of CNS tuberculosis, TBM
| Chemotherapy | Dose/route/frequency/duration* | Main side effects |
| Isoniazid (H) | 5-10 mg/kg/po/daily/12 months (300-600 mg daily) & pyridoxine 20-50 mg daily to prevent neuropathy | hepatitis, neuropathy |
| Rifampicin (R) | 10-12 mg/kg/po/daily/12 months (600 mg daily) | hepatitis |
| Pyrazinamide (Z) | 30 mg/kg/po/daily for 2 months 1.5-2.0 gm daily | nausea & vomiting & arthralgia, hepatitis |
| Ethambutol (E) or Streptomycin (S)** | 15 mg/kg/day/po/daily for 2 months (800 mg daily) 20 mg/kg/im/daily for 2 months (1 gm daily) | optic neuritis (rare) nerve deafness, nephrotoxicity |
| Dexamethasone or Prednisolone | 0.4 mg/kg/iv/po/daily (24 mg od) for 2 weeks and tapering over next 4 weeks 60 mg/po/daily for 2 weeks and tapering over next 4 weeks | hyperglycaemia, peptic ulcer, hypertension & psychosis |
* a longer course of treatment (18/12) is recommended in tuberculoma
** second line drug
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








