Fig. 12.1
(a) (surface histogram) The depression of RIII (“nociceptive”) flexion reflexes induced by SCS in a 35-year-old woman with electrodes at T10 level. Each trace is the rectified average of five single responses recorded at 15-s intervals. (b) Note that RIII was strongly depressed during stimulation (1) and regained basal values almost immediately after end of stimulation (2)
12.3 Material and Technique
There are two methods to implant electrode leads: percutaneous and open surgery. Different models of electrodes can be chosen according to clinical situations (Fig. 12.2). After multi-step assessment, the selected patients are operated on either through an open interlaminar approach or percutaneously.


Fig. 12.2
Electrodes for percutaneous and surgical approaches. Left: three different models of percutaneous electrodes with four contacts. Center: Specify 5-6-5 with 16 contacts for lumbar pain and hinged 2 × 4 electrode for radicular pain. Right: percutaneous electrodes with eight contacts. Different sizes and distances between contacts (©Medtronic France, 2013)
12.3.1 Percutaneous Stimulation Test and Implantation
The patient is placed in prone position on radiotransparent table. The epidural space is entered with a Tuohy needle. Under fluoroscopic control, the electrode is pushed through the needle to cranial direction in the posterior epidural space. The electrode is connected to an external stimulator. The best location for the electrode is at the upper level of the spinal cord segments corresponding to the painful territory. For lower thoracic positioning of the electrode (Fig. 12.3), percutaneous technique is preferred, with an entry point to the epidural space distal to the conus medullaris to lessen neurological risk. The percutaneous technique has the advantage to insert and place the electrode under local anesthesia. The most effective location is the one which induces evoked paresthesias to stimulation in the whole painful territory. The Tuohy needle is then withdrawn and the electrode is fixed to the subcutaneous tissue at the site of puncture. The electrode is switched to an external stimulator by a temporary cable tunneled distally. This temporary system is used for testing, for 3 days on average, before decision. If pain relief is obtained, the temporary cable is removed and the pulse generator is implanted under general anesthesia. If there is no evidence of pain relief the electrode is removed.
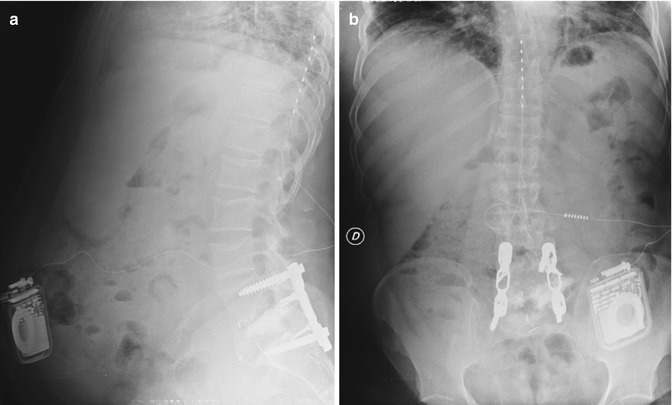

Fig. 12.3
SCS at T8–T9 level in a 39-year-old man with left sciatic and lumbar pain following twice lumbar disk surgery with no evidence of lumbar disk recurrence on CT. Pain was resistant to all medical treatments and transcutaneous electrical nerve stimulation. Patient had 75 % sciatic and 75 % lumbar pain relief after SCS. Implantation was using percutaneous technique (a) profile X-ray, (b) front X-ray
12.3.2 Surgical Implantation of the Spinal Cord System
Open implantation requires general anesthesia. The patient is placed in the prone position. For high thoracic and cervical positioning of the electrode for pain in upper limb (Fig. 12.4), an open surgical unilateral interlaminar approach is preferable. As a matter of fact the percutaneous technique at these levels would be less safe; it must only be reserved to very experienced hands. For pain in lower limbs, especially when lumbar pain is associated, midline interlaminar approach between T10–T11 and electrode placement in front of T8 vertebral body are favored, whatever percutaneous or open implantation (Fig. 12.5). In case of open surgery, under general anesthesia, absence of postoperative induced paresthesias or partial covering of the pain territory necessitate electrode repositioning. In all circumstances the best location of the electrode is at the upper level of the spinal cord segments corresponding to the painful territory.
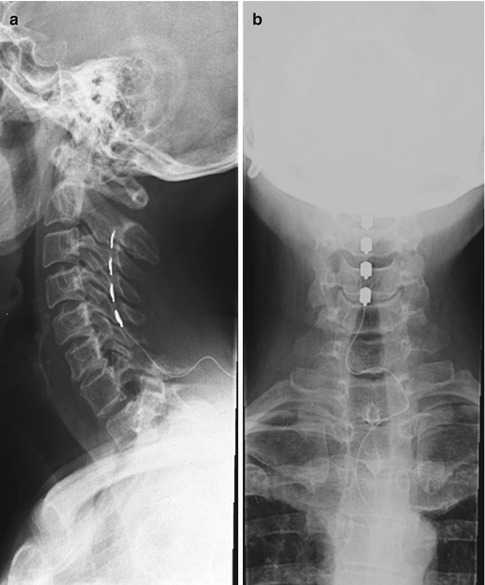
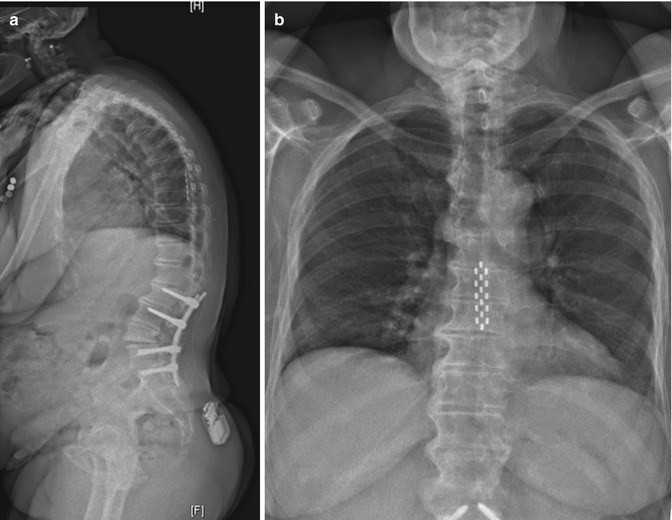

Fig. 12.4
Open interlaminar approach for a C5–C6 right insertion of a Resume TL (Medtronic) quadripolar electrode (a) profile X-ray, (b) front X-ray

Fig. 12.5
Specify 5-6-5 lead (Medtronic) at T8 level for failed back surgery syndrome including lumbar pain and radicular pain. (a) profile X-ray, (b) front X-ray
12.4 Indications, Selection of Patients, and Result
12.4.1 Neuropathic Pain
Neuropathic pain is defined as “the pain associated with primary injury of neural tissues – peripheral (peripheral nerves or roots) or central (spinal cord or brain)” (Gybels and Sweet 1989) .
Mechanism of Action of Stimulation
The neurophysiological effect of electrostimulation is most likely due to activation of the large primary afferents through stimulation of the dorsal columns (Burton 1975; Brown et al. 1973; Handwerker et al. 1975; Lindblom et al. 1977). Before considering indication for stimulation one must be sure that enough dorsal column fibers up to the brainstem are present and functionally valid. In a retrospective study of our series correlating the long-term effect of SCS on pain and the anatomical location of the pathological lesion, we found that interruption of primary afferent neurons at the radicular level, centrally to the dorsal root ganglion or at the spinal cord level, was associated with a high rate of failure of SCS (Keravel and Sindou 1985) (Fig. 12.6).
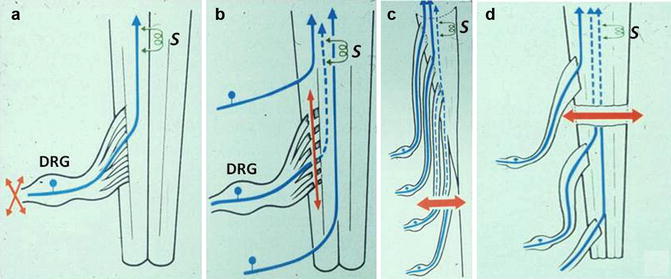

Fig. 12.6
Anatomical bases for SCS: functionality of the dorsal column fibers. When the lesion (orange arrows) is at the peripheral nerve level or at the radicular level distally to the dorsal root ganglion (DRG), axons going up to the brainstem sensory relay nuclei, through the dorsal columns, are intact (a). When the lesion interrupts the fibers centrally to the DRG, as for instance in brachial plexus avulsion (b), the dorsal column fibers had degenerated (dotted blue lines) up to the brainstem. When a lesion damaged the root centrally to the DRG at the lumbo-sacral level, as does a lumbar disc herniation not removed early enough, for instance, fibers had centrally degenerated (c). Same applies for spinal cord lesions (d). If dorsal column fibers are totally interrupted, SCS cannot be effective. Consideration of the location of the causal lesion, together with the degree of completeness of interruption of the dorsal column fibers, is of prime importance to predict effectiveness of SCS (S)
Indications
According to literature, neuropathic pain syndromes for which SCS can be helpful include: persistent pain due to radiculopathies in the frame of the so-called failed back surgery syndrome (FBSS), pain after peripheral nerve lesions, and post-amputation pain. SCS may be also indicated in diabetic neuropathy refractory to medical treatment except when autonomic neuropathy is present (Tesfaye et al. 1996; Kumar et al. 1996; Siegfried and Lazorthes 1982; Turner et al. 1995; Krainick et al. 1980).
Selection of Patients
Clinical and imaging examinations should determine whether the dorsal column fibers between the dorsal root ganglion (DRG) and the cuneate gracilis relay nuclei of brainstem are intact. The Somatosensory evoked potentials (SSEPs) examination is a valuable tool to assess their functional status (Mertens et al. 1992). Dorsal column function was assessed by measuring the central conduction time (CCT), namely the N13–N20 interval for the upper limb and the N22–P39 interval for the lower limb (Fig. 12.7). Sindou et al. (2003) established the value of SSEPs for predicting the long-lasting effects on pain after SCS. When preoperative CCT was significantly abnormal, the success rate of SCS, defined as at least 50 % long-term pain relief, was nil, whereas it was 75.4 % in patients with normal preoperative CCT (Table 12.1). Authors concluded that SSEPs are a useful tool for preoperative patient selection, and that if CCT is abolished or significantly altered, as seen in the presented illustrative case, the patient should not undergo SCS (Fig. 12.8).
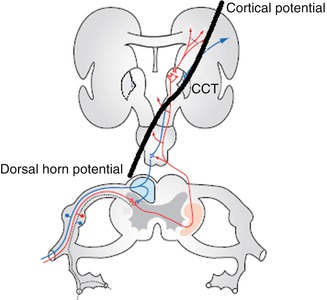
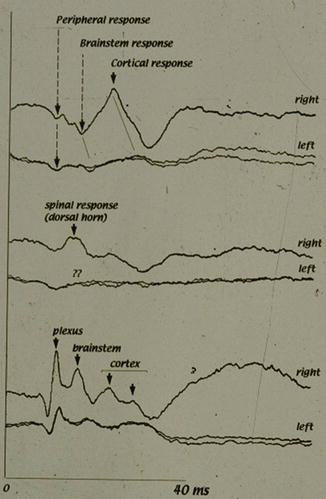
Table 12.1
Pain relief, according to preoperative CCT
Pre-operative CCT↓ | Post-op pain relief ≥50 % | Post-op pain relief <50 % |
|---|---|---|
Normal CCT (n = 111) | 93 (84 %) | 18 (16 %) |
Abnormal CCT (n = 32) | 0 (0 %) | 32 (100 %) |
Total (n = 143) | 93 (65 %) | 50 (35 %) |

Fig. 12.7
Assessment of neural conduction in the dorsal columns with the use of somatosensory evoked potentials (SSEPs). The central conduction time (CCT) measures the time from dorsal horn potential and cortical potential

Fig 12.8
Somato-sensory evoked potentials to median nerve stimulation in a patient with left brachial plexus avulsion; main locus of impaired transmission was central to the dorsal ganglion. In such case SCS would not be effective, according to authors’ experience
Results
In FBSS, results are better when pain is radicular and anatomically limited rather than diffuse, and when unilateral rather than bilateral. For FBSS, pain relief greater than 50 % is obtained in 60–75 % of the patients. North et al. (1993) found 52 % of patients with more than 50 % pain reduction at 7 years of follow-up. Low back pain in FBSS was considered in early series as a poor prognostic factor for SCS. However a high positioning of the electrode, between the seventh and tenth thoracic vertebrae, seems to give good results in at least 50 % of the patients so implanted. SCS can also be indicated in diabetic neuropathies refractory to medical treatment. In contrast, post-herpetic pain and intercostal neuralgias seem to respond less favorably.
12.4.2 CRPS
CRPS is a regional pain that affects the foot and the hand, which appears after surgery, trauma or even after immobilization. Excruciating pain results in functional impairment and trophic changes. When there is no evidence of nerve injury, the condition is defined as CRPS type I. When pain is associated with nerve injury, the condition is referred to as CRPS type II (Causalgia) (Stanton-Hicks 2006). Diagnostic criteria were established by International Association for the Study of Pain (Merskey and Bogduk 1994) (Table 12.2).
Table 12.2
Diagnostic criteria for CRPS-I and CRPS-II, according to International Association for the Study of Pain
CRPS-I (reflex sympathetic dystrophy) | CRPS-II (Causalgia) |
|---|---|
1. The presence of an initiating noxious event, or a cause of immobilization | 1. The presence of continuing pain, allodynia, or hyperalgesia after a nerve injury, not necessarily limited to the distribution of the injured nerve |
2. Continuing pain, allodynia, or hyperalgesia with which the pain is disproportionate to any inciting event | 2. Evidence at some time of edema, changes in skin blood flow, or abnormal sudomotor activity in the region of the pain |
3. Evidence at some time of edema, changes in skin blood flow, or abnormal sudomotor activity in the region of the pain | 3. This diagnosis is excluded by the existence of conditions that would otherwise account for the degree of pain and dysfunction |
4. This diagnosis is excluded by the existence of conditions that would otherwise account for the degree of pain and dysfunction
Stay updated, free articles. Join our Telegram channel
Full access? Get Clinical Tree
 Get Clinical Tree app for offline access
Get Clinical Tree app for offline access

|




