CHAPTER 14 Neurotology
The otolaryngologic subspecialty of otology, neurotology, and cranial base surgery has a focus on the anatomy, physiology, and abnormalities of the three cranial nerves that traverse the temporal bone—the cochlear, vestibular, and facial nerves—as well as specific tests of their functions.1 Surgical management of intractable vertigo is reviewed in Chapter 92; however, the indications for vestibular neurectomy are briefly introduced in this chapter in the context of vestibular assessment and discussion of various vestibular disorders. Facial nerve injury secondary to temporal bone fracture is reviewed in Chapters 338 and 339 and is included in this chapter only in the context of the cochlear and vestibular nerves, although more extensive reviews are offered elsewhere.2–4
Anatomy of the Inner Ear
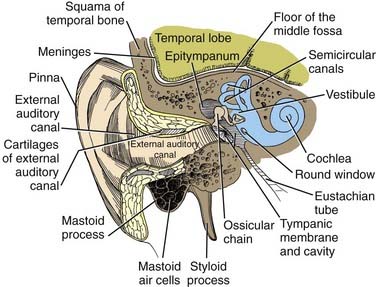
In postnatal infants and adults, the vestibulocochlear labyrinth consists of a continuous complex of bony spaces encased in the otic capsule and within the petrous bone, called the bony labyrinth (Figs. 14-1 to 14-4). It is filled with perilymph (biochemically similar to cerebrospinal fluid), and suspended within it is an almost true, miniature replica of the bony spaces—the membranous labyrinth. The labyrinth is filled with endolymph (see Fig. 14-2). The cochlea is located anteriorly and is divided from the vestibular labyrinth by the vestibular, cochlear, and facial nerves within the internal auditory canal (IAC). The vestibular portion of the labyrinth lies posterior to the cochlea and the IAC (see Figs. 14-3 and 14-4).
The Cochlear System
The basal turn of the cochlea forms a distinct bony promontory on the medial wall of the mesotympanum (i.e., the middle ear) and is the only portion of the cochlea that is visible during middle ear operations. The remaining turns of the cochlea are enclosed within the petrous bone. At the posterior aspect of the promontory are two fibrous barriers to communication with the inner ear: the oval window superiorly, which faces laterally and houses the footplate of the stapes, and the round window inferiorly, which faces posteriorly and is contained within the bony round window niche (see Figs. 14-1 and 14-2).
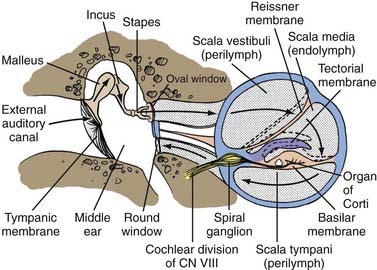
A transverse section through the cochlea shows that it is composed of three compartments: the scala vestibuli, scala media, and scala tympani (see Figs. 14-2 and 14-4). The scala media, which contains endolymph, is separated from the scala vestibuli and tympani by Reissner’s membrane and the basilar membrane, respectively. The scala vestibuli and scala tympani contain perilymph, and these two compartments communicate with each other at the cochlear apex through an opening at the tip of the basilar membrane called the helicotrema. At the base of the upper compartment, called the scala vestibuli, is the oval window, and at the base of the lower compartment, called the scala tympani, is the round window.
The basilar membrane has a complex structure resting on it that is best appreciated in cross section. The basilar membrane and the thin sloping membrane, called Reissner’s membrane, form a tube that ends blindly and is sealed at the helicotrema. This is the scala media, or cochlear duct, and it contains endolymph (see Figs. 14-2 and 14-4). Extending along the entire basilar membrane and spiraling with the cochlea is the organ of Corti. It contains the structurally complex sensory epithelium innervated by the cochlear nerve. As viewed from above, the basilar membrane is widest near the helicotrema and narrowest at the base. Maximal high-frequency vibration of the basilar membrane occurs at the base, and maximal low-frequency vibration occurs at the apex, thereby resulting in hair cell transduction of high frequencies at the base and low frequencies at the apex.
Physiology of Hearing
For an understanding of the various types of hearing loss, it is important to review the mechanism of transmission of sound to the neural receptors (see Fig. 14-2). Air vibrations impinge on the tympanic membrane and cause it and the malleus to vibrate. This physical vibration is transmitted through the incus to the stapes and by way of its footplate to the fluids of the labyrinth. The fluid vibrations produce a stimulus along the basilar membrane that activates the organ of Corti. Impulses are transmitted through the cochlear nerve endings to the cochlear nuclei in the pontomedullary junction. The impulses then continue to the auditory areas of the cortex.
Transformer Mechanism of the Tympanum
Because fluid is incompressible, the round window membrane acts as a compensating membrane to accommodate the vibrations of the stapes footplate (see Fig. 14-2). If sound vibrations were to reach the oval and round windows at the same time, a certain amount of cancellation of the sound would take place. This does not normally take place because of the phase difference between the windows, which is facilitated by several factors. The intact tympanic membrane protects the round window from direct sound impingement; the tympanic membrane is connected to the oval window through the ossicular chain, thus making direct transmission by this route faster; and the round window membrane faces backward, at right angles to the plane of the tympanic membrane, and is recessed within the niche. These factors delay the impingement of sound onto the round window membrane, which produces the phase difference.
Transmission in the Labyrinth
Cochlear signal transduction of high frequencies occurs near the basal portion, and transduction of low frequencies occurs near the apical regions of the basilar membrane and its organ of Corti. Detailed mechanisms of sound transduction and perception are beyond the scope of this chapter but can be found elsewhere.1
Measures of Auditory System Function
Subjective Measures of Hearing
Pure-Tone Audiometry
Air Conduction
Pure-tone threshold hearing sensitivity has developed to be the subjective procedure by which auditory sensitivity is determined. In the United States, the American National Standards Institute (ANSI) has established standards for the calibration of clinical audiometers. The output sound pressure level for standard circumaural or inserted earphones, or both, is specified when measured in a standard coupler, referred to as an artificial ear. The artificial ear simulates the impedance characteristics of the average human ear at the plane of the tympanic membrane. The decibel levels used in audiometers for the normal threshold for air conduction can be found elsewhere.5
Bone Conduction
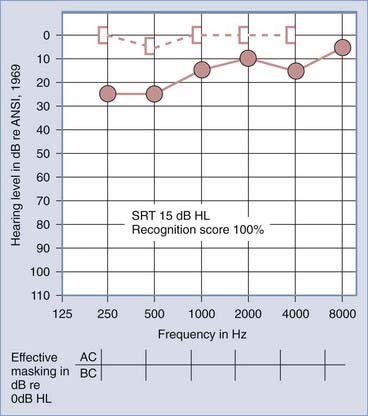
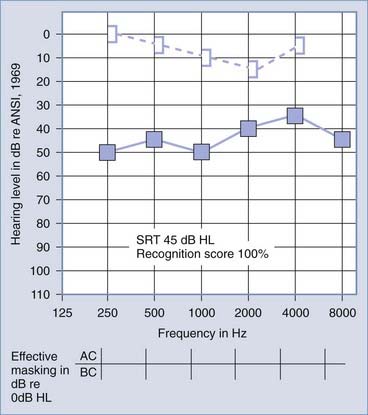
The primary audiologic tests used to distinguish conductive from sensorineural hearing loss are the comparative measures of air and bone conduction thresholds. The procedure for measuring bone conduction thresholds is similar to that for measuring air conduction thresholds, except that a vibrotactile stimulator transduces the signal, usually coupled to the mastoid of the ear under test. The diagnostic utility of the difference between air and bone conduction sensitivity is based primarily on two assumptions: that the air conduction threshold measures the function of the total auditory system, both conductive and sensorineural components, and that the threshold for bone conduction is primarily a measure of the integrity of the sensorineural auditory system and is not significantly influenced by the functional status of the external or middle ear. It has been demonstrated, however, that the external ear and middle ear do provide minor, but important contributions to the bone conduction threshold in the normal auditory system.6 Consequently, some conductive disorders do cause minor, but significant alterations in bone conduction sensitivity because of changes in the contribution of the middle ear to the bone-conducted signal reaching the cochlea. Despite this limitation, the difference between air and bone conduction pure-tone thresholds provides the most definitive indication of the effect of disorders in the external and middle ear on threshold sensitivity. A thorough review of the clinical principles of bone conduction testing is provided by Dirks.7 Examples of conductive and sensorineural hearing loss can be seen in Figures 14-5 and 14-6, respectively. Notice that in conductive hearing loss (see Fig. 14-5), hearing sensitivity by air conduction deviates from normal hearing (0-dB HL) but bone conduction sensitivity is within the normal limits. In sensorineural hearing loss (see Fig. 14-6), hearing sensitivity by both air conduction and bone conduction deviates equally from normal hearing (0-dB HL). Combinations of sensorineural and conductive hearing loss are called mixed hearing loss.
Masking
When a patient has a substantial difference in hearing sensitivity between the two ears, it is necessary to rule out the potential participation of the better hearing ear when testing the poorer hearing ear. Masking is defined by ANSI as the amount by which the threshold of audibility of a sound is raised by the presence of another (masking) sound.8 As early as 1940, Fletcher observed that a restricted bandwidth of frequencies contained within a broadband noise was sufficient to effectively mask a pure-tone threshold.9 Most clinical audiometers contain narrow bands of noise that encompass the critical band of frequencies necessary to mask frequency-specific stimuli.
In some circumstances of severe bilateral conductive hearing loss, it may be impossible to obtain a threshold for bone conduction (or possibly air conduction) without overmasking. Fortunately, as described later in this section, acoustic immittance studies can be performed without regard to “masking dilemmas” and can give additional diagnostic information on the functional status of the middle ear. Studebaker has described the rules for clinical masking in detail.10
Speech Audiometry
Speech Recognition Threshold
Traditionally, the SRT is measured with the use of spondaic words, that is, two-syllable words in which equal stress is placed on each syllable, such as hot-dog, baseball, cowboy, and sidewalk. No standardized method for presentation of the words has been accepted, although practical means for standardization have been suggested.11 The SRT is reported as the decibel HL below which the patient cannot successfully recognize the two-syllable words. It is expected that the SRT will approximately equal the average hearing loss for pure tones in the midfrequency region (500 to 2000 Hz), regardless of the type of hearing loss (i.e., conductive or sensorineural). The SRT has little differential diagnostic significance, except in cases of pseudohypacusis, but it is used to provide a descriptive measure of hearing loss for speech and to confirm the pure-tone air conduction sensitivity measures.
Speech Recognition Measures
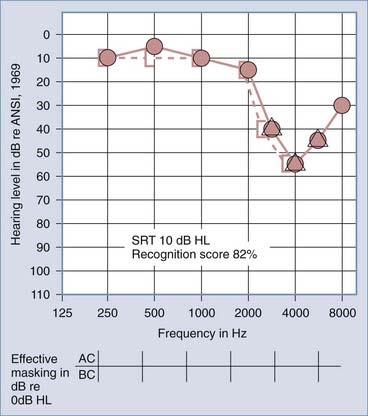
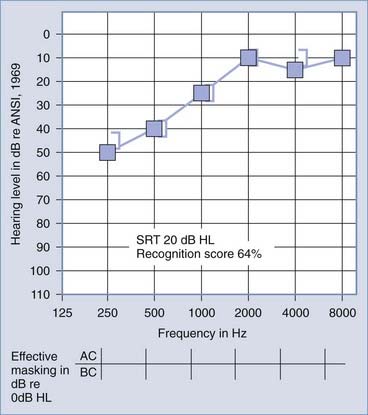
Figures 14-7 and 14-8 provide examples of the effects of conductive and sensorineural hearing loss on the SRT and speech recognition scores of two patients. Figure 14-7 is an example of conductive hearing loss secondary to otosclerosis. Notice the elevated pure-tone air conduction threshold and SRT, together with normal bone conduction sensitivity and excellent speech recognition score. Figure 14-8 is an example of sensorineural hearing loss secondary to Meniere’s disease, which is discussed later. Notice the low-frequency sensorineural hearing loss, the mildly elevated SRT, and the diminished speech recognition score (64%). This example reveals the potential effect of a cochlear lesion site on speech recognition ability. Early in the course of Meniere’s disease, it is unusual for speech discrimination ability to be abnormal. Review of Figures 14-5 and 14-6 reveals other examples in which conductive hearing loss from chronic otitis media (see Fig. 14-5) and sensorineural hearing loss from industrial noise trauma (see Fig. 14-6) affect speech and pure-tone results.
Objective Measures of Auditory System Function
Immittance Studies
Among the most significant advancements in the differential diagnosis of middle ear impairments and one that provides definitive information on cranial nerve VIII and low-brainstem function is measurement of acoustic immittance.12 This procedure requires no active participation by the patient but provides objective evidence of middle ear function and a means of testing the integrity of the acoustic reflex arc. The two procedures included in immittance studies are tympanometry and acoustic reflex measures.
Acoustic Reflex
The acoustic reflex refers to the reflexive contraction of the stapedius muscle on delivery of an acoustic stimulus. The stapedius muscle contracts reflexively and bilaterally on presentation of an acoustic stimulus.13 The muscle contraction results in a concomitant increase in impedance in the middle ear when measured at the plane of the tympanic membrane. Unfortunately, when middle ear pathologies introduce changes in middle ear impedance, it is not possible to measure evidence of further changes in impedance that might be introduced by contraction of the stapedius muscle. Only when tympanometry reveals the middle ear system to be functioning normally is it possible to test the integrity of the acoustic reflex arc.
Constant air pressure is maintained in the external auditory canal, and impedance or admittance is monitored over time. The intensity of a reflex-inducing acoustic stimulus is increased until a change in impedance or admittance is observed. The lowest intensity at which the reflex-inducing acoustic stimulus results in a change in acoustic impedance or admittance is specified as the acoustic reflex threshold. Typically, lesions in cochlear sites produce a change in the threshold of the acoustic reflex only for wide-band noise stimuli, not for pure-tone stimuli, until the hearing loss exceeds approximately the 60-dB HL. When the hearing loss is of cochlear origin and the loss exceeds 60 dB, there may be an increase in the threshold of the acoustic reflex even for pure-tone stimuli. The acoustic reflex threshold measure can be used in cases of sensorineural hearing loss to provide differential diagnostic information on the site of the sensorineural hearing loss.14
In patients with a retrocochlear site of the lesion (cranial nerve VIII and low brainstem), the acoustic reflex may be elevated or absent. An abnormally elevated or absent acoustic reflex in the presence of hearing loss at less than a 60-dB HL is audiologic evidence supporting a retrocochlear site of the lesion.15,16
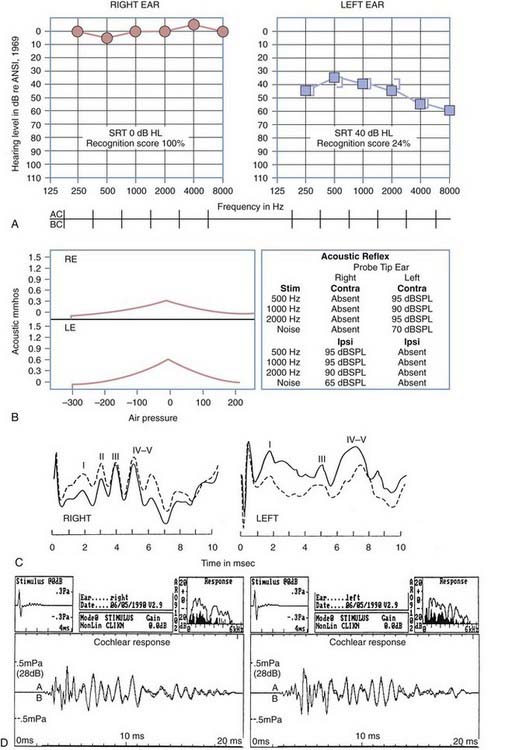
Figure 14-9 is an example of audiometric data from a patient with a left acoustic neuroma within the cerebellopontine angle. The pure-tone results reveal mild, left ear sensorineural hearing loss with a very poor speech recognition score (24%). The tympanograms were normal bilaterally, but there was no acoustic reflex identifiable with acoustic stimulation of the left ear. When the measuring tip was in the right ear, evidence of stapedius muscle contraction was observed only with ipsilateral stimulation. When the measuring tip was in the left ear, the stapedius muscle contracted only when the acoustic stimulus was presented contralaterally. As evidenced by acoustic reflex measures, this is the classic audiometric result seen in a patient with a left acoustic neuroma.
Auditory Brainstem Evoked Response Measures
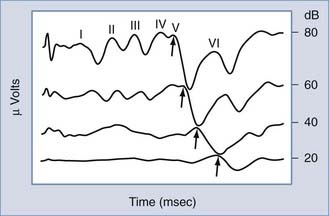
Possibly the most powerful audiologic test available today in differentiating between cochlear and retrocochlear lesions is measurement of the auditory brainstem evoked response (ABR). The ABR is one of several clinically useful evoked auditory potentials and the one most often applied in site-of-lesion testing.17 The ABR is typically evoked with a short-duration pulse delivered to the ear at a predetermined intensity. At high-intensity levels, the acoustic stimulus evokes as many as five amplitude peaks. The peaks were first identified and categorized by Jewett and Williston.18 Three of these peaks (waves I, III, and V) are the major peaks and are generally accepted as corresponding to firing of the first-order neurons of cranial nerve VIII (wave I), the superior olivary complex (wave III), and the inferior colliculus (wave V). These three major waves are present at approximately 2-msec intervals (in normal-hearing children and adults) after the onset of the acoustic stimulus at high-intensity levels (Fig. 14-10). As revealed in Figure 14-10, as the intensity of the stimulus is decreased, the amplitude of all peaks decreases, the latency of each peak increases, and the replicability of the early waves (I and III) decreases. Only wave V is identifiable at threshold levels.
Starr and Achor were among the earliest to use ABR results to describe a diverse set of patients with neurological disorders.19 Patients with cortical problems had normal ABR values, whereas patients with acoustic nerve and low-brainstem disorders had abnormal ABR results. This early evidence has been corroborated by numerous other reports in which ABR values were abnormal in patients with cranial nerve VIII and low-brainstem disorders.20,21
Auditory Neuropathy
Results from auditory evoked potential recordings combined with otoacoustic emission (OAE) testing provide objective measures that can identify patients with auditory neuropathy. The term auditory neuropathy, coined by Starr and colleagues,22 has been used to describe a group of patients with abnormal neural function demonstrated by absent or abnormal ABR results and absent middle ear reflexes but with normal outer hair cell function determined by normal OAE testing and cochlear microphonics. These patients also show evidence of poor speech discrimination, particularly in the presence of noise. Pure-tone thresholds vary widely in severity from normal to severe to profound and may be asymmetric or have a variety of configurations. Doyle and coworkers described the audiometric and electrophysiologic findings associated with auditory neuropathy.23 The results obtained after performing ABR and OAE testing suggest an abnormality of the auditory system at the level of the inner hair cells, at the synapse between hair cells and the cochlear nerve, at the level of the cochlear nerve itself, or a combination of these sites.
The ABR results in patients with cochlear hearing loss typically reveal normal ABR replicability and latencies at high-intensity levels, but with an elevated ABR “threshold” (a function of the elevated hearing thresholds secondary to cochlear pathology). In patients with a retrocochlear site of a lesion, the site of the auditory lesion affects the results. If a space-occupying lesion is present in the brainstem but after the first-order neurons of cranial nerve VIII, wave I may be normal, but all subsequent waves may be absent or significantly delayed in latency. If the lesion affects function of the first-order neurons of cranial nerve VIII, there may be no replicable waveforms evoked by the acoustic stimulus. Figure 14-9 presents the ABR results in a patient with an acoustic neuroma within the left cerebellopontine angle. The ABR result at a 75-dB normalized HL in the right ear reveals a replicable waveform with normal absolute and interwave latencies. The response from the left ear reveals poor replicability of waves III and V, increased latency of waves III and V, and consequently, abnormal I-III and I-V interwave latencies.
Electrically Evoked Auditory Potentials with Cochlear Implant Users
Electrically evoked auditory potentials have been studied for a number of purposes, including assessment of neural integrity, evaluation of cochlear implant function, and estimation of the psychophysical measures needed to program the cochlear implant speech processor, as well as an indication of performance after cochlear implantation.24–26 Preimplantation electrical recordings have included placement of a transtympanic needle electrode on the promontory and subsequent delivery of electrical current. In general, these results have been variable, and there has not been a clear relationship between these measures and postimplantation performance.27 Electrical auditory brainstem response (EABR) has been reliably recorded through a variety of cochlear implant devices and arrays. In general, four peaks can be identified, with the most robust being wave V. Wave I is usually obliterated by the stimulus artifact that occurs at the beginning of the recording. The EABR is affected by stimulus intensity, with a decrease in wave V amplitude and a slight increase in latency as the electrical current is decreased. On average, the latency of wave V is approximately 1.0 to 1.5 msec earlier than after the corresponding acoustic signal. Because the age at cochlear implantation has steadily decreased (now as young as 6 months of age), there is increased interest in the use of EABR to determine the threshold for each electrode and thereby assist in programming and fitting of the external speech processor.28 Several studies have compared the EABR wave V thresholds and behavior levels used in fitting of the speech processor. EABR wave V thresholds generally fall within the dynamic range for a given tested electrode and represent levels that are at least audible.24 Thresholds, amplitudes, and waveform morphologies differ across subjects and within individuals for different electrodes.24,25 The variations may be caused by differences in the population and pattern of surviving primary afferent neurons and dendrites within and across subjects.
Otoacoustic Emission Measures
Kemp was the first to report the presence of audiofrequency energy in the ear canal of subjects with normal hearing who were stimulated with a short-duration, broadband acoustic signal.29 Kemp identified these “emissions” as energy leakage from normal stimulation of cochlear structures. Since the early reports, evidence has accumulated confirming that such acoustic energy leakage is a biochemical property of the healthy, functioning cochlea.
Since identification of the phenomenon in 1978, research interests have focused on sources of the OAE30 and its clinical applications.31 OAE has been determined to be a product of the outer hair cells of the cochlea. Clinical evidence suggests that patients with thresholds of greater than 30-dB HL because of a cochlear-site lesion have no identifiable OAE to transient auditory stimulation. Any middle ear lesion typically precludes measurement of OAE. However, in the case of a retrocochlear lesion, when there has been no retrograde degeneration of the outer hair cells, normal OAE can be evoked in the presence of significant sensorineural hearing loss.31 OAE represents the first available auditory function test with which it may be possible to differentiate neural from cochlear sites of a lesion when the potential exists for each site to be implicated in sensorineural loss. As a consequence, OAE results may contribute information leading to more informed decisions by surgeons who need to address the potential for “hearing preservation” after excision of space-occupying lesions affecting cranial nerve VIII.
Figure 14-9D includes results from OAE measurements for the patient with an acoustic neuroma within the cerebellopontine angle presented earlier in this chapter. The results were obtained with a click delivered at an 80-dB sound pressure level. Results of the average OAE can be analyzed in terms of the replicability of the emission as a function of time after delivery of the emission-evoking stimulus (see Fig. 14-9D, lower section). The OAE response can be collapsed over time, and Fourier analysis of the energy provides a measure of the relative amplitude of the energy in the OAE as a function of the frequency (see Fig. 14-9D, upper right section). Noise in the external ear canal is represented by the dark spectrum; OAE energy is represented by the light spectrum. Despite left ear sensorineural hearing loss exceeding a 35-dB HL throughout the frequency range of 250 to 8000 Hz, there is OAE energy throughout the frequency range of 1000 to 4000 Hz. This observation reveals that the outer hair cells of the cochlea are functioning normally and that the source of the sensorineural hearing loss is neural, not cochlear. Postoperative measurements of OAE can reveal any changes in cochlear function caused by surgical intervention to remove the acoustic neuroma.
The Vestibular System
Anatomy
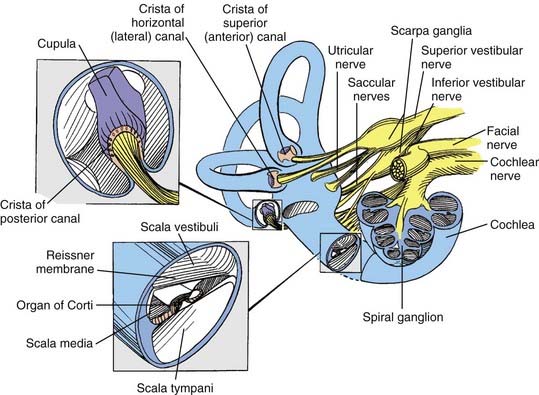
The bony vestibular labyrinth consists of an expansion that is continuous with the basal turn of the cochlea, called the vestibule, and three semicircular bony canals oriented at right angles to one another, each representing a dimension in space: a horizontal (lateral) canal, a superior (anterior) canal, and a posterior canal (Fig. 14-11; see also Fig. 14-4). Each canal originates from the lateral aspect of the vestibule and has its return insertion into the vestibule at a more medial location. The horizontal semicircular canal has its own insertion, but the vertical canals merge into a common crus before joining the vestibule (see Fig. 14-4). Near the origin of each canal is a bony expansion known as the ampulla.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree