Fig. 7.1
Postoperative radiograph example of subthalamic deep brain stimulation (STN-DBS). (a, b) Skull X-ray demonstrates electrode leads for STN-DBS (white arrow). (c) Chest X-ray demonstrates implantable pulse generators in the bilateral anterior chest (black arrowhead)
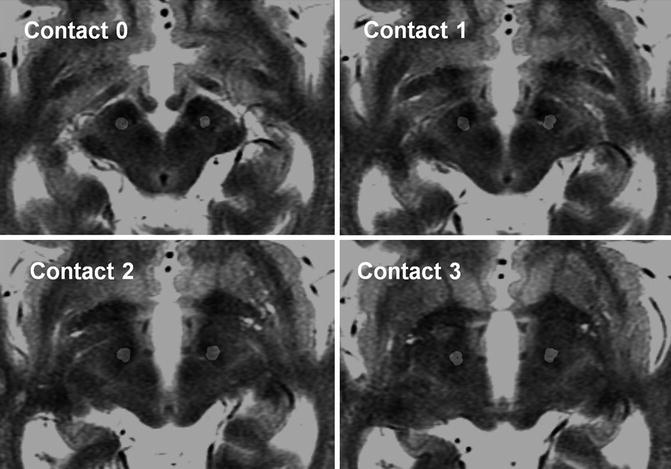
Fig. 7.2
Postoperative CT-preoperative MRI fusioned image shows artifacts of the electrodes placed within the STN
7.5 DBS Control After Surgery
After surgery, signals can be sent to the stimulation device from outside the body using a dedicated programmer. This allows changes to be made to the stimulation conditions and sites. Adjustments are necessary so that benefits can be optimized. The electrode position must be known when programming DBS, and intraoperative data and postoperative images are useful for electrode selection. Furthermore, thorough assessment of both the stimulus and adverse effects of all electrodes during initial programming makes subsequent electrode adjustments easier (Bronstein et al. 2011; Volkmann et al. 2006). Stimulating from the center of the STN to the dorsolateral area (contact 1 or 2) is effective for the cardinal symptoms of PD. An immediate effect is seen on rigidity as a result of stimulation, followed by a slightly delayed effect on tremors. The effect on bradykinesia is more individualized and is often observed over time. Stimulation is often initiated days or weeks after surgery because of unstable symptoms caused by postoperative drug adjustments, changes in resistance levels caused by environmental changes around the electrode, and micro-lesion effects caused by the surgical procedure.
Recent improvements in stimulation devices have enabled constant current stimulation (Okun et al. 2012) from the early postoperative period. After initiation of stimulation, its conditions (frequency, pulse width, stimulation intensity) are adjusted to eliminate bilateral symptom differences and gradually increase intensity with the aim of improving the off-period condition (Volkmann et al. 2006). Drugs are then tapered as appropriate according to dyskinesia. Some advanced PD patients have a very narrow therapeutic window for levodopa. These patients can be in an off-period condition during the daytime, with constant dyskinesia during the on-period. In some patients, the STN stimulation itself causes further exacerbation of dyskinesia.
When dyskinesia makes it difficult to adjust STN-DBS stimulation, control can be achieved by the selection of electrodes with a narrow distance between poles in active contact or by electrode configuration that enables direct stimulus application to the pallidofugal fibers outside and above the STN (Katayama et al. 2006). Some patients require regular stimulus frequency changes after surgery, but by 3–6 months, the need for further adjustment should be minimal.
Decreasing the amount of drug intake is often possible after STN-DBS initiation in many subjects. However, adjustments must carefully maintain the delicate balance between drug therapy and DBS. Dopaminergic medications should be tapered gradually and should not be excessively tapered at an early stage to avoid depression or apathy (Czernecki et al. 2008; Thobois et al. 2010). Witt et al. (2006) assessed the effects of the single levodopa challenge test and STN-DBS on depression and hedonic tone and found that both the single levodopa challenge test and STN-DBS ameliorated depression, whereas only levodopa ameliorated hedonic symptoms. This suggests that the excessive postoperative tapering of dopaminergic medication may cause anhedonia. Okun et al. (2009) performed the Cognition and Mood in Parkinson’s disease in STN versus GPi-DBS (COMPARE) trial and reported that subjects became confused, less energetic, less euphoric, and sad when the ventral STN was stimulated. Therefore, if these symptoms appear, the stimulation site should be changed to a more dorsolateral region.
7.6 Surgical Outcomes of STN-DBS
7.6.1 Effectiveness of STN-DBS
STN-DBS ameliorates the cardinal symptoms of PD, including bradykinesia, rigidity, and tremors, and activities of daily living (ADL). It primarily ameliorates motor symptoms and ADL during drug off-periods, shortens drug off-periods, allows dose tapering, and ameliorates dyskinesia, and improves QOL (Limousin et al. 1998; Kumar et al. 1998, 1999; Burchiel et al. 1999; Moro et al. 1999; Molinuevo et al. 2000; Deep-Brain Stimulation for Parkinson’s Disease Study Group 2001; Pinter et al. 1999; Houeto et al. 2000; Katayama et al. 2001; Simuni et al. 2002; Herzog et al. 2003; Kleiner-Fisman et al. 2003; Krack et al. 2003; Rodriguez-Oroz et al. 2004). Kleiner-Fisman et al. (2006) performed a systematic review of 37 cohort studies including 921 cases and reported that UPDRS ADL scores (UPDRS-II) and motor scores (UPDRS-III) improved by 50 and 52 %, respectively, when comparing postoperative off-periods and stimulation on-periods and preoperative drug off-periods. Levodopa dosage, dyskinesia, and daytime off-periods decreased by 55.9, 69.1, and 68.2 %, respectively. A 34.5 % improvement was observed in the PD questionnaire (PDQ-39).
Till date, three randomized controlled trials have compared drug therapy alone with STN-DBS for advanced PD. Deuschl et al. (2006) divided 156 PD patients aged ≥75 years into an STN-DBS group (n = 78) and a drug therapy alone group (n = 78) and compared their prognoses after 6 months using PDQ-39 and UPDRS-III. These outcomes significantly improved in the STN-DBS group compared with those in the drug therapy alone group. Weaver et al. (2009) divided 255 PD patients into a DBS group [STN (n = 60) or GPi (n = 61)] and a drug therapy alone group (n = 134) and compared symptoms during the on-period after 6 months. Their results revealed improved on-periods without dyskinesia or motor function problems and improved QOL in the DBS group. However, adverse effects associated with the surgical procedure were observed, with serious adverse effects being more common in the DBS group. These studies observed STN-DBS over short 6-month periods, whereas the PD-SURG trial, a large randomized trial evaluating the role of surgery as therapy for PD, compared treatment outcomes and complications after 1 year. Williams et al. (2010) allocated 366 PD patients to surgery (n = 183; STN-DBS group: 174 patients) or drug therapy alone (n = 183) and assessed their PDQ-39 scores after 1 year (PD-SURG trial). Improvements in the PDQ-39 summary index, mobility domain, ADL domain, and bodily discomfort domain were significantly greater in the STN-DBS group than in the drug therapy alone group. Complications associated with surgery were observed in 36 patients, with one procedure-related death. Twenty patients in the STN-DBS group and 13 in the drug therapy group experienced serious adverse effects associated with PD. The results indicated that STN-DBS was more effective than drug therapy alone.
7.6.2 The Characteristics of STN-DBS
The primary effect of STN-DBS is a significant decrease in the off-period duration. The treatment also affects the on-period, although less significantly. In patients with marked motor fluctuations with a good preoperative on-period state, the good state can be maintained after DBS (see Fig. 7.3, Case A). Conversely, in patients with advanced PD who already exhibit a poor response to levodopa, good outcomes are unlikely despite a slightly improved response to DBS (see Fig. 7.3, Case B). This applies equally to the long-term, where off-period improvements are maintained for a comparatively long period. However, even if an improvement is seen in the on-period, levels quickly return to their preoperative state, and there is often a subsequent exacerbation (Deuschl et al. 2006; Rodriguez-Oroz et al. 2005; Moro et al. 2010).
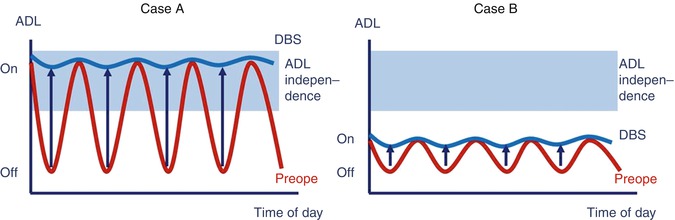

Fig. 7.3
Illustration of improvement of motor fluctuation in activities of daily living in the patients treated with STN-DBS
Levodopa dosage or the levodopa-equivalent dosage can be decreased after STN-DBS (Tavella et al. 2002; Thobois et al. 2002; Vingerhoets et al. 2002). While the extent of dose reduction varies among reports, most state that STN-DBS can decrease the dosage of anti-PD drugs. However, this effect is not significant for GPi-DBS (Scotto di Luzio et al. 2001; Volkmann et al. 2001; Anderson et al. 2005; Minguez-Castellanos et al. 2005; Weaver et al. 2005; Valldeoriola et al. 2002). In addition, amelioration of levodopa-induced dyskinesia can be expected, which is thought to be due to decreased dyskinesia resulting from anti-PD dose reductions (Deuschl et al. 2006; Anderson et al. 2005; Weaver et al. 2005; Valldeoriola et al. 2002). A direct effect has also been reported (Katayama et al. 2006).
7.6.3 The Long-Term Effect of STN-DBS
Several prospective studies with 3- to 5-year follow-up periods have examined the long-term effects of STN-DBS. Sustained improvements are observed in ADL, motor symptoms, rigidity, bradykinesia, and dose reductions (see Fig. 7.4) (Krack et al. 2003; Rodriguez-Oroz et al. 2005; Schüpbach et al. 2005; Østergaard and Aa Sunde 2006; Volkmann et al. 2009; Moro et al. 2010). The results of 8- or 10-year follow-ups have also been reported, and while tremors remain controlled in the long-term, there appears to be a decline in the effect on axial symptoms (Fasano et al. 2010; Castrioto et al. 2011).
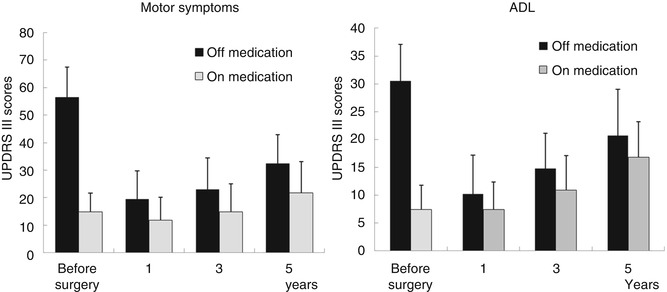

Fig. 7.4
Long-term effects of STN-DBS in motor UPDRS III subscore and ADL UPDRS II subscore in 132 PD patients who underwent bilateral STN-DBS in our institution. Significant improvement of off-medication state sustained up to 5 years after surgery
7.6.4 Symptoms for Which DBS Is Inadequate
DBS tends to be inadequate in patients with levodopa-resistant symptoms such as weakness, dysarthria, freezing of gait, and postural instability. Axial symptoms (particularly in the on-period) can be difficult to control. STN-DBS mainly ameliorates motor symptoms during the off-period (Deuschl et al. 2006; Anderson et al. 2005; Weaver et al. 2005), and axial symptoms such as postural instability and freezing of gait observed in the preoperative on-period often fail to ameliorate after surgery and appear during long-term follow-up of STN-DBS therapy (Welter et al. 2002; Tsai et al. 2009; Davis et al. 2006).
In a meta-analysis by St George at al. that was based on 11 articles, postural instability and gait disturbance deteriorated within 2 years, regardless of postoperative improvement, and tended to fall below preoperative levels (St George et al. 2010). In a 7-year follow-up reported by Muniz et al. (2010), freezing of gait (on initiation of walking) occurred more readily in the STN-DBS group than in the drug therapy alone group. In a study that examined the long-term effects of STN-DBS over 8–10 years, axial symptoms were the first to deteriorate (Fasano et al. 2010; Castrioto et al. 2011). Axial symptoms such as freezing and postural instability and related gait disturbance are motor symptoms that are difficult to control with STN-DBS, and their management is a topic for future study.
7.7 Complications of STN-DBS
DBS-induced adverse effects usually disappear when stimulation is discontinued, and they can be controlled by changing the stimulation conditions. Nevertheless, device-related issues such as disconnection or complications such as infection can occur. The most serious complication is ICH, which occurs in 3.9 % patients according to a systematic review by Kleiner-Fisman et al. (2006). Deuschl et al. (2006) reported that while life-threatening adverse effects such as ICH were more common with DBS than with drug therapy, overall treatment-related adverse effects were more common with drug therapy alone.
Previous studies have reported minor or transient complications related to cognitive and affective function that rarely affect ADL in a significant manner (Saint-Cyr et al. 2000; Kumar et al. 1999; Moro et al. 1999; Molinuevo et al. 2000; Pillon et al. 2000; Woods et al. 2002; Funkiewiez et al. 2004; Morrison et al. 2004). However, cognitive and behavioral dysfunction and psychiatric symptoms have recently been highlighted as consequences of STN-DBS. In a systematic review of 1,398 patients from 82 articles by Temel et al. (2006), cognitive dysfunction occurred in 41 % patients, depression in 8%, and hypomania in 4 %. In addition, a meta-analysis by Parsons et al. (2006) that was based on 612 patients from 28 cohort studies revealed that while STN-DBS with appropriate patient selection is a safe therapy in terms of cognitive function, a mild decline in executive function and verbal learning was observed along with a relatively marked decline in semantic and phonemic verbal fluency.
Many studies examining declining cognitive function have noted that a decline in verbal fluency is prominent and that attention should be paid to deteriorating language skills (Okun et al. 2009; Gironell et al. 2003; Smeding et al. 2006). Unfortunately, studies on cognitive function have concluded that it is difficult to distinguish a decline in cognitive function caused by STN-DBS from that caused by PD progression, making randomized controlled trials difficult. Witt et al. allocated patients either to STN-DBS (n = 60) or drug therapy alone (n = 63) and tested cognitive function after 6 months. While overall cognitive function scores did not decrease, scores for verbal fluency, the Stroop test, and executive function decreased after STN-DBS. More importantly, QOL improved in patients who experienced a decline in executive function after STN-DBS, which mitigated any clinical decrease in ADL after treatment (Witt et al. 2008). Subscores for elderly individuals, high doses of levodopa, and axial symptoms with high UPDRS scores are risk factors for deterioration in executive function after STN-DBS. However, these preoperative risk factors may reflect the degree of disease progression. Other factors, including surgical procedure, correct electrode placement, and postoperative management may also be involved in the deterioration of executive function after STN-DBS.
In order to examine whether the electrode trajectory affects changes in cognitive function, Witt et al. (2013) divided patients into an STN-DBS group (n = 31) and a drug therapy alone group (n = 31) and examined the association between changes in cognitive function and electrode entry point, region transected, and site of active contact within the STN. An electrode trajectory passing through the head of the caudate nucleus caused an overall cognitive decline as well as a decline in working memory. A site of active contact that deviated from within the STN resulted in a decline in semantic verbal fluency. Therefore, the electrode path should avoid the head of the caudate nucleus and the electrode should be placed correctly within the STN; together, this can decrease the risk of cognitive decline following STN-DBS.
Depression after STN-DBS is thought to occur in approximately 20–25% patients. However, in a review of 22 articles, Takeshita et al. (2005) found the incidence of postoperative depression to be 2–33.3 % and that of mania to be 4.2–8.1 %, with STN-DBS having an antidepressant effect in 16.7–76 % patients. Overall, they concluded that the depression scores remained unchanged. In two randomized controlled trials, the STN-DBS group experienced a slight antidepressant effect at 6 months compared with the drug therapy alone group. Postoperative risk factors for depression were identified to be an excessive tapering of dopaminergic medication and a history of depression (Weaver et al. 2009; Witt et al. 2008).
Some reports on suicides by patients who underwent STN-DBS have been published (Burkhard et al. 2004; Soulas et al. 2008). A review of 5,311 patients at 75 facilities by Voon et al. found the rate of successful suicide to be 0.45% and the rate of unsuccessful suicide to be 0.90 %. The incidence in the first year after surgery was significantly high and correlated with postoperative depression (Voon et al. 2008). Rodrigues et al. (2010) retrospectively examined suicide cases and reported levodopa dose tapering to be a significant factor.
Mood and impulsiveness are also thought to be involved in the limbic part of the STN, and stimulation of this region by STN-DBS may result in changes in mood and behavior. During the initial stage of STN-DBS, dopaminergic medication and stimulus adjustment are often difficult to balance and may increase mood-related complications. Therefore, excessive dose tapering in the early postoperative period is a significant risk factor, and drug and DBS adjustments must be appropriate, tolerable, and monitored.
7.8 Comparison of DBS Targets
DBS targeting the STN or GPi is a common treatment for motor fluctuations and dyskinesia associated with advanced PD. Several recent reports have seen little difference in the effectiveness of STN-DBS and GPi-DBS in ameliorating motor symptoms (Okun et al. 2009; Rodriguez-Oroz et al. 2005; Moro et al. 2010; Anderson et al. 2005; Follett et al. 2010). Weaver et al. (2005) conducted a meta-analysis of 31 papers on STN-DBS and 14 papers on GPi-DBS and concluded that motor function improvement (UPDRS-III) after 6 months was 54 % with STN-DBS and 40 % with GPi-DBS, indicating no significant difference between groups. ADL also improved by 40 % in both groups, although no significant difference was observed. While GPi-DBS ameliorated both the on-period and off-period symptoms, STN-DBS only ameliorated the off-period symptoms. In contrast, the drug dosage was tapered by 52 % with STN-DBS, whereas no tapering was possible with GPi-DBS. In a number of randomized controlled trials, improvements in motor function and ADL were comparable. However, adverse effects such as cognitive and mental dysfunction occurred more commonly with STN-DBS and rarely with GPi-DBS. Drug tapering was possible in these trials with STN-DBS, but not with GPi-DBS (Okun et al. 2009; Rodriguez-Oroz et al. 2005; Moro et al. 2010; Anderson et al. 2005; Follett et al. 2010).
The randomized controlled COMPARE trial compared the effectiveness of STN-DBS and GPi-DBS in ameliorating nonmotor symptoms and revealed a significant decline in verbal fluency and mental function (such as mood and cognitive function) in the STN-DBS group compared with that in the GPi-DBS group (Okun et al. 2009). Follett et al. (2010) allocated 299 patients to either an STN-DBS group (n = 147) or a GPi-DBS group (n = 152) and examined UPDRS-III score, self-reported function, QOL, neurocognitive function, and adverse events after 2 years. No difference in UPDRS-III score was observed between the two groups. Drug dosage was significantly tapered in the STN-DBS group compared with that in the GPi-DBS group; however, a decline in visuomotor processing speed was observed in the former group. Furthermore, depression was ameliorated in the GPi-DBS group but exacerbated in the STN-DBS group, and serious adverse effects were generally the same in both groups. Motor symptoms were also ameliorated by both STN-DBS and GPi-DBS, with individual differences in nonmotor function. Till date, the overall improvement rates for motor symptoms in randomized controlled trials have been 26–48 % with STN-DBS and 16.9–37 % with GPi-DBS. Therefore, the rate of amelioration of motor symptoms after STN-DBS tended to be lower in these trials.
In the recent Netherlands Subthalamic and Pallidal Stimulation (NSTAPS) study, patients were allocated either to an STN-DBS group (n = 63) or a GPi-DBS group (n = 65), and the weighted Academic Medical Center Linear Disability Scale (ALDS), cognitive function, mood, effects on behavior, UPDRS III score, QOL, adverse effects, and drug usage were examined after 1 year. No difference in weighted ALDS score was observed between the two groups. However, the off-period symptom improvement rate was 45.7 % in the STN-DBS group compared with 26 % in the GPi-DBS group, indicating a significant improvement. Compared with that in the GPi-DBS group, a significant tapering of drug dosage was possible in the STN-DBS group, resulting in significant amelioration of off-period symptoms as per ALDS scores and posture and gait as per UPDRS scores. In addition, there were no differences in cognitive, mood, and behavioral adverse effects between groups (Odekerken et al. 2013). Tapering of drug dosage was again possible in the STN-DBS group, which led to amelioration of levodopa-induced dyskinesia. In contrast, dose tapering was not possible in the GPi-DBS group, although a direct inhibitory effect on levodopa-induced dyskinesia was observed.
Stimulation of the ventral intermediate nucleus of the thalamus (Vim) has no effect on motor fluctuation and dyskinesia in patients with advanced PD, but it has a strong inhibitory effect on drug therapy-resistant tremors (Schuurman et al. 2000, 2008). Individualized target selection based on these differences in clinical efficacy is necessary.
7.9 Conclusions
DBS is indicated for advanced PD patients with motor fluctuations and dyskinesia whose symptoms are difficult to control with drugs or who suffer from drug therapy-resistant tremors. Further improvements in the safety and adjustability of DBS can be expected from future advances in DBS devices. Several crucial factors need to be considered to achieve favorable treatment outcomes: careful patient selection, optimally timed treatment initiation, technically correct surgical procedures, careful management of postoperative drugs, and controlled adjustment of DBS.
References
Anderson VC, Burchiel KJ, Hogarth P et al (2005) Pallidal vs subthalamic nucleus deep brain stimulation in Parkinson disease. Arch Neurol 62(4):554–560PubMedCrossRef
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree







