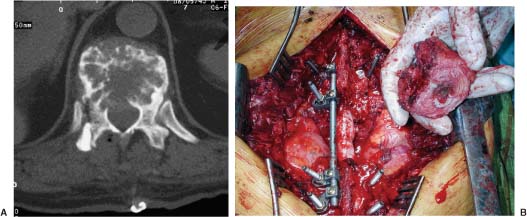
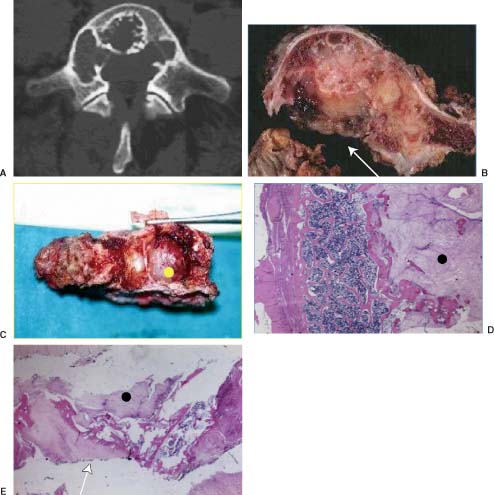
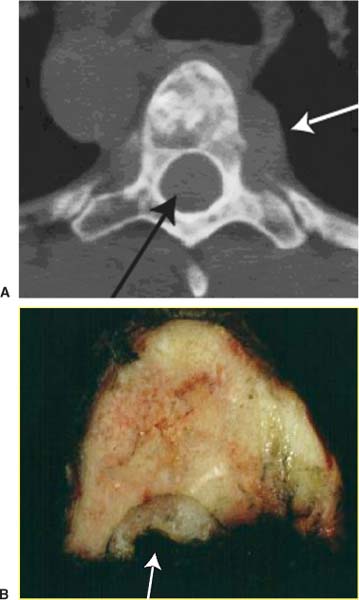
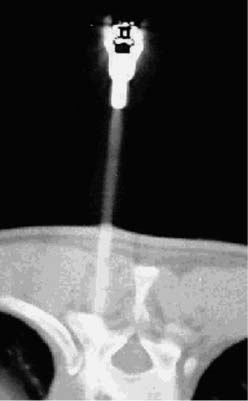
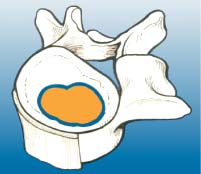
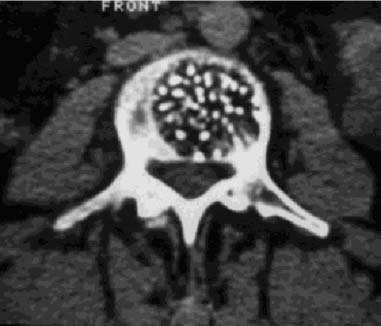
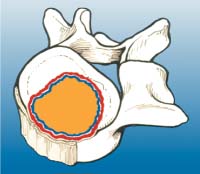
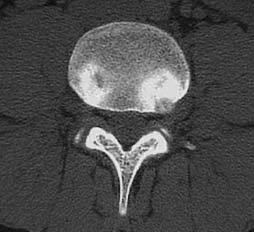
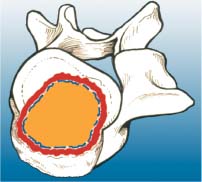
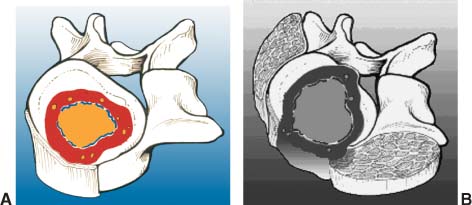
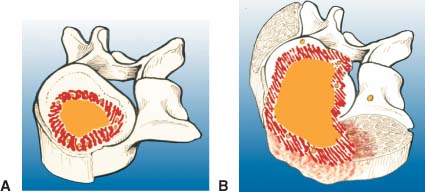
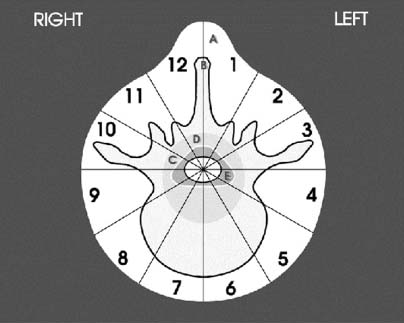
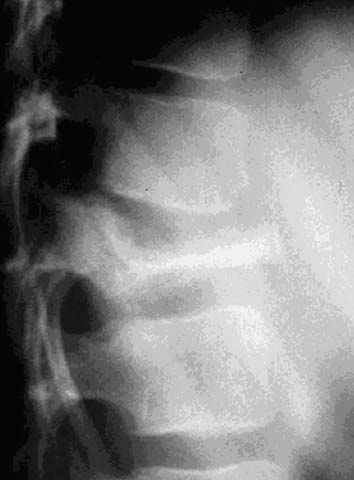
2 Primary bone tumors of the spine are rare, often unsuspected, and can be misdiagnosed. In a series of 1971 patients with musculoskeletal neoplasms followed over a 17-year period, only 29 patients (1.5%), including eight children, had primary osseous tumors of the thoracic and lumbar spine.1 Of 43,735 primary bone tumors,1–6 1851 (4.2%) were located in the spine. Compared with primary spinal tumors, metastatic tumors to the spine are much more common. A 20 to 70% incidence of spinal metastasis has been reported among common solid tumors. The importance of clinical problems related to spinal metastases is revealed by the incidence of paraplegia in the population (8.5 per 100,000) compared with traumatic spinal cord injuries (three to five per 100,000).7 Technically, the main concerns in the treatment of a spinal metastasis are to preserve mechanical and neurologic function. An oncologically appropriate treatment (based on early diagnosis and staging) can substantially improve the prognosis of patients with primary bone tumors. The most common symptom of a spinal tumor is pain. Pain is a nonspecific complaint, but pain associated with a tumor tends to be progressive, unrelenting, and unrelated to activity, and it increases during the night. Pain has several causes. Tumor growth may cause the bony cortex of the vertebral body to expand, resulting in pathologic fracture and invasion of paravertebral soft tissues. Pain is also associated with acute or chronic compression of the spinal cord, resulting in focal and radicular symptoms of pain, paraparesis, or paraplegia. Bowel and bladder dysfunction can be ominous signs. Spinal deformity also may be linked with spinal tumors. A spinal neoplasm must be suspected in adults when these symptoms are observed. Consequently, a complete radiographic evaluation must be obtained. In children, pain is not always expressed, but a change in personality, lethargy, instability, fever, a limp, and bruising are signs that must be investigated. Plain radiographs are the first step in the search for a spinal tumor. Anteroposterior (AP) and lateral radiographic views often help determine the nature and behavior of a lesion and can help identify specific tumor types. Bone scans are good for locating the site of a tumor when standard radiographs are negative. A bone scan should be the first examination when night pain is unresponsive to common analgesic medications. Further, radiography can distinguish between monostotic and polyostotic processes and can help locate the most accessible biopsy site in polyostotic disease. In the spine, computed tomography (CT) and magnetic resonance imaging (MRI) are probably the most helpful diagnostic tools for early detection of a tumor. Likewise, for surgical planning and perioperative planning, CT is often the most beneficial imaging modality.8 MRI has replaced myelography and is a useful tool for the diagnosis and treatment of spinal neoplasms. MRI is often used as the primary diagnostic imaging tool, whereas CT is often an adjunct to planning. Magnetic resonance imaging can help classify spinal tumors by location as intramedullary, intradural extramedullary, extradural, and osseous. Tumors can be located in more than one of these sites and in more than one site in the spine, particularly when metastatic disease involves the vertebral bodies and extends into or throughout the extradural space. Intramedullary spinal cord tumors are especially easy to identify by MRI. Involvement of the bone and its constituent parts can be more challenging. The microstructure of bone marrow consists of a bone framework surrounding fat cells, hematopoietic cells, reticulum cells, nerves, and vascular channels. MRIs of the bone marrow depend on the chemical composition of the marrow. Red marrow is ~40% water, 40% fat, and 20% protein. Yellow marrow is 15% water, 80% fat, and 5% protein. T1-weighted images optimize the signal from fat and provide excellent anatomic detail of the bone. Processes that replace fat in the bone marrow are best visualized on T1-weighted images.9 T2-weighted images help characterize abnormal tissue. Pathologic processes that increase the amount of free water demonstrate increased activity on T2-weighted images compared with normal fatty marrow. Disease processes that infiltrate or replace the red or yellow marrow can be delineated accurately on standard T1-weighted and T2-weighted images. Such processes include benign and malignant tumors, leukemia, lymphoma, myeloma, infection, marrow abnormalities with degenerative disease, and avascular necrosis.10 MRI also helps distinguish between benign and malignant processes associated with vertebral compression fractures. Likewise, it is an excellent tool for delineating vertebral and paravertebral extensions of tumor and can help surgeons to plan surgical interventions, margins for resection, and ports for radiation therapy. Depending on the types of resection and instrumentation used, MRI can be extremely helpful in detecting tumor recurrences. The purpose of a biopsy is to obtain a specimen of the tumor that is representative of the lesion and large enough to allow histologic and ultrastructural analysis as well as immunologic stains. The surgeon must be able to recognize the vital part of the tumor and distinguish it from the necrotic or reactive part. Three traditional forms of spinal biopsy are available: excisional, incisional, and needle biopsy. Excisional biopsy is considered only for conditions whose radiographic pattern is pathognomonic (e.g., osteoid osteoma). Most lesions of the spinal column require an incisional or needle biopsy. Needle biopsies are subject to sampling errors and provide small specimens for evaluation; better results can be achieved by using a trocar under CT control (Fig. 2-1). The overall incidence of inadequate or inappropriate tumor biopsy that significantly alters a patient’s care is greater than one in three11 and may be even higher with spinal lesions. The reasons are clear: it is rarely possible to excise the biopsy track during the excision of a spinal tumor. Being able to excise the biopsy tract during definitive surgery is the first rule of a well-planned biopsy. In the spine definitive surgery is possible only for some posterior tumors. What is unique to the spine is the epidural space, which is an extracompartmental area that should never be contaminated. Biopsy-linked risks can be reduced significantly when the biopsy is performed by the treating physician rather than by the referring physician. Cultures also may be obtained to rule out infection. When the differential diagnosis is narrow and limited to lesions that are easily distinguished histologically, a needle biopsy may be ideal. Specimens obtained from complex lesions and those from lesions with a challenging differential diagnosis most often prove inadequate. An incisional biopsy should be the last step in staging the patient, performed just before the definitive surgical resection. Both procedures may be performed under the same anesthetic setting if proper preoperative staging has been completed and the frozen section provides a clear diagnosis. The surgical technique used during biopsy influences both the yield of the procedure and the risk of postoperative complications. Several basic principles should be observed when performing a biopsy. The biopsy incision should be placed so that it can be excised with the tumor during the definitive procedure. Unlike limb tumors, this principle has not been the standard for spine tumor surgery. Transverse incisions and flaps should be avoided. The tumor should be approached in the most direct manner possible. Tissues should be handled carefully, and hemostasis should be meticulous. Bone should not be removed or windowed unless absolutely necessary. Bleeding from exposed bone or from uncauterized vessels and injured muscle will form a postoperative hematoma that can carry tumor cells beyond the margins of the intended excision and contaminate tissues proximal or distal to the primary lesion. Similar to limb surgery, tissue contaminated during the biopsy or by hematoma must be excised if surgical control is expected. Even a moderate-sized hematoma can make this goal impractical. Once a tumor is exposed, an adequate sample of tissue must be obtained. FIGURE 2-1 Trocar biopsy under computed tomography (CT) control. The patient is placed prone and local anesthesia is used. The trocar is a 2- to 3-mm needle capable of harvesting an appropriate amount of tumoral tissue for analysis. CT control permits the core of the tumor to be reached to obtain representative material. The epidural space must be respected to avoid tumoral contamination of this extracompartmental region. The specimen obtained should be large enough to allow histologic and ultrastructural analysis as well as immunologic stains. The margin of the soft tissue mass is often the most helpful to biopsy because central portions are frequently necrotic. The surgeon should maintain the architecture of the specimen and avoid crushing or distorting it. If a soft tissue component exists, a frozen section should be obtained. Finally, if the definitive excision is to follow the biopsy under the same anesthetic, all instruments used during the biopsy must be discarded. The field should be re-draped, and the surgeons should change gowns and gloves before the excision is begun. If fusion is planned, the bone graft should be obtained through a separate surgical setup. Sometimes biopsies are not indicated for primary spine tumors. Radiographic and other staging procedures often allow the surgeon to perform the definitive surgical procedure at one sitting (e.g., osteoid osteomas or select chondrosarcomas). The many hazards of biopsy can be avoided, and the opportunity to adhere to the principles of good tumor surgery is enhanced. Enneking’s classification is widely accepted for the study and treatment of bone tumors of the limbs; however, the same procedure (intralesional excision) used to treat long bones would not be the same as the one used to treat all spinal tumors. Spinal tumors are often resected without regard to the standard surgical principles applied to the treatment of almost all extremity tumors.12 However, the techniques of vertebral en bloc resection are well known, at least for the thoracolumbar spine,13–18 and these principles should be followed when possible. The lack of a logically organized classification makes the evaluation of results of treatment for primary spinal tumors confusing. Without a common terminology,19 applying the surgical principles of the Enneking’s oncologic staging system to the spine is difficult.20,21 The following terms are defined as used here: All surgical procedures directed toward a functional purpose (spinal cord decompression, fracture stabilization), with or without partial (piecemeal) removal of the tumor, are considered palliative. Palliative procedures are intended to obtain a diagnosis, to decrease pain, and possibly to improve function. The terms vertebrectomy (i.e., removal of all the elements of the vertebra), corpectomy, and somectomy (i.e., removal of the vertebral body) have no significance from an oncologic perspective if they are not qualified by the above-listed terms. Oncologic staging, as proposed by Enneking and coworkers,20,21 defines the biologic behavior of primary tumors and correlates with tumor aggressiveness, type of surgical resection (intralesional, marginal, wide or radical), and outcome (i.e., 5-year survival or recurrence rate) for limb lesions. This staging system divides benign tumors into three stages (S1, S2, S3) and localizes malignant tumors into four stages (IA, IB, IIA, IIB). Two further stages include metastatic high-grade intra- and extracompartmental malignant tumors (respectively, IIIA and IIIB). This classification is based on clinical features, radiographic patterns, CT and MRI data, and histologic findings. It has been described for long bone tumors and applied to spinal tumors in some reports.23–26 The first stage of benign tumors (S1, latent, inactive; Fig. 2-2) includes asymptomatic lesions bordered by a true capsule, which usually appears as a sclerotic rim on plain radiographs. These tumors do not grow or grow very slowly (Fig. 2-3). No treatment is required unless palliative surgery is needed for decompression or stabilization. Benign tumors stage 2 (S2, active; Fig. 2-4) grow slowly and cause mild symptoms. The tumor is bordered by a thin capsule and a layer of reactive tissue (Fig. 2-5), sometimes seen on plain radiographs as an enlargement of the tumor outline. Bone scans are positive. An intralesional excision can be performed with a low rate of recurrence.23 The incidence of recurrence can be lowered further by local adjuvant treatment (e.g., cryotherapy, embolization, radiation therapy). The third stage of benign tumors (S3, aggressive; Fig. 2-6) is composed of rapidly growing benign tumors. The capsule is very thin, discontinuous, or absent. The tumor invades neighboring compartments, and a wide reactive hypervascularized tissue (pseudocapsule) is often present, sometimes permeated by neoplastic digitations. Bone scans are strongly positive. Boundaries are fuzzy on plain radiographs. CT scans show an extracompartmental extension (Fig. 2-7A), and MRI clearly defines a pseudocapsule and its relationship to neurologic structures. Intralesional excision (curettage), even if augmented by radiation, can be associated with a significant rate of recurrence.27,28 En bloc excision is the treatment of choice (Fig. 2-7B). FIGURE 2-2 Illustration of a stage 1 benign tumor (so-called latent or inactive). The tumor is represented in yellow and the capsule in blue. FIGURE 2-3 Typical asymptomatic stage 1 hemangioma at L2 found incidentally. The continuous bone rim indicates that the lesion is not growing. FIGURE 2-4 Illustration of a so-called active stage 2 benign tumor. The red line indicates the pseudocapsule. FIGURE 2-5 CT scan of a stage 2 osteoblastoma. This small painful lesion is bordered by an osteosclerotic pseudocapsule. FIGURE 2-6 Illustration of a stage 3 benign aggressive tumor. The tumor is yellow, the thin and discontinuous blue line is the capsule, and the red line is the pseudocapsule. Note the extracompartmental growth. FIGURE 2-7 (A) Benign aggressive stage 3 hemangioendothelioma at T9. This osteolytic tumor of the vertebral body invades the spinal canal and has fuzzy margins. Treatment would include a vertebrectomy to achieve an en bloc tumor-free margin resection of the vertebral body. Because layer A has not been invaded, the techniques described by Roy Camille et al14 and Tomita et al18 are oncologically acceptable. (B) En bloc resection through a posterior approach. Tumor-free margins were confirmed histologically. Low-grade malignant tumors (Fig. 2-8) are included in stage I and subdivided into IA (the tumor remains inside the vertebra) and IB (tumor invades the paravertebral compartments). No true capsule is associated with these lesions, but a thick pseudocapsule of reactive tissue is permeated by small microscopic islands of tumor. A resection performed along the pseudocapsule often leaves residual foci of active tumor. Megavoltage radiation or proton-beam therapy can be added to reduce the risk of recurrence.29 The treatment of choice, if feasible, is a wide resection (Fig. 2-9). High-grade malignancies are defined as IIA and IIB (Fig. 10A). The neoplastic growth is so rapid that the host has no time to form a continuous reactive tissue. There is continuous seeding with neoplastic nodules (satellites). Moreover, these tumors can have neoplastic nodules at some distance from the main tumor mass (skip metastases). On plain radiographs, these malignancies appear as radiolucent and destructive. Many cases are associated with a pathologic fracture. Invasion of the epidural space is rapid in stage B, particularly in small-cell tumors (Ewing’s sarcomas, lymphomas). They are characterized by semifluid tissue and can occupy the epidural space after infiltrating the cortical border of the vertebra. FIGURE 2-8 (A) Illustration of a stage IA low-grade, intracompartmental malignant lesion. Tumor is represented in yellow, the capsule in blue, and the pseudocapsule in red. A small tumoral island is found within the pseudocapsule. (B) Illustration of a stage IB low-grade, extracompartmental malignant lesion. Tumor is represented in yellow, the capsule in blue, and the pseudocapsule in red. A small tumoral island is found within the pseudocapsule. The tumor is expanding outside the compartment of origin. The margin of the resection must be wide (a “radical” margin in the spine is impossible), and courses of radiation and chemotherapy (according to tumor type) must be considered for local control and to avoid distant metastasis. Anatomic constraints, however, can preclude an appropriate margin (Fig. 2-11). Stages IIIA and IIIB describe the same lesions as IIA and IIB but are associated with distant metastasis. FIGURE 2-9 (A) Stage 1B chordoma at L5. (B) Transverse section of the specimen after en bloc resection. Macroscopic analysis showed a continuous shell of healthy tissue around the mass. The margin can be defined as “wide” except in the epidural area (arrow). (C) Posterior view of specimen. The yellow dot is the longitudinal ligament covering the tumor (“marginal” margin). (D) Histologic slide showing a wide margin. Black dot = tumor; white arrow = normal bone. (E) Histologic slide showing “marginal” margin at the epidural space. Black dot = tumor; white arrow = longitudinal ligament. FIGURE 2-10 (A) Stage IIA high-grade, intracompartmental malignant lesion. Tumor is represented in yellow, the capsule is absent, and the pseudocapsule is in red, infiltrated by tumor. (B) Stage IIB high-grade, extracompartmental malignant lesion. Tumor is represented in yellow, the capsule is absent, and the pseudocapsules in red, infiltrated by tumor. The tumor grows outside the compartment, and a small island of tumor can be found far from the main tumor mass (skip metastasis). FIGURE 2-11 (A) Axial CT of an osteosarcoma at T5 shows tumor growth in layer A (white arrow) and layer D (black arrow) without interrupting the cortex. (B) Transverse section of the specimen after en bloc resection. The margin is intralesional in the layer D (white arrow). One year later the patient developed a local recurrence and distal bone and lung metastases. To apply this grading system correctly, a complete preoperative evaluation must be performed. The first attempt at staging primary spinal tumors for surgical purposes was introduced by Weinstein and McLain8 and subsequently modified in collaboration with the Rizzoli Institute.22,30 Weinstein described a systematic method for identifying each lesion. This staging system, now called the WBB system (Weinstein, Boriani, Biagini; Fig. 2-12), has since undergone several clinical evaluations.23–26 In the transverse plane, the WBB staging system22,30 divides the vertebra into 12 radiating zones (numbered 1 to 12 in a clockwise order) and into five layers (A to E from prevertebral to dural involvement). The longitudinal extent of the tumor is recorded by identifying the specific vertebrae involved. This system (Fig. 2-12) allows a rational approach to surgical planning, provided that surgery is performed along the required margins. The basic concept, a clock face with radiating zones, allows the crucial planning necessary to perform en bloc resections in the spinal cord and cauda equina around the longitudinal axis of the involved vertebra. To spare the spinal cord or cauda equina, each sector of the vertebra must be resected along the proposed planes (e.g., it is impossible to perform an en bloc resection of a vertebral body without first removing the posterior arch; the margins are the pedicles). Conversely, if the tumor occupies an eccentric area, the surgeon is often compelled to remove the healthy contralateral portion of the vertebra, to dislocate the dural sac, and to perform the bony resection of the margins using the radiating sections of the clock face as a guide. The resection is performed with an osteotome and other appropriate instruments. FIGURE 2-12 WBB (Weinstein-Boriani-Biagini) surgical staging system. The transverse extension of the vertebral tumor is described with reference to 12 radiating zones or “sectors” (numbered 1 to 12 in clockwise order) and to five concentric “layers” (Ato E from prevertebral to dural involvement). The longitudinal extent of the tumor is recorded too. So-called eosinophilic granulomas are benign, mostly self-limiting neoplasms that destroy bone. They are the solitary occurrence of X-histiocytosis (Langerhans cell histiocytosis). This condition usually occurs in children less than 10 years old, and the vertebrae are involved in 10 to 15% of cases. This disease is associated with a triad of syndromes: (1) isolated bone localizations (eosinophilic granulomas), (2) Hand-Schüller-Christian disease, or (3) Letterer-Siwe disease.31,32 Eosinophilic granulomas are the most benign form of Langerhans cell histiocytosis, and their pathogenesis is not yet clear. These lesions consist of an abnormal proliferation of lipid-containing histocytes from the reticuloendothelial system. Spinal eosinophilic granulomas can affect multiple spinal levels. They often become symptomatic with localized pain but rarely with neurologic deficits. The vertebral body is often involved, particularly the lumbar or thoracic spine, and is usually partially or totally destroyed. The destruction of the bone rarely leads to significant pain, and neurologic symptoms are exceptional. In younger children, the vertebral body collapses rapidly because most of it is involved, resulting in complete flattening or “vertebra plana.” In older children or adolescents, less involvement of the vertebral body can cause a wedge-shaped deformity or punched-out lytic lesions of the vertebral body (Fig. 2-13). Adjacent intervertebral disc spaces are often well maintained. Extraosseous paravertebral soft tissue masses are rare: This finding must raise suspicions of a different diagnosis (e.g., Ewing’s sarcoma).
Oncologic Classification of Vertebral Neoplasms
 Clinical Manifestations
Clinical Manifestations
 Diagnostic Imaging
Diagnostic Imaging
 Biopsy
Biopsy
 Terminology
Terminology
 Oncologic Staging
Oncologic Staging
Benign Tumors
Malignant Tumors
 Surgical Staging
Surgical Staging
 Types of Benign Tumors
Types of Benign Tumors
Eosinophilic Granulomas (Langerhans Cell Histiocytosis)
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree